
Letter from the Chairman
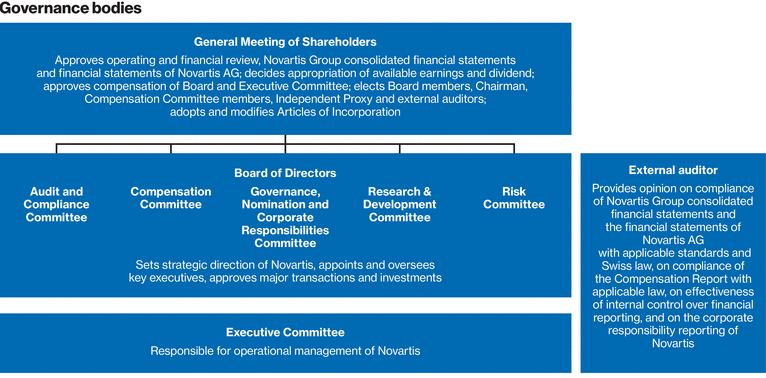
Our corporate governance approach
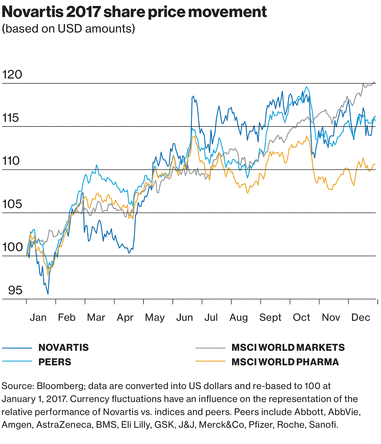
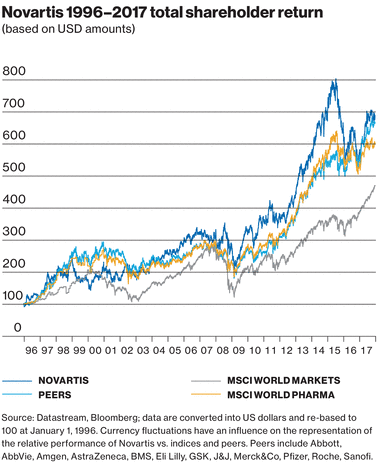
Our shares and our shareholders
|
Number of shares
|
|
||||||||
|
Year |
As of Jan 1 |
Changes
in shares |
As of Dec 31 |
Changes
in CHF |
|||||
|
2015
|
2 706 193 000
|
– 29 200 000
|
2 676 993 000
|
– 14 600 000
|
|||||
|
2016
|
2 676 993 000
|
– 49 878 180
|
2 627 114 820
|
– 24 939 090
|
|||||
|
2017
|
2 627 114 820
|
– 10 270 000
|
2 616 844 820
|
– 5 135 000
|
|||||
|
2017
|
2016
|
2015
|
|||||
|
Issued shares
|
2 616 844 820
|
2 627 114 820
|
2 676 993 000
|
||||
|
Treasury shares 1
|
299 388 321
|
253 055 807
|
303 098 183
|
||||
|
Outstanding shares at December 31
|
2 317 456 499
|
2 374 059 013
|
2 373 894 817
|
||||
|
Weighted average number of shares outstanding
|
2 345 783 843
|
2 378 474 555
|
2 402 806 352
|
||||
|
1 Approximately 131 million treasury shares (2016: 135 million; 2015: 137 million) are held in Novartis entities that restrict their availability for use.
|
|||||||
|
2017
|
2016
|
2015
|
|||||
|
Basic earnings per share (USD) from continuing operations
|
3.28
|
2.82
|
2.92
|
||||
|
Basic earnings per share (USD) from discontinued operations
|
|
|
4.48
|
||||
|
Total basic earnings per share (USD)
|
3.28
|
2.82
|
7.40
|
||||
|
Diluted earnings per share (USD) from continuing operations
|
3.25
|
2.80
|
2.88
|
||||
|
Diluted earnings per share (USD) from discontinued operations
|
|
|
4.41
|
||||
|
Total diluted earnings per share
|
3.25
|
2.80
|
7.29
|
||||
|
Operating cash flow (USD) from continuing operations
|
5.38
|
4.82
|
5.03
|
||||
|
Year-end equity for Novartis AG shareholders (USD)
|
32.00
|
31.52
|
32.46
|
||||
|
Dividend (CHF) 2
|
2.80
|
2.75
|
2.70
|
||||
|
1 Calculated on the weighted average number of shares outstanding, except year-end equity
|
|||||||
|
2 2017: proposal to shareholders for approval at the Annual General Meeting on March 2, 2018
|
|||||||
|
2017
|
2016
|
2015
|
|||||
|
Price/earnings ratio 1
|
25.7
|
25.7
|
11.9
|
||||
|
Price/earnings ratio from continuing operations 1
|
25.7
|
25.7
|
30.1
|
||||
|
Enterprise value/EBITDA from continuing operations
|
15
|
13
|
16
|
||||
|
Dividend yield (%) 1
|
3.4
|
3.7
|
3.1
|
||||
|
1 Based on the Novartis share price at December 31 of each year
|
|||||||
|
2017
|
2016
|
2015
|
|||||
|
Year-end ADR price (USD)
|
83.96
|
72.84
|
86.04
|
||||
|
High 1
|
86.65
|
86.21
|
106.12
|
||||
|
Low 1
|
70.03
|
67.59
|
83.96
|
||||
|
Number of ADRs outstanding 2
|
320 833 039
|
315 349 314
|
299 578 398
|
||||
|
|
|||||||
|
1 Based on the daily closing prices
|
|||||||
|
2 The depositary, JPMorgan Chase Bank, N.A., holds one Novartis AG share for every ADR issued.
|
|||||||
|
2017
|
2016
|
2015
|
|||||
|
Year-end share price
|
82.40
|
74.10
|
86.80
|
||||
|
High 1
|
85.15
|
86.45
|
102.30
|
||||
|
Low 1
|
69.55
|
68.15
|
82.20
|
||||
|
Year-end market capitalization (USD billions) 2
|
195.5
|
172.0
|
208.3
|
||||
|
Year-end market capitalization (CHF billions) 2
|
191.0
|
175.9
|
206.1
|
||||
|
1 Based on the daily closing prices
|
|||||||
|
2 Market capitalization is calculated based on the number of shares outstanding (excluding treasury shares). Market capitalization in USD is based on the market capitalization in CHF converted at the year-end CHF/USD exchange rate.
|
|||||||
|
As of December 31, 2017 |
Number of
registered shareholders |
% of registered share capital |
|||
|
1–100
|
24 970
|
0.06
|
|||
|
101–1'000
|
101 722
|
1.62
|
|||
|
1'001–10'000
|
36 938
|
3.93
|
|||
|
10'001–100'000
|
3 244
|
3.21
|
|||
|
100'001–1'000'000
|
463
|
5.25
|
|||
|
1'000'001–5'000'000
|
72
|
5.58
|
|||
|
5'000'001 or more 1
|
32
|
50.24
|
|||
|
Total registered shareholders/shares
|
167 441
|
69.89
|
|||
|
Unregistered shares
|
|
30.11
|
|||
|
Total
|
|
100.00
|
|||
|
1 Including significant registered shareholders as listed above
|
|||||
|
As of December 31, 2017
|
Shareholders in %
|
Shares in %
|
|||
|
Individual shareholders
|
96.31
|
13.36
|
|||
|
Legal entities 1
|
3.63
|
35.25
|
|||
|
Nominees, fiduciaries and ADS depositary
|
0.06
|
51.39
|
|||
|
Total
|
100.00
|
100.00
|
|||
|
1 Excluding 6.4% of the share capital held as treasury shares by Novartis AG and its subsidiaries
|
|||||
|
As of December 31, 2017
|
Shareholders in %
|
Shares in %
|
|||
|
Belgium
|
0.13
|
3.82
|
|||
|
France
|
2.23
|
0.38
|
|||
|
Germany
|
5.35
|
2.13
|
|||
|
Japan
|
0.18
|
0.71
|
|||
|
Switzerland 1
|
88.42
|
42.56
|
|||
|
United Kingdom
|
0.49
|
22.22
|
|||
|
United States
|
0.34
|
25.82
|
|||
|
Other countries
|
2.86
|
2.36
|
|||
|
Total
|
100.00
|
100.00
|
|||
|
Registered shares held by nominees are shown in the country where the company/affiliate entered in the Novartis Share Register as shareholder has its registered seat.
|
|||||
|
1 Excluding 6.4% of the share capital held as treasury shares by Novartis AG and its subsidiaries
|
|||||
Our Board of Directors
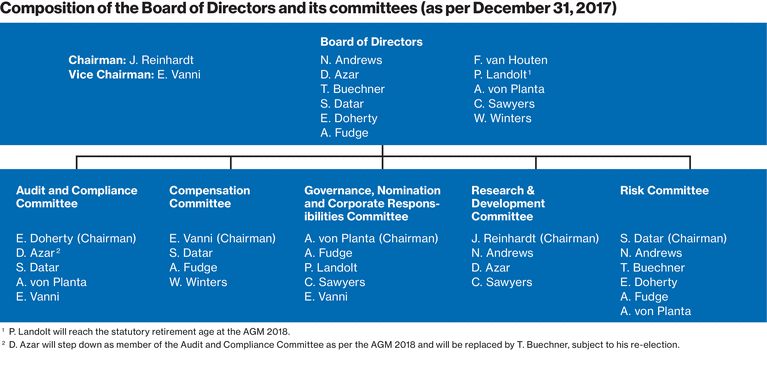
|
Name |
Nationality |
Year of birth |
First election
at AGM |
||||
|
Joerg Reinhardt, Ph.D.
|
D
|
1956
|
2013
|
||||
|
Enrico Vanni, Ph.D.
|
CH
|
1951
|
2011
|
||||
|
Nancy C. Andrews, M.D., Ph.D.
|
US
|
1958
|
2015
|
||||
|
Dimitri Azar, M.D.
|
US
|
1959
|
2012
|
||||
|
Ton Buechner
|
NLD
|
1965
|
2016
|
||||
|
Srikant Datar, Ph.D.
|
US
|
1953
|
2003
|
||||
|
Elizabeth Doherty
|
GB
|
1957
|
2016
|
||||
|
Ann Fudge
|
US
|
1951
|
2008
|
||||
|
Frans van Houten
|
NLD
|
1960
|
2017
|
||||
|
Pierre Landolt, Ph.D.
|
CH
|
1947
|
1996
|
||||
|
Andreas von Planta, Ph.D.
|
CH
|
1955
|
2006
|
||||
|
Charles L. Sawyers, M.D.
|
US
|
1959
|
2013
|
||||
|
William T. Winters
|
GB/US
|
1961
|
2013
|
||||
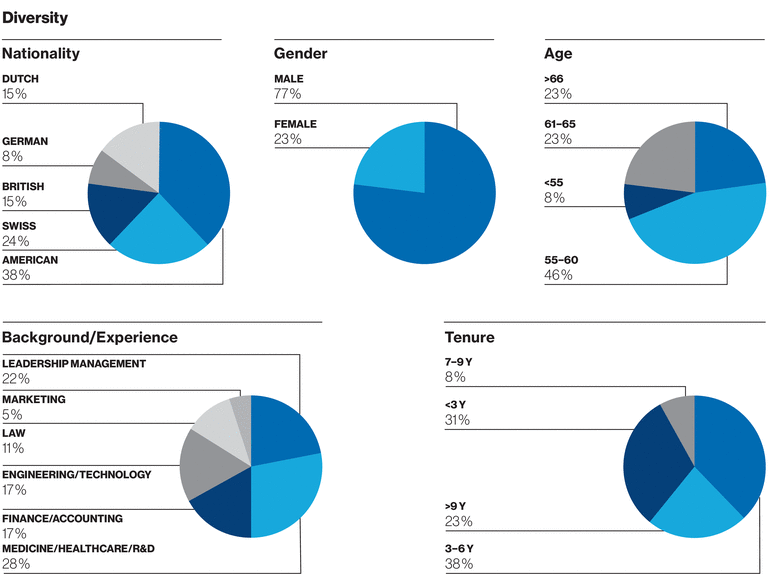
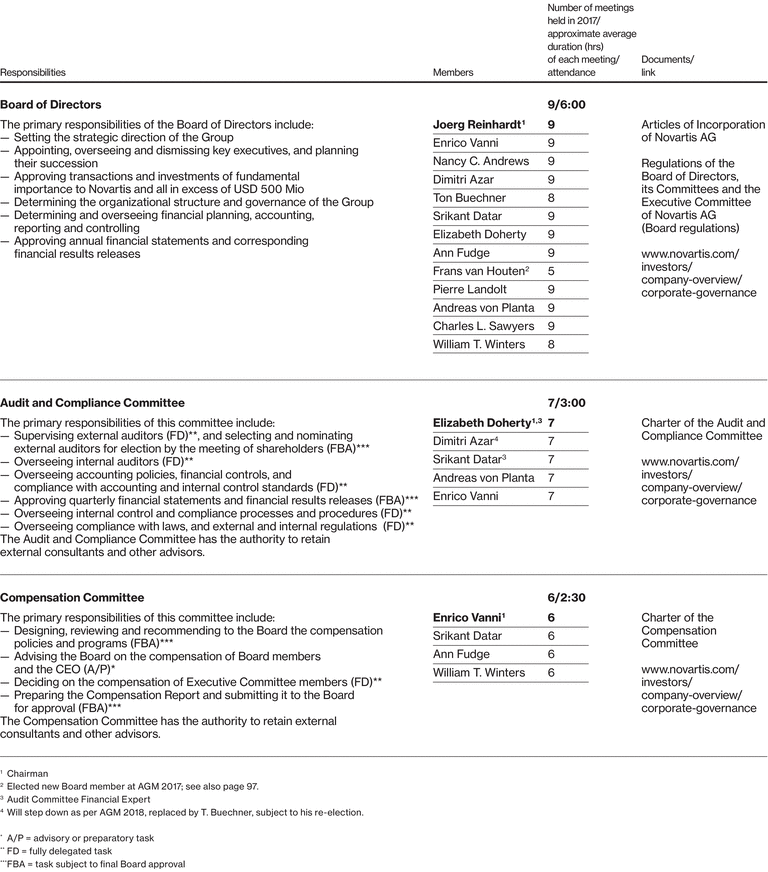


Board of Directors






Board of Directors (continued)







Our management

Executive Committee








Executive Committee (continued)



Our independent external auditors
|
2017
USD million |
2016
USD million |
||||
|
Audit services
|
24.6
|
26.7
|
|||
|
Audit-related services
|
7.2
|
2.9
|
|||
|
Tax services
|
0.8
|
0.7
|
|||
|
Other services
|
1.4
|
1.3
|
|||
|
Total
|
34.0
|
31.6
|
|||
|
|
|||||
Our corporate governance framework
Further information
|
Topic
|
Information
|
||
|
Share capital
|
Articles of Incorporation of Novartis AG
www.novartis.com/investors/company-overview/corporate-governance Novartis key share data www.novartis.com/key-share-data |
||
|
Shareholder rights
|
Articles of Incorporation of Novartis AG
www.novartis.com/investors/company-overview/corporate-governance Investor Relations information www.novartis.com/investors |
||
|
Board regulations
|
Board regulations
www.novartis.com/investors/company-overview/corporate-governance |
||
|
Executive Committee
|
Executive Committee
www.novartis.com/our-company/executive-committee |
||
|
Novartis code for senior financial officers
|
Novartis Code of Ethical Conduct for CEO and Senior Financial Officers
www.novartis.com/investors/company-overview/corporate-governance |
||
|
Additional information
|
Novartis Investor Relations
www.novartis.com/investors |
||
Compensation Committee Chairman’s letter
Dear Shareholder,
Executive Committee compensation at a glance (pages 127 to 142)


Board compensation at a glance (pages 146 to 150)
|
CHF 000s |
AGM 2017-2018
annual fee |
||
|
Chairman of the Board
|
3 800
|
||
|
Board membership
|
300
|
||
|
Vice Chairman
|
50
|
||
|
Chair of the Audit and Compliance Committee
|
120
|
||
|
Chair of the following committees:
• Compensation Committee • Governance, Nomination and Corporate Responsibilities Committee • Research & Development Committee • Risk Committee |
60 |
||
|
Membership of the Audit and Compliance Committee
|
60
|
||
|
Membership of the following committees:
• Compensation Committee • Governance, Nomination and Corporate Responsibilities Committee • Research & Development Committee • Risk Committee |
30 |
||
|
CHF 000s |
2017
total compensation 1 |
||
|
Chairman of the Board
|
3 805
|
||
|
Other 12 members of the Board
|
4 591
|
||
|
Total
|
8 396
|
||
|
1 Includes an amount of CHF 15 622 for mandatory employer contributions for all Board members paid by Novartis to Swiss governmental social security systems. This amount is out of total employer contributions of CHF 298 206, and provides a right to the maximum future insured government pension benefit for the Board member.
|
|||
Compensation governance at a glance
|
Decision on
|
Decision-making authority
|
||
|
Compensation of Chairman and other Board members
|
Board of Directors
|
||
|
Compensation of CEO
|
Board of Directors
|
||
|
Compensation of other Executive Committee members
|
Compensation Committee
|
||
Executive Committee compensation philosophy and principles
Executive Committee compensation policies

Executive Committee performance management process

2017 Executive Committee compensation

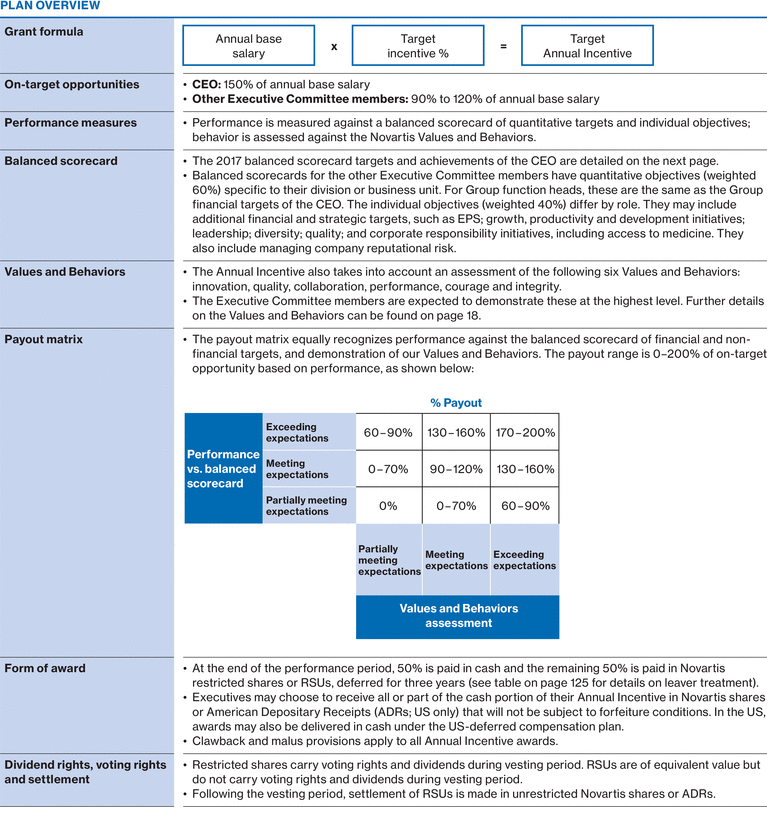


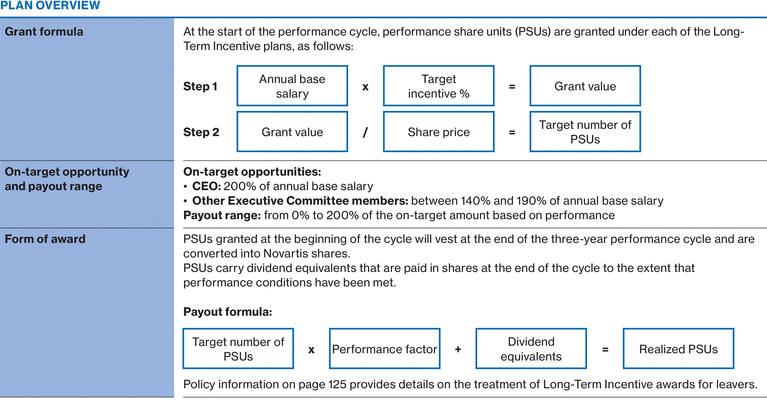




|
|
2017 annual base salary
|
2017 pension benefits
|
2017 Annual Incentive1
|
Long-Term Incentives
|
Other 2017 Compensation2
|
||||||||||||||
|
|
|
|
|
|
LTPP 2015–2017 cycle
|
LTRPP 2015–2017 cycle
|
|
|
|||||||||||
|
Currency |
Cash (amount) |
Amount |
Cash |
Equity 1 |
Equity (value at vesting date) 2 |
Equity (value at vesting date) 2 |
Amount 3 |
Total realized
compensation (incl. Share price movement) 4 |
|||||||||||
|
Executive Committee members active on December 31, 2017
|
|||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|||||||||||
|
Joseph Jimenez (CEO)
|
CHF
|
2 100 000
|
166 397
|
1 968 750
|
1 968 792
|
5 068 337
|
0
|
72 186
|
11 344 462
|
||||||||||
|
Aggregate realized compensation of the other 10 ECN members
|
CHF
|
9 310 740
|
1 675 398
|
5 841 107
|
7 743 069
|
8 355 739
|
0
|
3 248 419
|
36 174 472
|
||||||||||
|
Total 5
|
CHF
|
11 410 740
|
1 841 795
|
7 809 857
|
9 711 861
|
13 424 076
|
0
|
3 320 605
|
47 518 934
|
||||||||||
|
See page 134 for 2016 comparative figures.
|
|||||||||||||||||||
|
1 The portion of the Annual Incentive delivered in equity is rounded up to the nearest share, based on the closing share price on the grant date (January 18, 2018) of CHF 82.90 per Novartis share and USD 86.41 per ADR.
|
|||||||||||||||||||
|
2 The amounts represent the underlying share value of the 160 733 PSUs vesting on January 21, 2018 to the CEO and other Executive Committee members for the performance cycle 2015-2017, inclusive of earned dividend equivalents for the three-year cycle. The value is determined using the closing share price on the last trading day (January 19, 2018) before the vesting date of CHF 83.38 per Novartis share and USD 86.94 per ADR. For two members of the Executive Committee, the vesting value is reported pro-rata based on the period they were an Executive Committee member during the performance cycle.
|
|||||||||||||||||||
|
3 Includes any other perquisites, benefits in kind, international assignment benefits as per the global mobility policy (e.g., housing, international health insurance, children's school fees, tax equalization).
|
|||||||||||||||||||
|
4 All amounts are before deduction of the social security contribution and income tax due by the Executive Committee member.
|
|||||||||||||||||||
|
5 Amounts for Executive Committee members paid in USD were converted at a rate of CHF 1.00 = USD 1.015, which is the same average exchange rate used in the Group's 2017 consolidated financial statements.
|
|||||||||||||||||||
|
|
2016 annual base salary
|
2016 pension benefits
|
2016 Annual Incentive1
|
Long-Term Incentives
|
Other 2016 Compensation2
|
||||||||||||||
|
|
|
|
|
|
LTPP 2014–2016 cycle
|
LTRPP 2014–2016 cycle
|
|
|
|||||||||||
|
Currency |
Cash (amount) |
Amount |
Cash |
Equity 1 |
Equity (value at vesting date) 2 |
Equity (value at vesting date) 2 |
Amount 3 |
Total realized
compensation (incl. Share price movement) 4 |
|||||||||||
|
Executive Committee members active on December 31, 2016 and members who stepped down during financial year 2016
|
|||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|||||||||||
|
Joseph Jimenez (CEO)
|
CHF
|
2 093 417
|
160 283
|
1 417 500
|
1 417 510
|
4 950 334
|
442 013
|
75 628
|
10 556 685
|
||||||||||
|
Aggregate realized compensation of the other 13 ECN members 5
|
CHF
|
8 778 483
|
1 675 484
|
4 825 680
|
6 516 148
|
12 190 674
|
733 656
|
9 684 126
|
44 404 251
|
||||||||||
|
Total 6
|
CHF
|
10 871 900
|
1 835 767
|
6 243 180
|
7 933 658
|
17 141 008
|
1 175 669
|
9 759 754
|
54 960 936
|
||||||||||
|
1 The portion of the Annual Incentive delivered in equity is rounded up to the nearest share, based on the closing share price on the grant date (January 17, 2017) of CHF 71.35 per Novartis share and USD 71.99 per ADR.
|
|||||||||||||||||||
|
2 The amounts represent the underlying share value of the PSUs vesting to Executive Committee members for the performance cycle 2014-2016, based on the closing share price on the vesting date (January 17, 2017) of CHF 71.35 per Novartis share and USD 71.99 per ADR, plus earned dividend equivalents during the three-year cycle.
|
|||||||||||||||||||
|
3 Includes any other perquisites, benefits in kind, international assignment benefits as per the global mobility policy (e.g., housing, international health insurance, children's school fees, tax equalization). In addition, for the three Executive Committee members who stepped down during 2016, it includes, inter alia, their pro-rata compensation from the date they stepped down from the Executive Committee to December 31, 2016.
|
|||||||||||||||||||
|
4 All amounts are before deduction of the social security contribution and income tax due by the Executive Committee member.
|
|||||||||||||||||||
|
5 This represents realized compensation of ten Executive Committee members who were active on December 31, 2016 as well as three members who stepped down during 2016.
|
|||||||||||||||||||
|
6 Amounts for Executive Committee members paid in USD were converted at a rate of CHF 1.00 = USD 1.015, which is the same average exchange rate used in the Group's 2016 consolidated financial statements.
|
|||||||||||||||||||
|
|
Fixed compensation and pension benefits
|
Variable compensation
|
|
|
|||||||||||||||
|
|
Actual compensation paid or granted for 2017
|
Long-Term Incentive 2017-2019 cycle grants at target
|
|
|
|||||||||||||||
|
|
2017 annual base salary
|
2017 pension benefits
|
2017 Annual Incentive (performance achieved)
|
LTPP 2017–2019 cycle
|
LTRPP 2017–2019 cycle
|
Other 2017 compensation
|
Total compensation paid, promised or granted 2017
|
||||||||||||
|
Currency |
Cash (amount) |
Amount 1 |
Cash |
Equity
(value at grant date) 2 |
PSUs
(target value at grant date) 3 |
PSUs
(target value at grant date) 3 |
Amount 4 |
Amount 5 |
|||||||||||
|
Executive Committee members active on December 31, 2017
|
|||||||||||||||||||
|
Joseph Jimenez (CEO)
|
CHF
|
2 100 000
|
166 397
|
1 968 750
|
1 968 792
|
4 200 018
|
2 625 038
|
72 186
|
13 101 181
|
||||||||||
|
Steven Baert
|
CHF
|
775 000
|
154 652
|
663 000
|
663 034
|
1 170 069
|
468 056
|
119 218
|
4 013 029
|
||||||||||
|
F. Michael Ball
|
USD
|
1 120 000
|
203 546
|
873 600
|
873 605
|
1 792 047
|
784 043
|
293 289
|
5 940 130
|
||||||||||
|
James Bradner
|
USD
|
1 066 385
|
117 394
|
898 800
|
898 837
|
1 819 043
|
856 033
|
45 855
|
5 702 347
|
||||||||||
|
Felix R. Ehrat
|
CHF
|
928 333
|
137 334
|
223 200
|
892 833
|
1 581 045
|
558 028
|
15 034
|
4 335 807
|
||||||||||
|
Richard Francis
|
CHF
|
841 667
|
176 362
|
425 000
|
425 028
|
1 360 002
|
510 010
|
1 112 948
|
4 851 017
|
||||||||||
|
Paul Hudson
|
CHF
|
958 333
|
203 485
|
950 400
|
950 449
|
1 536 023
|
672 046
|
197 101
|
5 467 837
|
||||||||||
|
Harry Kirsch
|
CHF
|
1 038 333
|
153 854
|
800 800
|
800 814
|
1 768 053
|
832 012
|
58 710
|
5 452 576
|
||||||||||
|
Vasant Narasimhan
|
CHF
|
841 667
|
168 562
|
807 500
|
807 529
|
1 360 002
|
510 010
|
50 603
|
4 545 873
|
||||||||||
|
Bruno Strigini (until December 31, 2017) 6
|
CHF
|
898 333
|
210 613
|
225 000
|
225 074
|
1 440 057
|
540 048
|
50 000
|
3 589 125
|
||||||||||
|
André Wyss
|
CHF
|
875 000
|
154 339
|
0
|
1 232 060
|
1 408 021
|
528 061
|
70 526
|
4 268 007
|
||||||||||
|
Total 7
|
CHF
|
11 410 740
|
1 841 795
|
7 809 857
|
9 711 861
|
19 381 014
|
8 859 147
|
2 080 458
|
61 094 873
|
||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
Based on assumption of 100% payout at target. Actual payout (0-200% of target) will be known at the end of the three-year cycle in January 2020.
|
|
|
|
|
|
|||
|
See page 136 for 2016 comparative figures.
|
|||||||||||||||||||
|
1 Includes mandatory employer contributions of CHF 4 336 for the CEO and CHF 50 227 for the other Executive Committee members paid by Novartis to governmental social security systems. This amount is out of total employer contributions of CHF 2 710 445 paid in 2017 for all Executive Committee members, and provides a right to the maximum future insured government pension benefit for the Executive Committee member.
|
|||||||||||||||||||
|
2 The portion of the Annual Incentive delivered in equity is rounded up to the nearest share, based on the closing share price on the grant date (January 18, 2018) of CHF 82.90 per Novartis share and USD 86.41 per ADR.
|
|||||||||||||||||||
|
3 The amounts represent the underlying share value of the target number of PSUs granted to Executive Committee members for the performance cycle 2017-2019, based on the closing share price on the grant date (January 17, 2017) of CHF 71.35 per Novartis share and USD 71.99 per ADR.
|
|||||||||||||||||||
|
4 Includes any other perquisites, benefits in kind, and international assignment benefits as per the global mobility policy (e.g., housing, international health insurance, children's school fees, tax equalization).
|
|||||||||||||||||||
|
5 All amounts are before deduction of the social security contribution and income tax due by the Executive Committee member.
|
|||||||||||||||||||
|
6 Bruno Strigini stepped down from the Executive Committee at the end of the 2017 business year. The LTPP and LTRPP grants for the 2017-19 performance cycle, included in the table above, will vest at the end of the performance cycle on a pro-rata basis per his contractual agreement and subject to the plan rules .
|
|||||||||||||||||||
|
7 Amounts in USD for F. Michael Ball and James Bradner were converted at a rate of CHF 1.00 = USD 1.015, which is the same average exchange rate used in the Group's 2017 consolidated financial statements.
|
|||||||||||||||||||
|
|
Fixed compensation and pension benefits
|
Variable compensation
|
|
|
|||||||||||||||
|
|
Actual compensation paid or granted for 2016
|
Long-Term Incentive 2016-2018 cycle grants at target
|
|
|
|||||||||||||||
|
|
2016 annual base salary
|
2016 pension benefits
|
2016 Annual Incentive (performance achieved)
|
LTPP 2016–2018 cycle
|
LTRPP 2016–2018 cycle
|
Other 2016 compensation
|
Total compensation paid, promised or granted 2016
|
||||||||||||
|
Currency |
Cash (amount) |
Amount 1 |
Cash |
Equity
(value at grant date) 2 |
PSUs
(target value at grant date) 3 |
PSUs
(target value at grant date) 3 |
Amount 4 |
Amount 5 |
|||||||||||
|
Executive Committee members active on December 31, 2016 6
|
|||||||||||||||||||
|
Joseph Jimenez (CEO)
|
CHF
|
2 093 417
|
160 283
|
1 417 500
|
1 417 510
|
4 200 031
|
2 625 079
|
75 628
|
11 989 448
|
||||||||||
|
Steven Baert
|
CHF
|
721 667
|
147 442
|
554 730
|
554 746
|
1 050 048
|
350 042
|
139 159
|
3 517 834
|
||||||||||
|
F. Michael Ball (from February 1, 2016)
|
USD
|
1 012 308
|
60 574
|
553 574
|
553 603
|
1 742 284
|
762 269
|
4 040 748
|
8 725 360
|
||||||||||
|
James Bradner (from March 1, 2016)
|
USD
|
888 462
|
58 859
|
579 393
|
579 448
|
1 687 473
|
794 195
|
1 155 169
|
5 742 999
|
||||||||||
|
Felix R. Ehrat
|
CHF
|
915 833
|
148 122
|
202 400
|
809 680
|
1 564 033
|
552 002
|
14 852
|
4 206 922
|
||||||||||
|
Richard Francis
|
CHF
|
786 667
|
188 738
|
520 000
|
520 070
|
1 280 062
|
480 033
|
1 116 054
|
4 891 624
|
||||||||||
|
Paul Hudson (from July 1, 2016)
|
CHF
|
475 000
|
108 818
|
288 945
|
288 968
|
0
|
0
|
3 090 313
|
4 252 044
|
||||||||||
|
Harry Kirsch
|
CHF
|
1 025 000
|
141 510
|
736 450
|
736 475
|
1 751 009
|
824 018
|
51 361
|
5 265 823
|
||||||||||
|
Vasant Narasimhan (from February 1, 2016)
|
CHF
|
764 993
|
157 348
|
537 531
|
537 551
|
1 093 245
|
364 468
|
102 868
|
3 558 004
|
||||||||||
|
Bruno Strigini (from July 1, 2016)
|
CHF
|
445 000
|
109 057
|
211 863
|
211 910
|
1 074 442
|
268 670
|
45 696
|
2 366 638
|
||||||||||
|
André Wyss
|
CHF
|
830 834
|
146 289
|
0
|
1 275 025
|
1 360 001
|
425 040
|
95 595
|
4 132 784
|
||||||||||
|
Subtotal 7
|
CHF
|
9 931 091
|
1 425 275
|
5 585 643
|
7 468 241
|
16 751 942
|
7 422 814
|
9 850 656
|
58 435 662
|
||||||||||
|
Executive Committee members who stepped down during 2016 8
|
|||||||||||||||||||
|
David Epstein (until June 30, 2016)
|
USD
|
699 767
|
290 385
|
428 400
|
428 412
|
1 285 264
|
642 632
|
4 529 809
|
8 304 669
|
||||||||||
|
Mark C. Fishman (until February 29, 2016)
|
USD
|
175 154
|
107 706
|
195 000
|
0
|
0
|
0
|
126 454
|
604 314
|
||||||||||
|
Jeff George (until January 31, 2016)
|
USD
|
80 000
|
18 558
|
44 000
|
43 986
|
0
|
0
|
2 996 905
|
3 183 449
|
||||||||||
|
Subtotal 7
|
CHF
|
940 809
|
410 492
|
657 537
|
465 417
|
1 266 270
|
633 135
|
7 540 067
|
11 913 726
|
||||||||||
|
Total 7
|
CHF
|
10 871 900
|
1 835 767
|
6 243 180
|
7 933 658
|
18 018 212
|
8 055 949
|
17 390 723
|
70 349 389
|
||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
Based on assumption of 100% payout at target. Actual payout (0-200% of target) will be known at the end of the three-year cycle in January 2019.
|
|
|
|
|
|
|||
|
1 Includes mandatory employer contributions of CHF 4 336 for the CEO and CHF 70 880 for the other Executive Committee members paid by Novartis to governmental social security systems. This amount is out of total employer contributions of CHF 3 263 989 paid in 2016 for all Executive Committee members, and provides a right to the maximum future insured government pension benefit for the Executive Committee member.
|
|||||||||||||||||||
|
2 The portion of the Annual Incentive delivered in equity is rounded up to the nearest share, based on the closing share price on the grant date (January 17, 2017) of CHF 71.35 per Novartis share and USD 71.99 per ADR.
|
|||||||||||||||||||
|
3 The amounts represent the underlying share value of the target number of PSUs granted to Executive Committee members for the performance cycle 2016-2018, based on the closing share price on the grant date (January 20, 2016) of CHF 79.70 per Novartis share and USD 80.49 per ADR. For F. Michael Ball, the target PSUs were granted on February 1, 2016, at the closing share price of the same date (USD 77.27 per ADR).
|
|||||||||||||||||||
|
4 Includes any other perquisites, benefits in kind, international assignment benefits as per the global mobility policy (e.g., housing, international health insurance, children's school fees, tax equalization), compensation granted for forfeited entitlements at previous employers and, for F. Michael Ball, a one-off performance award with target value at grant date of USD 3.9 million. In addition, for Executive Committee members who stepped down during 2016, it includes, inter alia, their pro-rata compensation from the date they stepped down from the Executive Committee to December 31, 2016 (see also note 8 below).
|
|||||||||||||||||||
|
5 All amounts are before deduction of the social security contribution and income tax due by the Executive Committee member.
|
|||||||||||||||||||
|
6 For those members who joined the Executive Committee in 2016, the information under the columns "annual base salary", "pension benefits" and "Annual Incentive" includes their pro-rata compensation from the date they joined the Executive Committee to December 31, 2016. The information under "LTPP" and "LTRPP" columns reflects their pro-rata compensation at target for the period to December 31, 2018.
|
|||||||||||||||||||
|
7 Amounts in USD for Mr. Ball, James Bradner, David Epstein, Mark C. Fishman and Jeff George were converted at a rate of CHF 1.00 = USD 1.015, which is the same average exchange rate used in the Group's 2016 consolidated financial statements.
|
|||||||||||||||||||
|
8 For those members who stepped down from the Executive Committee in 2016, the information under the columns "annual base salary", "pension benefits", "Annual Incentive", "LTPP" and "LTRPP" reflects the pro-rata value during 2016 for the period they were an Executive Committee member. The information under the column "Other 2016 compensation" includes, inter alia, the aggregated pro-rata value from the date they stepped down from the Executive Committee to December 31, 2016.
|
|||||||||||||||||||
|
Variable compensation
|
|||||||||||
|
2017 Annual Incentive (performance achieved)
|
LTPP 2017–2019 cycle
|
LTRPP 2017–2019 cycle
|
|||||||||
|
Equity
(number) 2 |
PSUs
(target number) 3 |
PSUs
(target number) 3 |
|||||||||
|
Executive Committee members active on December 31, 2017
|
|||||||||||
|
Joseph Jimenez (CEO)
|
23 749
|
58 865
|
36 791
|
||||||||
|
Steven Baert
|
7 998
|
16 399
|
6 560
|
||||||||
|
F. Michael Ball
|
10 110
|
24 893
|
10 891
|
||||||||
|
James Bradner
|
10 402
|
25 268
|
11 891
|
||||||||
|
Felix R. Ehrat
|
10 770
|
22 159
|
7 821
|
||||||||
|
Richard Francis
|
5 127
|
19 061
|
7 148
|
||||||||
|
Paul Hudson
|
11 465
|
21 528
|
9 419
|
||||||||
|
Harry Kirsch
|
9 660
|
24 780
|
11 661
|
||||||||
|
Vasant Narasimhan
|
9 741
|
19 061
|
7 148
|
||||||||
|
Bruno Strigini (until December 31, 2017) 4
|
2 715
|
20 183
|
7 569
|
||||||||
|
André Wyss
|
14 862
|
19 734
|
7 401
|
||||||||
|
Total
|
116 599
|
271 931
|
124 300
|
||||||||
|
See page 140 for 2016 comparative figures.
|
|||||||||||
|
1 The values of the awards are reported in the table "2017 compensation at grant value for the CEO and other Executive Committee members" on page 135.
|
|||||||||||
|
2 Vested shares, restricted shares and/or RSUs granted under the Annual Incentive for performance period 2017
|
|||||||||||
|
3 Target number of PSUs granted under the LTPP and LTRPP as applicable for the performance cycle 2017-2019
|
|||||||||||
|
4 Bruno Strigini stepped down from the Executive Committee at the end of the 2017 business year. The LTPP and LTRPP grants for the 2017-19 performance cycle, included in the table above, will vest at the end of the performance cycle on a pro-rata basis per his contractual agreement and subject to the plan rules
|
|||||||||||
|
Variable compensation
|
|||||||||
|
2016 Annual Incentive (performance achieved)
|
LTPP 2016–2018 cycle
|
LTRPP 2016–2018 cycle
|
Other
|
||||||
|
Equity
(number) 2 |
PSUs
(target number) 3 |
PSUs
(target number) 3 |
Equity/PSUs
(number) |
||||||
|
Executive Committee members active on December 31, 2016
|
|||||||||
|
Joseph Jimenez (CEO)
|
19 867
|
52 698
|
32 937
|
0
|
|||||
|
Steven Baert
|
7 775
|
13 175
|
4 392
|
0
|
|||||
|
F. Michael Ball (from February 1, 2016)
|
7 690
|
22 548
|
9 865
|
50 000
|
|||||
|
James Bradner (from March 1, 2016)
|
8 049
|
20 965
|
9 867
|
3 607
|
|||||
|
Felix R. Ehrat
|
11 348
|
19 624
|
6 926
|
0
|
|||||
|
Richard Francis
|
7 289
|
16 061
|
6 023
|
0
|
|||||
|
Paul Hudson (from July 1, 2016) 4
|
4 050
|
0
|
0
|
34 502
|
|||||
|
Harry Kirsch
|
10 322
|
21 970
|
10 339
|
0
|
|||||
|
Vasant Narasimhan (from February 1, 2016)
|
7 534
|
13 717
|
4 573
|
0
|
|||||
|
Bruno Strigini (from July 1, 2016)
|
2 970
|
13 549
|
3 388
|
0
|
|||||
|
André Wyss
|
17 870
|
17 064
|
5 333
|
0
|
|||||
|
Subtotal
|
104 764
|
211 371
|
93 643
|
88 109
|
|||||
|
Executive Committee members who stepped down during 2016
|
|||||||||
|
David Epstein (until June 30, 2016)
|
5 951
|
15 968
|
7 984
|
29 902
|
|||||
|
Mark C. Fishman (until February 29, 2016) 4
|
0
|
0
|
0
|
0
|
|||||
|
Jeff George (until January 31, 2016) 4
|
611
|
0
|
0
|
6 724
|
|||||
|
Subtotal
|
6 562
|
15 968
|
7 984
|
36 626
|
|||||
|
Total
|
111 326
|
227 339
|
101 627
|
124 735
|
|||||
|
1 The values of the awards are reported in the table "2016 compensation at grant value for the CEO and other Executive Committee members" on page 136.
|
|||||||||
|
2 Vested shares, restricted shares and/or RSUs granted under the Annual Incentive for performance period 2016
|
|||||||||
|
3 Target number of PSUs granted under the LTPP and LTRPP as applicable for the performance cycle 2016-2018
|
|||||||||
|
4 Paul Hudson, Mark C. Fishman and Jeff George were not granted LTPP and LTRPP awards for the performance cycle 2016-2018.
|
|||||||||
|
Function
|
Ownership level
|
||
|
CEO
|
5 x base compensation
|
||
|
Other Executive Committee members
|
3 x base compensation
|
||
|
Vested shares and ADRs |
Unvested shares and other equity rights 2 |
Equity ownership level
as a multiple of annual base salary 3 |
Unvested target PSUs (e.g., LTPP / LTRPP) 4 |
Matching shares under the LSSP 5 |
Total at December 31, 2017 |
||||||||
|
Joseph Jimenez (CEO)
|
287 699
|
62 693
|
14x
|
225 685
|
0
|
576 077
|
|||||||
|
Steven Baert
|
10 955
|
21 410
|
3x
|
33 715
|
0
|
66 080
|
|||||||
|
F. Michael Ball
|
0
|
7 690
|
1x
|
101 532
|
0
|
109 222
|
|||||||
|
James Bradner
|
0
|
13 234
|
1x
|
34 130
|
0
|
47 364
|
|||||||
|
Felix R. Ehrat
|
189 940
|
23 541
|
19x
|
79 764
|
19 950
|
313 195
|
|||||||
|
Richard Francis
|
35 117
|
17 305
|
5x
|
40 453
|
0
|
92 875
|
|||||||
|
Paul Hudson
|
6 616
|
6 498
|
1x
|
29 695
|
0
|
42 809
|
|||||||
|
Harry Kirsch
|
64 769
|
30 309
|
8x
|
58 792
|
6 277
|
160 147
|
|||||||
|
Vasant Narasimhan
|
16 279
|
58 887
|
7x
|
23 413
|
3 426
|
102 005
|
|||||||
|
Bruno Strigini
|
27 871
|
39 844
|
6x
|
38 930
|
0
|
106 645
|
|||||||
|
André Wyss
|
51 183
|
22 784
|
7x
|
40 456
|
0
|
114 423
|
|||||||
|
Total
|
690 429
|
304 195
|
|
706 565
|
29 653
|
1 730 842
|
|||||||
|
1 Includes holdings of "persons closely linked" to Executive Committee members (see definition on page 142)
|
|||||||||||||
|
2 Includes unvested shares and ADRs as well as other equity rights applicable for the determination of equity amounts for the share ownership requirements, as per the definition above
|
|||||||||||||
|
3 The multiple is calculated based on the full year annual base salary and the closing share price as at the end of the 2017 Financial Year. The share price on the final trading day of 2017 was CHF 82.40 / USD 83.96 as at December 29, 2017.
|
|||||||||||||
|
4 Target number of PSUs are disclosed pro-rata to December 31, 2017, unless the award qualified for full vesting under the relevant plan rules.
|
|||||||||||||
|
5 Matching shares under the Leveraged Share Savings Plan (LSSP) are disclosed pro-rata to December 31, 2017, unless the award qualified for full vesting under the plan rules. LSSP participation for Executive Committee members ceased in 2014 and no new LSSP awards have been made since then. Outstanding awards will vest five years from the grant date, subject to the LSSP plan rules.
|
|||||||||||||
|
Annual
base salary 1 |
Variable
compensation 2 |
||||
|
Joseph Jimenez (CEO)
|
16.3%
|
83.7%
|
|||
|
Steven Baert
|
20.7%
|
79.3%
|
|||
|
F. Michael Ball
|
20.6%
|
79.4%
|
|||
|
James Bradner
|
19.3%
|
80.7%
|
|||
|
Felix R. Ehrat
|
22.2%
|
77.8%
|
|||
|
Richard Francis
|
23.6%
|
76.4%
|
|||
|
Paul Hudson
|
18.9%
|
81.1%
|
|||
|
Harry Kirsch
|
19.8%
|
80.2%
|
|||
|
Vasant Narasimhan
|
19.5%
|
80.5%
|
|||
|
Bruno Strigini
|
27.0%
|
73.0%
|
|||
|
André Wyss
|
21.6%
|
78.4%
|
|||
|
Total
|
20.0%
|
80.0%
|
|||
|
1 Excludes pension and other benefits
|
|||||
|
2 See table “2017 compensation at grant value for the CEO and other Executive Committee members" on page 135 with regard to the disclosure principles of variable compensation.
|
|||||
2018 Executive Committee compensation
|
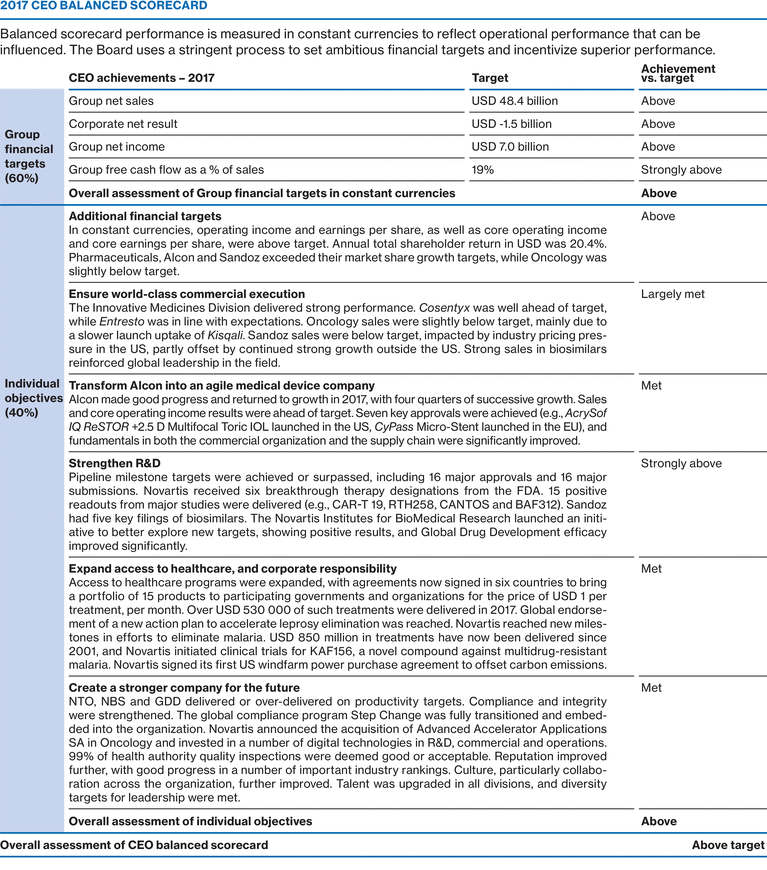
CEO Balanced Scorecard - Key Metrics
|
|
|
Group financial targets (60% weighting)
|
|
|
• Group net sales
|
|
|
• Group operating income
|
|
|
• Group FCF as % of sales
|
|
|
• Share of peers
|
|
|
Strategic objectives (40% weighting)
|
|
|
• Innovation
|
|
|
• Access to healthcare
|
|
|
• People and culture
|
|
|
• Data and digital
|
|
|
Performance
|
Payout
|
||
|
Outstanding
|
170–200%
|
||
|
Exceeds expectations
|
130–160%
|
||
|
Meets expectations
|
80–120%
|
||
|
Partially meets expectations
|
40–70%
|
||
|
Below expectations
|
0–30%
|
||
|
Novartis position
in the peer group |
Payout range
(% of target) |
||
|
Positions 1–2
|
170–200%
|
||
|
Positions 3–5
|
130–160%
|
||
|
Positions 6–8
|
80–120%
|
||
|
Positions 9–16
|
0%
|
||
2017 Board compensation
|
CHF |
AGM 2017-2018
annual fee |
||
|
Chairman of the Board
|
3 800 000
|
||
|
Board membership
|
300 000
|
||
|
Vice Chairman
|
50 000
|
||
|
Chair of the Audit and Compliance Committee
|
120 000
|
||
|
Chair of the following committees:
• Compensation Committee • Governance, Nomination and Corporate Responsibilities Committee • Research & Development Committee • Risk Committee |
60 000 |
||
|
Membership of the Audit and Compliance Committee
|
60 000
|
||
|
Membership of the following committees:
• Compensation Committee • Governance, Nomination and Corporate Responsibilities Committee • Research & Development Committee • Risk Committee |
30 000 |
||
|
Board membership
|
Vice Chairman
|
Audit and Compliance Committee
|
Compensation Committee
|
Governance, Nomination and Corporate Responsibilities Committee
|
Research & Development Committee
|
Risk Committee
|
Shares (number) 1
|
Cash (CHF) (A)
|
Shares (CHF) (B)
|
Other (CHF) (C) 2
|
Total (CHF) (A)+(B)+(C) 3
|
||||||||||||||
|
Board members active on December 31, 2017
|
|||||||||||||||||||||||||
|
Joerg Reinhardt 4
|
Chair
|
|
|
|
|
Chair
|
|
24 407
|
1 900 000
|
1 900 000
|
4 336
|
3 804 336
|
|||||||||||||
|
Enrico Vanni
|
•
|
•
|
•
|
Chair
|
•
|
|
|
3 210
|
250 000
|
250 000
|
3 475
|
503 475
|
|||||||||||||
|
Nancy Andrews
|
•
|
|
|
|
|
•
|
•
|
2 311
|
180 000
|
180 000
|
–
|
360 000
|
|||||||||||||
|
Dimitri Azar
|
•
|
|
•
|
|
|
•
|
|
2 504
|
195 000
|
195 000
|
–
|
390 000
|
|||||||||||||
|
Ton Buechner
|
•
|
|
|
|
|
|
• 5
|
4 039
|
–
|
325 000
|
–
|
325 000
|
|||||||||||||
|
Srikant Datar
|
•
|
|
• 7
|
•
|
|
|
Chair 5
|
2 989
|
227 500
|
227 500
|
–
|
455 000
|
|||||||||||||
|
Elizabeth Doherty
|
•
|
|
Chair 5
|
|
|
|
• 5
|
2 591
|
217 500
|
217 500
|
–
|
435 000
|
|||||||||||||
|
Ann Fudge
|
•
|
|
|
•
|
•
|
|
•
|
2 504
|
195 000
|
195 000
|
–
|
390 000
|
|||||||||||||
|
Frans van Houten (from February 28, 2017)
|
•
|
|
|
|
|
|
|
1 305
|
75 000
|
175 000
|
–
|
250 000
|
|||||||||||||
|
Pierre Landolt 6
|
•
|
|
|
|
•
|
|
|
4 238
|
–
|
330 000
|
3 475
|
333 475
|
|||||||||||||
|
Andreas von Planta
|
•
|
|
•
|
|
Chair
|
|
• 8
|
2 989
|
227 500
|
227 500
|
4 336
|
459 336
|
|||||||||||||
|
Charles L. Sawyers
|
•
|
|
|
|
•
|
•
|
|
2 311
|
180 000
|
180 000
|
–
|
360 000
|
|||||||||||||
|
William T. Winters
|
•
|
|
|
•
|
|
|
|
4 238
|
–
|
330 000
|
–
|
330 000
|
|||||||||||||
|
Total
|
|
|
|
|
|
|
|
59 636
|
3 647 500
|
4 732 500
|
15 622
|
8 395 622
|
|||||||||||||
|
See next page for 2016 comparative figures.
|
|||||||||||||||||||||||||
|
1 The shown amounts represent the gross number of shares delivered to each Board member in 2017 for the respective Board member’s service period. The number of shares reported in this column represent: (i) the second and final equity installment delivered in February 2017 for the services from the 2016 AGM to the 2017 AGM, and (ii) the first of two equity installments delivered in August 2017 for the services from the 2017 AGM to the 2018 AGM. The second and final equity installment for the services from the 2017 AGM to the 2018 AGM will take place in February 2018.
|
|||||||||||||||||||||||||
|
2 Includes an amount of CHF 15 622 for mandatory employer contributions for all Board members paid by Novartis to Swiss governmental social security systems. This amount is out of total employer contributions of CHF 298 206, and provides a right to the maximum future insured government pension benefit for the Board member.
|
|||||||||||||||||||||||||
|
3 All amounts are before deduction of the social security contribution and income tax due by the Board member.
|
|||||||||||||||||||||||||
|
4 No additional committee fees for chairing the Research & Development Committee were delivered to Dr. Reinhardt.
|
|||||||||||||||||||||||||
|
5 From February 28, 2017
|
|||||||||||||||||||||||||
|
6 According to Pierre Landolt, the Sandoz Family Foundation is the economic beneficiary of the compensation.
|
|||||||||||||||||||||||||
|
7 Until February 27, 2017, Chair of the Audit and Compliance Committee
|
|||||||||||||||||||||||||
|
8 Until February 27, 2017, Chair of the Risk Committee
|
|||||||||||||||||||||||||
|
Board membership
|
Vice Chairman
|
Audit and Compliance Committee
|
Compensation Committee
|
Governance, Nomination and Corporate Responsibilities Committee
|
Research & Development Committee
|
Risk Committee
|
Shares (number) 1
|
Cash (CHF) (A)
|
Shares (CHF) (B)
|
Other (CHF) (C) 2
|
Total (CHF) (A)+(B)+(C) 3
|
||||||||||||||
|
Board members active on December 31, 2016
|
|||||||||||||||||||||||||
|
Joerg Reinhardt 4
|
Chair
|
|
|
|
|
Chair
|
|
25 020
|
1 900 000
|
1 900 000
|
4 336
|
3 804 336
|
|||||||||||||
|
Enrico Vanni
|
•
|
•
|
•
|
Chair
|
• 5
|
• 6
|
|
3 291
|
250 000
|
250 000
|
4 336
|
504 336
|
|||||||||||||
|
Nancy Andrews
|
•
|
|
|
|
|
•
|
• 5
|
2 265
|
177 500
|
177 500
|
–
|
355 000
|
|||||||||||||
|
Dimitri Azar
|
•
|
|
•
|
|
|
•
|
|
2 567
|
195 000
|
195 000
|
–
|
390 000
|
|||||||||||||
|
Ton Buechner (from February 24, 2016)
|
•
|
|
|
|
|
|
|
1 864
|
–
|
250 000
|
–
|
250 000
|
|||||||||||||
|
Srikant Datar
|
•
|
|
Chair
|
•
|
|
|
•
|
3 159
|
240 000
|
240 000
|
–
|
480 000
|
|||||||||||||
|
Elizabeth Doherty (from February 24, 2016)
|
•
|
|
•
|
|
|
|
|
1 118
|
150 000
|
150 000
|
–
|
300 000
|
|||||||||||||
|
Ann Fudge
|
•
|
|
|
•
|
•
|
|
•
|
2 567
|
195 000
|
195 000
|
–
|
390 000
|
|||||||||||||
|
Pierre Landolt 7
|
•
|
|
|
|
• 8
|
|
|
4 553
|
–
|
335 000
|
3 475
|
338 475
|
|||||||||||||
|
Andreas von Planta
|
•
|
|
•
|
|
Chair 5
|
|
Chair
|
3 055
|
237 500
|
237 500
|
4 336
|
479 336
|
|||||||||||||
|
Charles L. Sawyers
|
•
|
|
|
|
•
|
•
|
|
2 369
|
180 000
|
180 000
|
–
|
360 000
|
|||||||||||||
|
William T. Winters
|
•
|
|
|
•
|
|
|
|
4 344
|
–
|
330 000
|
–
|
330 000
|
|||||||||||||
|
Subtotal
|
|
|
|
|
|
|
|
56 172
|
3 525 000
|
4 440 000
|
16 483
|
7 981 483
|
|||||||||||||
|
Board members who stepped down at the 2016 AGM
|
|||||||||||||||||||||||||
|
Verena A. Briner (until February 23, 2016)
|
•
|
|
|
|
|
|
•
|
1 147
|
27 500
|
27 500
|
579
|
55 579
|
|||||||||||||
|
Subtotal
|
|
|
|
|
|
|
|
1 147
|
27 500
|
27 500
|
579
|
55 579
|
|||||||||||||
|
Total
|
|
|
|
|
|
|
|
57 319
|
3 552 500
|
4 467 500
|
17 062
|
8 037 062
|
|||||||||||||
|
1 The shown amounts represent the gross number of shares delivered to each Board member in 2016 for the respective Board member’s service period. The number of shares reported in this column represent: (i) the second and final equity installment delivered in February 2016 for the services from the 2015 AGM to the 2016 AGM, and (ii) the first of two equity installments delivered in August 2016 for the services from the 2016 AGM to the 2017 AGM. The second and final equity installment for the services from the 2016 AGM to the 2017 AGM will take place in February 2017.
|
|||||||||||||||||||||||||
|
2 Includes an amount of CHF 17 062 for mandatory employer contributions for all Board members paid by Novartis to Swiss governmental social security systems. This amount is out of total employer contributions of CHF 387 308, and provides a right to the maximum future insured government pension benefit for the Board member.
|
|||||||||||||||||||||||||
|
3 All amounts are before deduction of the social security contribution and income tax due by the Board member.
|
|||||||||||||||||||||||||
|
4 Does not include EUR 1 045 800 paid to Joerg Reinhardt on January 31, 2016, for lost entitlements at his former employer. This amount is the third and final of three installments totaling EUR 2 665 051, which compensates him for lost entitlements at his former employer. The lost entitlements of EUR 2 665 051 were included in full on page 124 of the 2014 Compensation Report. No additional committee fees for chairing the Research & Development Committee were delivered to Dr. Reinhardt.
|
|||||||||||||||||||||||||
|
5 From February 24, 2016.
|
|||||||||||||||||||||||||
|
6 Until February 23, 2016.
|
|||||||||||||||||||||||||
|
7 According to Pierre Landolt, the Sandoz Family Foundation is the economic beneficiary of the compensation.
|
|||||||||||||||||||||||||
|
8 Until February 23, 2016, Chair of the Governance, Nomination and Corporate Responsibilities Committee.
|
|||||||||||||||||||||||||
|
CHF
|
Compensation earned for the respective financial year (A) 1
|
Compensation earned for the period from January 1 to the AGM (2 months) of the financial year (B)
|
Compensation to be earned for the period from January 1 to the AGM (2 months) in the year following the financial year (C)
|
Total compensation earned from AGM to AGM (A)-(B)+(C)
|
Amount approved by shareholders at the respective AGM
|
Amount within the amount approved by shareholders at the respective AGM
|
|||||||
|
|
|
|
|
|
|
||||||||
|
2017
|
January 1, 2017 to 2017 AGM
|
January 1, 2018 to 2018 AGM 2
|
2017 AGM to 2018 AGM
|
2017 AGM
|
2017 AGM
|
||||||||
|
Joerg Reinhardt
|
3 804 336
|
633 334
|
633 334
|
3 804 336
|
3 805 000
|
Yes
|
|||||||
|
Other Board members
|
4 591 286
|
713 334
|
773 334
|
4 651 286
|
4 720 000
|
Yes
|
|||||||
|
Total
|
8 395 622
|
1 346 668
|
1 406 668
|
8 455 622
|
8 525 000
|
Yes
|
|||||||
|
|
|
|
|
|
|
||||||||
|
|
|
|
|
|
|
||||||||
|
2016
|
January 1, 2016 to 2016 AGM
|
January 1, 2017 to 2017 AGM
|
2016 AGM to 2017 AGM
|
2016 AGM
|
2016 AGM
|
||||||||
|
Joerg Reinhardt
|
3 804 336
|
633 334
|
633 334
|
3 804 336
|
3 805 000
|
Yes
|
|||||||
|
Other Board members
|
4 232 726
|
653 334
|
713 334
|
4 292 726
|
4 355 000
|
Yes
|
|||||||
|
Total
|
8 037 062
|
1 286 668
|
1 346 668
|
8 097 062
|
8 160 000
|
Yes
|
|||||||
|
1 See page 147 for 2017 Board member compensation.
|
|||||||||||||
|
2 To be confirmed and reported in the 2018 Compensation Report
|
|||||||||||||
|
Number of shares
At December 31, 2017 1,2 |
|||
|
Joerg Reinhardt
|
518 310
|
||
|
Enrico Vanni
|
20 101
|
||
|
Nancy Andrews
|
4 042
|
||
|
Dimitri Azar
|
13 094
|
||
|
Ton Buechner
|
4 428
|
||
|
Srikant Datar
|
37 239
|
||
|
Elizabeth Doherty
|
2 761
|
||
|
Ann Fudge
|
15 457
|
||
|
Frans van Houten
|
978
|
||
|
Pierre Landolt 3
|
61 029
|
||
|
Andreas von Planta
|
130 634
|
||
|
Charles L. Sawyers
|
7 763
|
||
|
William T. Winters
|
12 397
|
||
|
Total
|
828 233
|
||
|
1 Includes holdings of "persons closely linked" to Board members (see definition on page 142)
|
|||
|
2 Each share provides entitlement to one vote.
|
|||
|
3 According to Pierre Landolt, the Sandoz Family Foundation is the economic beneficiary of the shares
|
|||
2018 Board compensation
|
CHF |
AGM 2018-2019
annual fee |
||
|
Chairman of the Board
|
3 800 000
|
||
|
Board membership
|
280 000
|
||
|
Vice Chairman
|
50 000
|
||
|
Chair of the Audit and Compliance Committee
|
130 000
|
||
|
Chair of the Compensation Committee
|
90 000
|
||
|
Chair of the following committees:
• Governance, Nomination and Corporate Responsibilities Committee • Research & Development Committee • Risk Committee |
70 000 |
||
|
Membership of the Audit and Compliance Committee
|
70 000
|
||
|
Membership of the following committees:
• Compensation Committee • Governance, Nomination and Corporate Responsibilities Committee • Research & Development Committee • Risk Committee |
40 000 |
||
Compensation governance

|
Decision on
|
Decision-making authority
|
||
|
Compensation of Chairman and other Board members
|
Board of Directors
|
||
|
Compensation of CEO
|
Board of Directors
|
||
|
Compensation of other Executive Committee members
|
Compensation Committee
|
||
Novartis Group consolidated financial statements
|
(USD millions unless indicated otherwise)
|
Note
|
2017
|
2016
|
2015
|
|||||
|
Net sales to third parties from continuing operations
|
3
|
49 109
|
48 518
|
49 414
|
|||||
|
Sales to discontinued segments
|
|
|
|
26
|
|||||
|
Net sales from continuing operations
|
3
|
49 109
|
48 518
|
49 440
|
|||||
|
Other revenues
|
|
1 026
|
918
|
947
|
|||||
|
Cost of goods sold
|
|
– 17 175
|
– 17 520
|
– 17 404
|
|||||
|
Gross profit from continuing operations
|
|
32 960
|
31 916
|
32 983
|
|||||
|
Marketing & Sales
|
|
– 12 861
|
– 11 998
|
– 11 772
|
|||||
|
Research & Development
|
|
– 8 972
|
– 9 039
|
– 8 935
|
|||||
|
General & Administration
|
|
– 2 136
|
– 2 194
|
– 2 475
|
|||||
|
Other income
|
|
1 969
|
1 927
|
2 049
|
|||||
|
Other expense
|
|
– 2 331
|
– 2 344
|
– 2 873
|
|||||
|
Operating income from continuing operations
|
3
|
8 629
|
8 268
|
8 977
|
|||||
|
Income from associated companies
|
4
|
1 108
|
703
|
266
|
|||||
|
Interest expense
|
5
|
– 777
|
– 707
|
– 655
|
|||||
|
Other financial income and expense
|
5
|
39
|
– 447
|
– 454
|
|||||
|
Income before taxes from continuing operations
|
|
8 999
|
7 817
|
8 134
|
|||||
|
Taxes
|
6
|
– 1 296
|
– 1 119
|
– 1 106
|
|||||
|
Net income from continuing operations
|
|
7 703
|
6 698
|
7 028
|
|||||
|
Net income from discontinued operations
|
29
|
|
|
10 766
|
|||||
|
Net income
|
|
7 703
|
6 698
|
17 794
|
|||||
|
Attributable to:
|
|
|
|
|
|||||
|
Shareholders of Novartis AG
|
|
7 703
|
6 712
|
17 783
|
|||||
|
Non-controlling interests
|
|
0
|
– 14
|
11
|
|||||
|
|
|
|
|
||||||
|
Basic earnings per share (USD) from continuing operations
|
|
3.28
|
2.82
|
2.92
|
|||||
|
Basic earnings per share (USD) from discontinued operations
|
|
|
|
4.48
|
|||||
|
Total basic earnings per share (USD)
|
7
|
3.28
|
2.82
|
7.40
|
|||||
|
|
|
|
|
||||||
|
Diluted earnings per share (USD) from continuing operations
|
|
3.25
|
2.80
|
2.88
|
|||||
|
Diluted earnings per share (USD) from discontinued operations
|
|
|
|
4.41
|
|||||
|
Total diluted earnings per share (USD)
|
7
|
3.25
|
2.80
|
7.29
|
|||||
|
The accompanying Notes form an integral part of the consolidated financial statements.
|
|||||||||
|
(USD millions)
|
Note
|
2017
|
2016
|
2015
|
|||||
|
Net income
|
|
7 703
|
6 698
|
17 794
|
|||||
|
Other comprehensive income to be eventually recycled into the consolidated income statement:
|
|
|
|
|
|||||
|
Fair value adjustments on marketable securities, net of taxes
|
8.1
|
38
|
– 113
|
28
|
|||||
|
Fair value adjustments on deferred cash flow hedges, net of taxes
|
8.1
|
12
|
15
|
20
|
|||||
|
Total fair value adjustments on financial instruments, net of taxes
|
8.1
|
50
|
– 98
|
48
|
|||||
|
Novartis share of other comprehensive income recognized by associated companies, net of taxes
|
|
– 37
|
671
|
– 48
|
|||||
|
Net investment hedge
|
|
– 237
|
|
|
|||||
|
Currency translation effects
|
8.2
|
2 210
|
– 2 391
|
– 1 662
|
|||||
|
Total of items to eventually recycle
|
|
1 986
|
– 1 818
|
– 1 662
|
|||||
|
Other comprehensive income never to be recycled into the consolidated income statement:
|
|
|
|
|
|||||
|
Actuarial gains/(losses) from defined benefit plans, net of taxes
|
8.3
|
851
|
– 515
|
– 147
|
|||||
|
Total comprehensive income
|
|
10 540
|
4 365
|
15 985
|
|||||
|
Attributable to:
|
|
|
|
|
|||||
|
Shareholders of Novartis AG
|
|
10 538
|
4 382
|
15 977
|
|||||
|
Continuing operations
|
|
10 538
|
4 382
|
5 238
|
|||||
|
Discontinued operations
|
|
|
|
10 739
|
|||||
|
Non-controlling interests
|
|
2
|
– 17
|
8
|
|||||
|
The accompanying Notes form an integral part of the consolidated financial statements.
|
|||||||||
|
(USD millions)
|
Note
|
2017
|
2016
|
||||
|
Assets
|
|
|
|
||||
|
Non-current assets
|
|
|
|
||||
|
Property, plant & equipment
|
9
|
16 464
|
15 641
|
||||
|
Goodwill
|
10
|
31 750
|
30 980
|
||||
|
Intangible assets other than goodwill
|
10
|
29 997
|
31 340
|
||||
|
Investments in associated companies
|
4
|
15 370
|
14 304
|
||||
|
Deferred tax assets
|
11
|
8 229
|
10 034
|
||||
|
Financial assets
|
12
|
2 243
|
2 196
|
||||
|
Other non-current assets
|
12
|
818
|
698
|
||||
|
Total non-current assets
|
|
104 871
|
105 193
|
||||
|
Current assets
|
|
|
|
||||
|
Inventories
|
13
|
6 867
|
6 255
|
||||
|
Trade receivables
|
14
|
8 600
|
8 202
|
||||
|
Income tax receivables
|
|
202
|
156
|
||||
|
Marketable securities, commodities, time deposits and derivative financial instruments
|
15
|
625
|
770
|
||||
|
Cash and cash equivalents
|
15
|
8 860
|
7 007
|
||||
|
Other current assets
|
16
|
3 054
|
2 541
|
||||
|
Total current assets
|
|
28 208
|
24 931
|
||||
|
Total assets
|
|
133 079
|
130 124
|
||||
|
|
|
|
|||||
|
Equity and liabilities
|
|
|
|
||||
|
Equity
|
|
|
|
||||
|
Share capital
|
17
|
969
|
972
|
||||
|
Treasury shares
|
17
|
– 100
|
– 76
|
||||
|
Reserves
|
|
73 299
|
73 936
|
||||
|
Issued share capital and reserves attributable to Novartis AG shareholders
|
|
74 168
|
74 832
|
||||
|
Non-controlling interests
|
|
59
|
59
|
||||
|
Total equity
|
|
74 227
|
74 891
|
||||
|
Liabilities
|
|
|
|
||||
|
Non-current liabilities
|
|
|
|
||||
|
Financial debts
|
18
|
23 224
|
17 897
|
||||
|
Deferred tax liabilities
|
11
|
5 168
|
6 657
|
||||
|
Provisions and other non-current liabilities
|
19
|
7 057
|
8 470
|
||||
|
Total non-current liabilities
|
|
35 449
|
33 024
|
||||
|
Current liabilities
|
|
|
|
||||
|
Trade payables
|
|
5 169
|
4 873
|
||||
|
Financial debts and derivative financial instruments
|
20
|
5 308
|
5 905
|
||||
|
Current income tax liabilities
|
|
1 723
|
1 603
|
||||
|
Provisions and other current liabilities
|
21
|
11 203
|
9 828
|
||||
|
Total current liabilities
|
|
23 403
|
22 209
|
||||
|
Total liabilities
|
|
58 852
|
55 233
|
||||
|
Total equity and liabilities
|
|
133 079
|
130 124
|
||||
|
The accompanying Notes form an integral part of the consolidated financial statements.
|
|||||||
|
(USD millions) |
Note |
Share capital |
Treasury shares |
Retained earnings |
Total value adjustments |
Issued share
capital and reserves attributable to Novartis shareholders |
Non- controlling interests |
Total equity |
|||||||||
|
Total equity at January 1, 2015
|
|
1 001
|
– 103
|
72 433
|
– 2 565
|
70 766
|
78
|
70 844
|
|||||||||
|
Net income
|
|
|
|
17 783
|
|
17 783
|
11
|
17 794
|
|||||||||
|
Other comprehensive income
|
8
|
|
|
– 48
|
– 1 758
|
– 1 806
|
– 3
|
– 1 809
|
|||||||||
|
Total comprehensive income
|
|
|
|
17 735
|
– 1 758
|
15 977
|
8
|
15 985
|
|||||||||
|
Dividends
|
17.1
|
|
|
– 6 643
|
|
– 6 643
|
|
– 6 643
|
|||||||||
|
Purchase of treasury shares
|
17.2
|
|
– 33
|
– 6 086
|
|
– 6 119
|
|
– 6 119
|
|||||||||
|
Reduction of share capital
|
|
– 10
|
15
|
– 5
|
|
|
|
|
|||||||||
|
Exercise of options and employee transactions
|
17.2
|
|
14
|
1 578
|
|
1 592
|
|
1 592
|
|||||||||
|
Equity-based compensation
|
17.2
|
|
6
|
809
|
|
815
|
|
815
|
|||||||||
|
Decrease of treasury share repurchase obligation under a share buyback trading plan
|
17.4
|
|
|
658
|
|
658
|
|
658
|
|||||||||
|
Changes in non-controlling interests
|
17.3
|
|
|
|
|
|
– 10
|
– 10
|
|||||||||
|
Fair value adjustments related to divestments
|
8
|
|
|
– 100
|
100
|
|
|
|
|||||||||
|
Total of other equity movements
|
|
– 10
|
2
|
– 9 789
|
100
|
– 9 697
|
– 10
|
– 9 707
|
|||||||||
|
Total equity at December 31, 2015
|
|
991
|
– 101
|
80 379
|
– 4 223
|
77 046
|
76
|
77 122
|
|||||||||
|
Net income
|
|
|
|
6 712
|
|
6 712
|
– 14
|
6 698
|
|||||||||
|
Other comprehensive income
|
8
|
|
|
671
|
– 3 001
|
– 2 330
|
– 3
|
– 2 333
|
|||||||||
|
Total comprehensive income
|
|
|
|
7 383
|
– 3 001
|
4 382
|
– 17
|
4 365
|
|||||||||
|
Dividends
|
17.1
|
|
|
– 6 475
|
|
– 6 475
|
|
– 6 475
|
|||||||||
|
Purchase of treasury shares
|
17.2
|
|
– 7
|
– 985
|
|
– 992
|
|
– 992
|
|||||||||
|
Reduction of share capital
|
|
– 19
|
25
|
– 6
|
|
|
|
|
|||||||||
|
Exercise of options and employee transactions
|
17.2
|
|
2
|
212
|
|
214
|
|
214
|
|||||||||
|
Equity-based compensation
|
17.2
|
|
5
|
659
|
|
664
|
|
664
|
|||||||||
|
Impact of change in ownership of consolidated entities
|
17.5
|
|
|
– 7
|
|
– 7
|
|
– 7
|
|||||||||
|
Fair value adjustments related to divestments
|
8
|
|
|
– 12
|
12
|
|
|
|
|||||||||
|
Total of other equity movements
|
|
– 19
|
25
|
– 6 614
|
12
|
– 6 596
|
|
– 6 596
|
|||||||||
|
Total equity at December 31, 2016
|
|
972
|
– 76
|
81 148
|
– 7 212
|
74 832
|
59
|
74 891
|
|||||||||
|
Net income
|
|
|
|
7 703
|
|
7 703
|
|
7 703
|
|||||||||
|
Other comprehensive income
|
8
|
|
|
– 37
|
2 872
|
2 835
|
2
|
2 837
|
|||||||||
|
Total comprehensive income
|
|
|
|
7 666
|
2 872
|
10 538
|
2
|
10 540
|
|||||||||
|
Dividends
|
17.1
|
|
|
– 6 495
|
|
– 6 495
|
|
– 6 495
|
|||||||||
|
Purchase of treasury shares
|
17.2
|
|
– 36
|
– 5 538
|
|
– 5 574
|
|
– 5 574
|
|||||||||
|
Reduction of share capital
|
|
– 3
|
5
|
– 2
|
|
|
|
|
|||||||||
|
Exercise of options and employee transactions
|
17.2
|
|
2
|
253
|
|
255
|
|
255
|
|||||||||
|
Equity-based compensation
|
17.2
|
|
5
|
607
|
|
612
|
|
612
|
|||||||||
|
Changes in non-controlling interests
|
17.3
|
|
|
|
|
|
– 2
|
– 2
|
|||||||||
|
Total of other equity movements
|
|
– 3
|
– 24
|
– 11 175
|
|
– 11 202
|
– 2
|
– 11 204
|
|||||||||
|
Total equity at December 31, 2017
|
|
969
|
– 100
|
77 639
|
– 4 340
|
74 168
|
59
|
74 227
|
|||||||||
|
The accompanying Notes form an integral part of the consolidated financial statements.
|
|||||||||||||||||
|
(USD millions)
|
Note
|
2017
|
2016
|
2015
|
|||||
|
Net income from continuing operations
|
|
7 703
|
6 698
|
7 028
|
|||||
|
Reversal of non-cash items
|
22.1
|
7 058
|
8 437
|
9 070
|
|||||
|
Dividends received from associated companies and others
|
|
987
|
899
|
432
|
|||||
|
Interest received
|
|
97
|
43
|
34
|
|||||
|
Interest paid
|
|
– 708
|
– 723
|
– 646
|
|||||
|
Other financial receipts
|
|
|
|
714
|
|||||
|
Other financial payments
|
|
– 272
|
– 155
|
– 23
|
|||||
|
Taxes paid 1
|
|
– 1 611
|
– 2 111
|
– 2 454
|
|||||
|
Cash flows before working capital and provision changes from continuing operations
|
|
13 254
|
13 088
|
14 155
|
|||||
|
Payments out of provisions and other net cash movements in non-current liabilities
|
|
– 877
|
– 1 536
|
– 1 207
|
|||||
|
Change in net current assets and other operating cash flow items
|
22.2
|
244
|
– 77
|
– 863
|
|||||
|
Cash flows from operating activities from continuing operations
|
|
12 621
|
11 475
|
12 085
|
|||||
|
Cash flows used in operating activities from discontinued operations 1
|
|
|
|
– 188
|
|||||
|
Total cash flows from operating activities
|
|
12 621
|
11 475
|
11 897
|
|||||
|
Purchase of property, plant & equipment
|
|
– 1 696
|
– 1 862
|
– 2 367
|
|||||
|
Proceeds from sales of property, plant & equipment
|
|
92
|
161
|
237
|
|||||
|
Purchase of intangible assets
|
|
– 1 050
|
– 1 017
|
– 1 138
|
|||||
|
Proceeds from sales of intangible assets
|
|
640
|
847
|
621
|
|||||
|
Purchase of financial assets
|
|
– 468
|
– 247
|
– 264
|
|||||
|
Proceeds from sales of financial assets
|
|
330
|
247
|
166
|
|||||
|
Purchase of other non-current assets
|
|
– 42
|
– 149
|
– 82
|
|||||
|
Proceeds from sales of other non-current assets
|
|
1
|
|
1
|
|||||
|
Divestments of interests in associated companies
|
|
29
|
|
|
|||||
|
Acquisitions and divestments of businesses, net
|
22.3
|
– 784
|
– 765
|
– 16 507
|
|||||
|
Purchase of marketable securities and commodities
|
|
– 580
|
– 530
|
– 595
|
|||||
|
Proceeds from sales of marketable securities and commodities
|
|
549
|
622
|
262
|
|||||
|
Cash flows used in investing activities from continuing operations
|
|
– 2 979
|
– 2 693
|
– 19 666
|
|||||
|
Cash flows used in/from investing activities from discontinued operations 1
|
22.4
|
– 140
|
– 748
|
8 882
|
|||||
|
Total cash flows used in investing activities
|
|
– 3 119
|
– 3 441
|
– 10 784
|
|||||
|
Dividends paid to shareholders of Novartis AG
|
|
– 6 495
|
– 6 475
|
– 6 643
|
|||||
|
Acquisition of treasury shares
|
|
– 5 490
|
– 1 109
|
– 6 071
|
|||||
|
Proceeds from exercise options and other treasury share transactions
|
|
252
|
214
|
1 581
|
|||||
|
Increase in non-current financial debts
|
22.5
|
4 933
|
1 935
|
4 596
|
|||||
|
Repayment of non-current financial debts
|
22.5
|
– 188
|
– 1 696
|
– 3 086
|
|||||
|
Change in current financial debts
|
22.5
|
– 755
|
1 816
|
451
|
|||||
|
Impact of change in ownership of consolidated entities
|
|
0
|
– 6
|
0
|
|||||
|
Dividends paid to non-controlling interests and other financing cash flows
|
|
10
|
7
|
– 4
|
|||||
|
Cash flows used in financing activities
|
|
– 7 733
|
– 5 314
|
– 9 176
|
|||||
|
Effect of exchange rate changes on cash and cash equivalents
|
|
84
|
– 387
|
– 286
|
|||||
|
Net change in cash and cash equivalents
|
|
1 853
|
2 333
|
– 8 349
|
|||||
|
Cash and cash equivalents at January 1
|
|
7 007
|
4 674
|
13 023
|
|||||
|
Cash and cash equivalents at December 31
|
|
8 860
|
7 007
|
4 674
|
|||||
|
The accompanying Notes form an integral part of the consolidated financial statements.
|
|||||||||
|
|
|||||||||
|
1 In 2016, the total net tax payment amounted to USD 2 299 million, of which USD 188 million was included in the cash flows used in investing activities from discontinued operations.
|
|||||||||
|
In 2015, the total net tax payment amounted to USD 3 325 million, of which a refund of USD 94 million was included in the cash flows used in operating activities from discontinued operations, and a USD 965 million payment in the cash flows from investing activities of discontinued operations.
|
|||||||||
Notes to the Novartis Group consolidated financial statements
|
Useful life
|
|||
|
Buildings
|
20 to 40 years
|
||
|
Machinery and other equipment
|
|
||
|
Machinery and equipment
|
7 to 20 years
|
||
|
Furniture and vehicles
|
5 to 10 years
|
||
|
Computer hardware
|
3 to 7 years
|
||
|
Useful life |
Income statement location
for amortization and impairment charges |
||||
|
Currently marketed products
|
5 to 20 years
|
"Cost of goods sold"
|
|||
|
Marketing know-how
|
25 years
|
"Cost of goods sold"
|
|||
|
Technologies
|
10 to 20 years
|
"Cost of goods sold" or "Research and Development"
|
|||
|
Other (including computer software)
|
3 to 7 years
|
In the respective functional expense
|
|||
|
Alcon brand name
|
Not amortized, indefinite useful life
|
Not applicable
|
|||
|
Innovative Medicines
|
Sandoz
|
Alcon
|
Corporate (including eliminations)
|
Group
|
|||||||||||||||||
|
(USD millions)
|
2017
|
2016
|
2017
|
2016
|
2017
|
2016
|
2017
|
2016
|
2017
|
2016
|
|||||||||||
|
Net sales to third parties
|
33 025
|
32 562
|
10 060
|
10 144
|
6 024
|
5 812
|
|
|
49 109
|
48 518
|
|||||||||||
|
Sales to other segments
|
668
|
624
|
118
|
104
|
3
|
|
– 789
|
– 728
|
|
|
|||||||||||
|
Net sales
|
33 693
|
33 186
|
10 178
|
10 248
|
6 027
|
5 812
|
– 789
|
– 728
|
49 109
|
48 518
|
|||||||||||
|
Other revenues
|
898
|
815
|
37
|
37
|
3
|
4
|
88
|
62
|
1 026
|
918
|
|||||||||||
|
Cost of goods sold
|
– 9 007
|
– 9 331
|
– 5 800
|
– 5 971
|
– 3 231
|
– 3 092
|
863
|
874
|
– 17 175
|
– 17 520
|
|||||||||||
|
Gross profit
|
25 584
|
24 670
|
4 415
|
4 314
|
2 799
|
2 724
|
162
|
208
|
32 960
|
31 916
|
|||||||||||
|
Marketing & Sales
|
– 9 089
|
– 8 435
|
– 1 811
|
– 1 681
|
– 1 961
|
– 1 882
|
|
|
– 12 861
|
– 11 998
|
|||||||||||
|
Research & Development
|
– 7 630
|
– 7 709
|
– 774
|
– 814
|
– 568
|
– 516
|
|
|
– 8 972
|
– 9 039
|
|||||||||||
|
General & Administration
|
– 986
|
– 978
|
– 315
|
– 300
|
– 383
|
– 410
|
– 452
|
– 506
|
– 2 136
|
– 2 194
|
|||||||||||
|
Other income
|
1 027
|
1 091
|
204
|
185
|
47
|
48
|
691
|
603
|
1 969
|
1 927
|
|||||||||||
|
Other expense
|
– 1 124
|
– 1 213
|
– 351
|
– 259
|
– 124
|
– 96
|
– 732
|
– 776
|
– 2 331
|
– 2 344
|
|||||||||||
|
Operating income
|
7 782
|
7 426
|
1 368
|
1 445
|
– 190
|
– 132
|
– 331
|
– 471
|
8 629
|
8 268
|
|||||||||||
|
Income from associated companies
|
– 1
|
|
23
|
6
|
|
|
1 086
|
697
|
1 108
|
703
|
|||||||||||
|
Interest expense
|
|
|
|
|
|
|
|
|
– 777
|
– 707
|
|||||||||||
|
Other financial income and expense
|
|
|
|
|
|
|
|
|
39
|
– 447
|
|||||||||||
|
Income before taxes
|
|
|
|
|
|
|
|
|
8 999
|
7 817
|
|||||||||||
|
Taxes
|
|
|
|
|
|
|
|
|
– 1 296
|
– 1 119
|
|||||||||||
|
Net income
|
|
|
|
|
|
|
|
|
|
|
|
7 703
|
6 698
|
|
|||||||
|
Attributable to:
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
|
Shareholders of Novartis AG
|
|
|
|
|
|
|
|
|
7 703
|
6 712
|
|||||||||||
|
Non-controlling interests
|
|
|
|
|
|
|
|
|
0
|
– 14
|
|||||||||||
|
|
|
|
|
|
|
|
|
|
|
||||||||||||
|
Included in net income are:
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
|
Interest income
|
|
|
|
|
|
|
|
|
110
|
43
|
|||||||||||
|
Depreciation of property, plant & equipment
|
– 916
|
– 883
|
– 270
|
– 260
|
– 217
|
– 229
|
– 117
|
– 117
|
– 1 520
|
– 1 489
|
|||||||||||
|
Amortization of intangible assets
|
– 2 291
|
– 2 470
|
– 447
|
– 450
|
– 942
|
– 929
|
– 10
|
– 12
|
– 3 690
|
– 3 861
|
|||||||||||
|
Impairment charges on property, plant & equipment, net
|
– 84
|
– 93
|
– 73
|
– 2
|
|
– 5
|
|
– 2
|
– 157
|
– 102
|
|||||||||||
|
Impairment charges on intangible assets, net
|
– 591
|
– 522
|
– 61
|
– 65
|
– 57
|
– 4
|
|
|
– 709
|
– 591
|
|||||||||||
|
Impairment charges and fair value gains on financial assets, net
|
– 42
|
– 55
|
|
|
– 29
|
|
– 185
|
– 77
|
– 256
|
– 132
|
|||||||||||
|
Additions to restructuring provisions
|
– 122
|
– 236
|
– 61
|
– 46
|
– 8
|
– 36
|
– 3
|
– 25
|
– 194
|
– 343
|
|||||||||||
|
Equity-based compensation of Novartis equity plans
|
– 593
|
– 582
|
– 52
|
– 47
|
– 71
|
– 53
|
– 208
|
– 164
|
– 924
|
– 846
|
|||||||||||
|
|
|||||||||||||||||||||
|
|
|||||||||||||||||||||
|
Innovative Medicines
|
Sandoz
|
Alcon
|
Corporate (including eliminations)
|
Group
|
|||||||||||||||||
|
(USD millions)
|
2016
|
2015
|
2016
|
2015
|
2016
|
2015
|
2016
|
2015
|
2016
|
2015
|
|||||||||||
|
Net sales to third parties from continuing operations
|
32 562
|
33 345
|
10 144
|
10 070
|
5 812
|
5 999
|
|
|
48 518
|
49 414
|
|||||||||||
|
Sales to other segments
|
624
|
518
|
104
|
128
|
|
|
– 728
|
– 620
|
|
26
|
|||||||||||
|
Net sales from continuing operations
|
33 186
|
33 863
|
10 248
|
10 198
|
5 812
|
5 999
|
– 728
|
– 620
|
48 518
|
49 440
|
|||||||||||
|
Other revenues
|
815
|
792
|
37
|
25
|
4
|
23
|
62
|
107
|
918
|
947
|
|||||||||||
|
Cost of goods sold
|
– 9 331
|
– 9 204
|
– 5 971
|
– 5 844
|
– 3 092
|
– 3 145
|
874
|
789
|
– 17 520
|
– 17 404
|
|||||||||||
|
Gross profit from continuing operations
|
24 670
|
25 451
|
4 314
|
4 379
|
2 724
|
2 877
|
208
|
276
|
31 916
|
32 983
|
|||||||||||
|
Marketing & Sales
|
– 8 435
|
– 8 430
|
– 1 681
|
– 1 679
|
– 1 882
|
– 1 663
|
|
|
– 11 998
|
– 11 772
|
|||||||||||
|
Research & Development
|
– 7 709
|
– 7 685
|
– 814
|
– 782
|
– 516
|
– 468
|
|
|
– 9 039
|
– 8 935
|
|||||||||||
|
General & Administration
|
– 978
|
– 1 031
|
– 300
|
– 346
|
– 410
|
– 450
|
– 506
|
– 648
|
– 2 194
|
– 2 475
|
|||||||||||
|
Other income
|
1 091
|
1 149
|
185
|
109
|
48
|
54
|
603
|
737
|
1 927
|
2 049
|
|||||||||||
|
Other expense
|
– 1 213
|
– 1 639
|
– 259
|
– 381
|
– 96
|
– 69
|
– 776
|
– 784
|
– 2 344
|
– 2 873
|
|||||||||||
|
Operating income from continuing operations
|
7 426
|
7 815
|
1 445
|
1 300
|
– 132
|
281
|
– 471
|
– 419
|
8 268
|
8 977
|
|||||||||||
|
Income from associated companies
|
|
|
6
|
2
|
|
|
697
|
264
|
703
|
266
|
|||||||||||
|
Interest expense
|
|
|
|
|
|
|
|
|
– 707
|
– 655
|
|||||||||||
|
Other financial income and expense
|
|
|
|
|
|
|
|
|
– 447
|
– 454
|
|||||||||||
|
Income before taxes from continuing operations
|
|
|
|
|
|
|
|
|
7 817
|
8 134
|
|||||||||||
|
Taxes
|
|
|
|
|
|
|
|
|
– 1 119
|
– 1 106
|
|||||||||||
|
Net income from continuing operations
|
|
|
|
|
|
|
|
|
6 698
|
7 028
|
|||||||||||
|
Net income from discontinued operations
|
|
|
|
|
|
|
|
|
|
10 766
|
|||||||||||
|
Net income
|
|
|
|
|
|
|
|
|
|
|
|
|
6 698
|
17 794
|
|
||||||
|
Attributable to:
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
|
Shareholders of Novartis AG
|
|
|
|
|
|
|
|
|
6 712
|
17 783
|
|||||||||||
|
Non-controlling interests
|
|
|
|
|
|
|
|
|
– 14
|
11
|
|||||||||||
|
|
|
|
|
|
|
|
|
|
|
||||||||||||
|
Included in net income from continuing operations are:
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
|
Interest income
|
|
|
|
|
|
|
|
|
43
|
33
|
|||||||||||
|
Depreciation of property, plant & equipment
|
– 883
|
– 839
|
– 260
|
– 277
|
– 229
|
– 237
|
– 117
|
– 117
|
– 1 489
|
– 1 470
|
|||||||||||
|
Amortization of intangible assets
|
– 2 470
|
– 2 384
|
– 450
|
– 450
|
– 929
|
– 912
|
– 12
|
– 9
|
– 3 861
|
– 3 755
|
|||||||||||
|
Impairment charges on property, plant & equipment, net
|
– 93
|
39
|
– 2
|
– 97
|
– 5
|
– 1
|
– 2
|
– 21
|
– 102
|
– 80
|
|||||||||||
|
Impairment charges on intangible assets, net
|
– 522
|
– 138
|
– 65
|
– 27
|
– 4
|
– 1
|
|
|
– 591
|
– 166
|
|||||||||||
|
Impairment charges and fair value gains on financial assets, net
|
– 55
|
– 32
|
|
|
|
|
– 77
|
– 72
|
– 132
|
– 104
|
|||||||||||
|
Additions to restructuring provisions
|
– 236
|
– 232
|
– 46
|
– 93
|
– 36
|
– 25
|
– 25
|
– 49
|
– 343
|
– 399
|
|||||||||||
|
Equity-based compensation of Novartis equity plans
|
– 582
|
– 620
|
– 47
|
– 53
|
– 53
|
– 66
|
– 164
|
– 164
|
– 846
|
– 903
|
|||||||||||
|
Innovative Medicines
|
Sandoz
|
Alcon
|
Corporate (including eliminations)
|
Group
|
|||||||||||||||||
|
(USD millions)
|
2017
|
2016
|
2017
|
2016
|
2017
|
2016
|
2017
|
2016
|
2017
|
2016
|
|||||||||||
|
Total assets
|
54 075
|
51 911
|
18 231
|
17 611
|
22 014
|
22 970
|
38 759
|
37 632
|
133 079
|
130 124
|
|||||||||||
|
Total liabilities
|
– 11 457
|
– 10 007
|
– 3 459
|
– 3 168
|
– 1 893
|
– 2 520
|
– 42 043
|
– 39 538
|
– 58 852
|
– 55 233
|
|||||||||||
|
Total equity
|
|
|
|
|
|
|
|
|
74 227
|
74 891
|
|||||||||||
|
Net debt
|
|
|
|
|
|
|
|
|
19 047
|
16 025
|
|||||||||||
|
Net operating assets
|
42 618
|
41 904
|
14 772
|
14 443
|
20 121
|
20 450
|
|
|
93 274
|
90 916
|
|||||||||||
|
|
|
|
|
|
|
|
|
|
|
||||||||||||
|
Included in assets and liabilities are:
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
|
Total property, plant & equipment
|
10 857
|
10 410
|
2 525
|
2 374
|
2 403
|
2 163
|
679
|
694
|
16 464
|
15 641
|
|||||||||||
|
Additions to property, plant & equipment 1
|
877
|
996
|
326
|
316
|
431
|
396
|
94
|
127
|
1 728
|
1 835
|
|||||||||||
|
Total goodwill and intangible assets
|
31 571
|
31 630
|
10 993
|
10 774
|
16 176
|
16 914
|
3 007
|
3 002
|
61 747
|
62 320
|
|||||||||||
|
Additions to goodwill and intangible assets 1
|
984
|
865
|
64
|
45
|
82
|
63
|
16
|
5
|
1 146
|
978
|
|||||||||||
|
Total investment in associated companies
|
41
|
16
|
7
|
18
|
|
|
15 322
|
14 270
|
15 370
|
14 304
|
|||||||||||
|
Additions to investment in associated companies
|
6
|
4
|
|
|
|
|
40
|
37
|
46
|
41
|
|||||||||||
|
Cash and cash equivalents, marketable securities, commodities, time deposits and derivative financial instruments
|
|
|
|
|
|
|
9 485
|
7 777
|
9 485
|
7 777
|
|||||||||||
|
Financial debts and derivative financial instruments
|
|
|
|
|
|
|
28 532
|
23 802
|
28 532
|
23 802
|
|||||||||||
|
Current income tax and deferred tax liabilities
|
|
|
|
|
|
|
6 891
|
8 260
|
6 891
|
8 260
|
|||||||||||
|
1 Excluding impact of business combinations
|
|||||||||||||||||||||
|
|
|||||||||||||||||||||
|
Net sales1
|
Total of selected non-current assets2
|
||||||||||||||||||||
|
(USD millions)
|
2017
|
%
|
2016
|
%
|
2015
|
%
|
2017
|
%
|
2016
|
%
|
|||||||||||
|
Country
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
|
Switzerland
|
836
|
2
|
830
|
2
|
774
|
2
|
43 920
|
47
|
44 413
|
48
|
|||||||||||
|
United States
|
16 935
|
34
|
17 117
|
35
|
18 079
|
37
|
28 476
|
30
|
28 484
|
31
|
|||||||||||
|
United Kingdom
|
1 160
|
2
|
1 182
|
2
|
1 277
|
3
|
7 957
|
9
|
6 892
|
7
|
|||||||||||
|
Germany
|
3 690
|
8
|
3 634
|
7
|
3 262
|
7
|
3 128
|
3
|
2 733
|
3
|
|||||||||||
|
France
|
2 490
|
5
|
2 390
|
5
|
2 269
|
5
|
284
|
|
199
|
|
|||||||||||
|
Japan
|
3 177
|
6
|
3 267
|
7
|
3 163
|
6
|
148
|
|
145
|
|
|||||||||||
|
Other
|
20 821
|
43
|
20 098
|
42
|
20 590
|
40
|
9 668
|
11
|
9 399
|
11
|
|||||||||||
|
Group
|
49 109
|
100
|
48 518
|
100
|
49 414
|
100
|
93 581
|
100
|
92 265
|
100
|
|||||||||||
|
|
|
|
|
|
|
|
|
|
|
||||||||||||
|
Region
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
|
Europe
|
17 492
|
36
|
17 079
|
35
|
16 472
|
33
|
61 699
|
66
|
59 879
|
65
|
|||||||||||
|
Americas
|
20 899
|
42
|
20 998
|
43
|
22 414
|
45
|
29 113
|
31
|
29 831
|
32
|
|||||||||||
|
Asia/Africa/Australasia
|
10 718
|
22
|
10 441
|
22
|
10 528
|
22
|
2 769
|
3
|
2 555
|
3
|
|||||||||||
|
Group
|
49 109
|
100
|
48 518
|
100
|
49 414
|
100
|
93 581
|
100
|
92 265
|
100
|
|||||||||||
|
|
|||||||||||||||||||||
|
1 Net sales from operations by location of third-party customer
|
|||||||||||||||||||||
|
2 Total of property, plant and equipment; goodwill; intangible assets; and investment in associated companies
|
|||||||||||||||||||||
|
2017 USD millions |
2016 USD millions |
Change
(2016 to 2017) USD % |
2015 USD millions |
Change
(2015 to 2016) USD % |
|||||||
|
Oncology
|
|
|
|
|
|
||||||
|
Gleevec/Glivec
|
1 943
|
3 323
|
– 42
|
4 658
|
– 29
|
||||||
|
Tasigna
|
1 841
|
1 739
|
6
|
1 632
|
7
|
||||||
|
Sandostatin
|
1 612
|
1 646
|
– 2
|
1 630
|
1
|
||||||
|
Afinitor/Votubia
|
1 525
|
1 516
|
1
|
1 607
|
– 6
|
||||||
|
Exjade/Jadenu
|
1 059
|
956
|
11
|
917
|
4
|
||||||
|
Tafinlar + Mekinist
|
873
|
672
|
30
|
453
|
nm
|
||||||
|
Promacta/Revolade
|
867
|
635
|
37
|
402
|
nm
|
||||||
|
Votrient
|
808
|
729
|
11
|
565
|
nm
|
||||||
|
Jakavi
|
777
|
581
|
34
|
410
|
42
|
||||||
|
Kisqali
|
76
|
0
|
nm
|
0
|
nm
|
||||||
|
Other
|
893
|
993
|
– 10
|
1 030
|
– 4
|
||||||
|
Total Oncology business unit
|
12 274
|
12 790
|
– 4
|
13 304
|
– 4
|
||||||
|
|
|
|
|
|
|||||||
|
Ophthalmology
|
|
|
|
|
|
||||||
|
Lucentis
|
1 888
|
1 835
|
3
|
2 060
|
– 11
|
||||||
|
Travoprost Group
|
589
|
619
|
– 5
|
631
|
– 2
|
||||||
|
Systane Group
|
400
|
377
|
6
|
380
|
– 1
|
||||||
|
Topical Olopatadine Group
|
284
|
335
|
– 15
|
457
|
– 27
|
||||||
|
Other
|
2 207
|
2 297
|
– 4
|
2 395
|
– 4
|
||||||
|
Total Ophthalmology
|
5 368
|
5 463
|
– 2
|
5 923
|
– 8
|
||||||
|
|
|
|
|
|
|||||||
|
Immunology and Dermatology
|
|
|
|
|
|
||||||
|
Cosentyx
|
2 071
|
1 128
|
84
|
261
|
nm
|
||||||
|
Neoral/Sandimmun(e)
|
488
|
515
|
– 5
|
570
|
– 10
|
||||||
|
Zortress/Certican
|
414
|
398
|
4
|
335
|
19
|
||||||
|
Ilaris
|
402
|
283
|
42
|
236
|
20
|
||||||
|
Myfortic
|
378
|
383
|
– 1
|
441
|
– 13
|
||||||
|
Other
|
288
|
308
|
– 6
|
294
|
5
|
||||||
|
Total Immunology and Dermatology
|
4 041
|
3 015
|
34
|
2 137
|
41
|
||||||
|
|
|
|
|
|
|||||||
|
Neuroscience
|
|
|
|
|
|
||||||
|
Gilenya
|
3 185
|
3 109
|
2
|
2 776
|
12
|
||||||
|
Other
|
102
|
124
|
– 18
|
141
|
– 12
|
||||||
|
Total Neuroscience
|
3 287
|
3 233
|
2
|
2 917
|
11
|
||||||
|
2017 USD millions |
2016 USD millions |
Change
(2016 to 2017) USD % |
2015 USD millions |
Change
(2015 to 2016) USD % |
|||||||
|
|
|
|
|
|
|||||||
|
Respiratory
|
|
|
|
|
|
||||||
|
Ultibro Breezhaler
|
411
|
363
|
13
|
260
|
40
|
||||||
|
Seebri Breezhaler
|
151
|
149
|
1
|
150
|
– 1
|
||||||
|
Onbrez Breezhaler
|
112
|
143
|
– 22
|
166
|
– 14
|
||||||
|
Subtotal COPD1 portfolio
|
674
|
655
|
3
|
576
|
14
|
||||||
|
Xolair 2
|
920
|
835
|
10
|
755
|
11
|
||||||
|
Other
|
23
|
31
|
– 26
|
37
|
– 16
|
||||||
|
Total Respiratory
|
1 617
|
1 521
|
6
|
1 368
|
11
|
||||||
|
|
|
|
|
|
|||||||
|
Cardio-Metabolic
|
|
|
|
|
|
||||||
|
Entresto
|
507
|
170
|
198
|
21
|
nm
|
||||||
|
Other
|
17
|
14
|
21
|
0
|
nm
|
||||||
|
Total Cardio-Metabolic
|
524
|
184
|
185
|
21
|
nm
|
||||||
|
|
|
|
|
|
|||||||
|
Established Medicines
|
|
|
|
|
|
||||||
|
Galvus
|
1 233
|
1 193
|
3
|
1 140
|
5
|
||||||
|
Exforge
|
960
|
926
|
4
|
1 047
|
– 12
|
||||||
|
Diovan/Co-Diovan
|
957
|
1 073
|
– 11
|
1 284
|
– 16
|
||||||
|
Voltaren/Cataflam
|
465
|
525
|
– 11
|
558
|
– 6
|
||||||
|
Exelon/Exelon Patch
|
381
|
444
|
– 14
|
728
|
– 39
|
||||||
|
Ritalin/Focalin
|
236
|
282
|
– 16
|
365
|
– 23
|
||||||
|
Other
|
1 682
|
1 913
|
– 12
|
2 553
|
– 25
|
||||||
|
Total Established Medicines
|
5 914
|
6 356
|
– 7
|
7 675
|
– 17
|
||||||
|
|
|
|
|
|
|||||||
|
Total Pharmaceutical business unit
|
20 751
|
19 772
|
5
|
20 041
|
– 1
|
||||||
|
|
|
|
|
|
|||||||
|
Total division net sales
|
33 025
|
32 562
|
1
|
33 345
|
– 2
|
||||||
|
1 Chronic obstructive pulmonary disease
|
|||||||||||
|
2 Net sales reflect Xolair sales for all indications (e.g., including Xolair SAA and Xolair CSU, which is managed by the Immunology and Dermatology franchise)
|
|||||||||||
|
|
|||||||||||
|
nm = not meaningful
|
|||||||||||
|
Net income statement effect
|
Other comprehensive income effect
|
Total comprehensive income effect
|
|||||||||||||||||
|
(USD millions)
|
2017
|
2016
|
2015
|
2017
|
2016
|
2015
|
2017
|
2016
|
2015
|
||||||||||
|
Roche Holding AG, Switzerland
|
456
|
464
|
343
|
108
|
– 39
|
– 149
|
564
|
425
|
194
|
||||||||||
|
GlaxoSmithKline Consumer Healthcare Holdings Ltd., UK
|
629
|
234
|
– 79
|
– 145
|
710
|
– 4
|
484
|
944
|
– 83
|
||||||||||
|
Others
|
23
|
5
|
2
|
|
|
|
23
|
5
|
2
|
||||||||||
|
Associated companies related to continuing operations
|
1 108
|
703
|
266
|
– 37
|
671
|
– 153
|
1 071
|
1 374
|
113
|
||||||||||
|
Balance sheet value
|
|||||
|
(USD millions) |
December 31,
2017 |
December 31,
2016 |
|||
|
Roche Holding AG, Switzerland
|
8 121
|
7 644
|
|||
|
GlaxoSmithKline Consumer Healthcare Holdings Ltd., UK
|
7 020
|
6 448
|
|||
|
Others
|
229
|
212
|
|||
|
Total
|
15 370
|
14 304
|
|||
|
(CHF billions) |
Current assets |
Non-current
assets |
Current
liabilities |
Non-current
liabilities |
|||||
|
December 31, 2016
|
28.7
|
61.4
|
22.6
|
27.8
|
|||||
|
June 30, 2017
|
26.7
|
56.9
|
20.6
|
26.0
|
|||||
|
(CHF billions) |
Revenue |
Net income |
Other
comprehen- sive income |
Total
comprehen- sive income |
|||||
|
December 31, 2016
|
50.6
|
7.5
|
0.7
|
8.2
|
|||||
|
June 30, 2017
|
26.3
|
4.4
|
0.2
|
4.6
|
|||||
|
(USD millions) |
December 31,
2017 |
||
|
Novartis share of Roche’s estimated net assets
|
2 412
|
||
|
Novartis share of re-appraised intangible assets
|
673
|
||
|
Implicit Novartis goodwill
|
2 915
|
||
|
Current value of share in net identifiable assets and goodwill
|
6 000
|
||
|
Accumulated equity accounting adjustments and translation effects less dividends received
|
2 121
|
||
|
Balance sheet value
|
8 121
|
||
|
(USD millions)
|
2017
|
2016
|
2015
|
||||
|
Novartis share of Roche's estimated current-year consolidated net income
|
669
|
678
|
650
|
||||
|
Prior-year adjustment
|
– 67
|
– 68
|
– 157
|
||||
|
Amortization of fair value adjustments relating to intangible assets, net of taxes of USD 42 million (2016: USD 42 million; 2015: USD 41 million)
|
– 146
|
– 146
|
– 150
|
||||
|
Net income effect
|
456
|
464
|
343
|
||||
|
(USD millions) |
December 31,
2017 |
||
|
Novartis share of GSK Consumer Healthcare’s estimated net assets
|
1 505
|
||
|
Novartis share of re-appraised intangible assets
|
3 852
|
||
|
Implicit Novartis goodwill
|
1 763
|
||
|
Current value of share in net identifiable assets and goodwill
|
7 120
|
||
|
Accumulated equity accounting adjustments and translation effects less dividends received
|
– 100
|
||
|
Balance sheet value
|
7 020
|
||
|
(GBP billions) |
Current assets |
Non-current
assets |
Current
liabilities |
Non-current
liabilities |
|||||
|
December 31, 2016
|
4.0
|
21.1
|
3.1
|
2.1
|
|||||
|
September 30, 2017
|
3.3
|
20.6
|
2.6
|
2.0
|
|||||
|
(GBP billions) |
Revenue |
Net income |
Other
comprehen- sive income |
Total
comprehen- sive income |
|||||
|
December 31, 2016
|
6.5
|
0.6
|
1.6
|
2.2
|
|||||
|
September 30, 2017
|
5.3
|
0.6
|
– 0.4
|
0.2
|
|||||
|
(USD millions)
|
2017
|
2016
|
2015
|
||||
|
Novartis share of GSK Consumer Healthcare's estimated current-year consolidated net income
|
589
|
268
|
– 17
|
||||
|
Prior-year adjustment
|
47
|
– 22
|
|
||||
|
Amortization of fair value adjustments relating to intangible assets and inventory, net of taxes of USD 1 million (2016: USD 2 million; 2015: USD 18 million)
|
– 7
|
– 12
|
– 62
|
||||
|
Net income effect
|
629
|
234
|
– 79
|
||||
|
(USD millions)
|
2017
|
2016
|
2015
|
||||
|
Interest expense
|
– 758
|
– 709
|
– 669
|
||||
|
(Expense)/ income arising from discounting long-term liabilities
|
– 19
|
2
|
14
|
||||
|
Total interest expense
|
– 777
|
– 707
|
– 655
|
||||
|
(USD millions)
|
2017
|
2016
|
2015
|
||||
|
Interest income
|
110
|
43
|
33
|
||||
|
Dividend income
|
1
|
1
|
1
|
||||
|
Net capital losses on available-for-sale securities
|
– 1
|
– 1
|
– 8
|
||||
|
Income on forward contracts and options
|
|
|
1
|
||||
|
Impairment of commodities and available-for-sale securities, net
|
12
|
7
|
– 132
|
||||
|
Other financial expense
|
– 25
|
– 20
|
– 23
|
||||
|
Monetary loss from hyperinflation accounting
|
|
|
– 72
|
||||
|
Currency result, net
|
– 58
|
– 477
|
– 254
|
||||
|
Total other financial income and expense
|
39
|
– 447
|
– 454
|
||||
|
(USD millions)
|
2017
|
2016
|
2015
|
||||
|
Switzerland
|
5 289
|
3 110
|
5 765
|
||||
|
Foreign
|
3 710
|
4 707
|
2 369
|
||||
|
Income before taxes from continuing operations
|
8 999
|
7 817
|
8 134
|
||||
|
Income before taxes from discontinued operations
|
|
|
12 479
|
||||
|
Total income before taxes
|
8 999
|
7 817
|
20 613
|
||||
|
(USD millions)
|
2017
|
2016
|
2015
|
||||
|
Switzerland
|
– 462
|
– 709
|
– 317
|
||||
|
Foreign
|
– 1 594
|
– 1 418
|
– 1 333
|
||||
|
Current income tax expense from continuing operations
|
– 2 056
|
– 2 127
|
– 1 650
|
||||
|
Switzerland
|
– 298
|
765
|
– 68
|
||||
|
Foreign
|
1 058
|
243
|
612
|
||||
|
Deferred tax income from continuing operations
|
760
|
1 008
|
544
|
||||
|
Income tax expense from continuing operations
|
– 1 296
|
– 1 119
|
– 1 106
|
||||
|
Income tax expense from discontinued operations
|
|
|
– 1 713
|
||||
|
Total income tax expense
|
– 1 296
|
– 1 119
|
– 2 819
|
||||
|
(As a percentage)
|
2017
|
2016
|
2015
|
||||
|
Applicable tax rate
|
14.5
|
13.2
|
12.4
|
||||
|
Effect of disallowed expenditures
|
3.4
|
3.5
|
3.5
|
||||
|
Effect of utilization of tax losses brought forward from prior periods
|
– 0.1
|
– 0.2
|
– 0.2
|
||||
|
Effect of income taxed at reduced rates
|
– 0.2
|
– 0.2
|
– 0.3
|
||||
|
Effect of tax credits and allowances
|
– 2.2
|
– 2.8
|
– 2.7
|
||||
|
Effect of release of contingent consideration liability
|
– 1.2
|
0.0
|
0.0
|
||||
|
Effect of tax rate change on current and deferred tax assets and liabilities 1
|
0.7
|
0.2
|
– 0.5
|
||||
|
Effect of write-off of deferred tax assets
|
0.0
|
0.5
|
0.0
|
||||
|
Effect of write down and reversal of write-down of investments in subsidiaries
|
– 1.1
|
– 1.0
|
– 0.9
|
||||
|
Effect of tax benefits expiring in 2017
|
– 0.8
|
– 0.5
|
– 0.4
|
||||
|
Effect of non-deductible losses in Venezuela
|
0.0
|
1.3
|
1.2
|
||||
|
Effect of prior year items
|
1.2
|
0.2
|
1.0
|
||||
|
Effect of other items 2
|
0.2
|
0.1
|
0.5
|
||||
|
Effective tax rate for continuing operations
|
14.4
|
14.3
|
13.6
|
||||
|
Effective tax rate for discontinued operations
|
|
|
13.7
|
||||
|
Effective tax rate
|
14.4
|
14.3
|
13.7
|
||||
|
|
|||||||
|
1 Included in 2017 is a 0.7% impact related to the revaluation of the deferred tax assets and liabities and a portion of current tax payables. This revaluation resulted from the US tax reform legislation enacted on December 22, 2017, refer to Note 11 for additional disclosures.
|
|||||||
|
2 Other items in 2016 (+0.1%) include one-time impacts for the deferred tax effects on the net assets of certain subsidiaries resulting from the change in their tax status (-6.2%), the changes in uncertain tax positions (+5.1%) and other items (+1.2%).
|
|||||||
|
2017
|
2016
|
2015
|
|||||
|
Net income attributable to shareholders of Novartis AG (USD millions)
|
|
|
|
||||
|
– Continuing operations
|
7 703
|
6 712
|
7 025
|
||||
|
– Discontinued operations
|
|
|
10 758
|
||||
|
– Total
|
7 703
|
6 712
|
17 783
|
||||
|
|
|
|
|||||
|
Number of shares (in millions)
|
|
|
|
||||
|
Weighted average number of shares outstanding used in basic earnings per share
|
2 346
|
2 378
|
2 403
|
||||
|
Adjustment for vesting of restricted shares, restricted share units and dilutive shares from options
|
25
|
22
|
35
|
||||
|
Weighted average number of shares in diluted earnings per share
|
2 371
|
2 400
|
2 438
|
||||
|
|
|
|
|||||
|
Basic earnings per share (USD)
|
|
|
|
||||
|
– Continuing operations
|
3.28
|
2.82
|
2.92
|
||||
|
– Discontinued operations
|
|
|
4.48
|
||||
|
– Total
|
3.28
|
2.82
|
7.40
|
||||
|
|
|
|
|||||
|
Diluted earnings per share (USD)
|
|
|
|
||||
|
– Continuing operations
|
3.25
|
2.80
|
2.88
|
||||
|
– Discontinued operations
|
|
|
4.41
|
||||
|
– Total
|
3.25
|
2.80
|
7.29
|
||||
|
(USD millions) |
Fair value
adjustments on marketable securities |
Fair value
adjustments on deferred cash flow hedges |
Actuarial
gains/(losses) from defined benefit plans |
Net investment hedge |
Cumulative
currency translation effects |
Total value adjustments |
|||||||
|
Value adjustments at January 1, 2015
|
433
|
– 38
|
– 5 366
|
|
2 406
|
– 2 565
|
|||||||
|
Fair value adjustments on financial instruments
|
28
|
20
|
|
|
|
48
|
|||||||
|
Net actuarial losses from defined benefit plans 1
|
|
|
– 147
|
|
|
– 147
|
|||||||
|
Currency translation effects 2
|
|
|
|
|
– 1 659
|
– 1 659
|
|||||||
|
Total value adjustments in 2015
|
28
|
20
|
– 147
|
|
– 1 659
|
– 1 758
|
|||||||
|
Fair value adjustments related to divestments
|
|
|
100
|
|
|
100
|
|||||||
|
Value adjustments at December 31, 2015
|
461
|
– 18
|
– 5 413
|
|
747
|
– 4 223
|
|||||||
|
Fair value adjustments on financial instruments
|
– 113
|
15
|
|
|
|
– 98
|
|||||||
|
Net actuarial losses from defined benefit plans
|
|
|
– 514
|
|
|
– 514
|
|||||||
|
Currency translation effects
|
|
|
|
|
– 2 389
|
– 2 389
|
|||||||
|
Total value adjustments in 2016
|
– 113
|
15
|
– 514
|
|
– 2 389
|
– 3 001
|
|||||||
|
Fair value adjustments related to divestments
|
|
|
12
|
|
|
12
|
|||||||
|
Value adjustments at December 31, 2016
|
348
|
– 3
|
– 5 915
|
|
– 1 642
|
– 7 212
|
|||||||
|
Fair value adjustments on financial instruments
|
38
|
12
|
|
|
|
50
|
|||||||
|
Net investment hedge
|
|
|
|
– 237
|
|
– 237
|
|||||||
|
Net actuarial gains from defined benefit plans
|
|
|
851
|
|
|
851
|
|||||||
|
Currency translation effects
|
|
|
|
|
2 208
|
2 208
|
|||||||
|
Total value adjustments in 2017
|
38
|
12
|
851
|
– 237
|
2 208
|
2 872
|
|||||||
|
Value adjustments at December 31, 2017
|
386
|
9
|
– 5 064
|
– 237
|
566
|
– 4 340
|
|||||||
|
1 Net actuarial gains of USD 10 million in 2015 were attributable to discontinued operations up to the respective divestment dates
|
|||||||||||||
|
2 Currency translation losses of USD 29 million in 2015 were attributable to discontinued operations up to the respective divestment dates
|
|||||||||||||
|
(USD millions) |
Fair value
adjustments on marketable securities |
Fair value
adjustments on deferred cash flow hedges |
Total |
||||
|
Fair value adjustments at January 1, 2017
|
348
|
– 3
|
345
|
||||
|
Changes in fair value:
|
|
|
|
||||
|
– Available-for-sale marketable securities
|
11
|
|
11
|
||||
|
– Available-for-sale financial investments
|
47
|
|
47
|
||||
|
Realized net gains transferred to the consolidated income statement:
|
|
|
|
||||
|
– Other financial assets sold
|
– 109
|
|
– 109
|
||||
|
Amortized net losses on cash flow hedges transferred to the consolidated income statement
|
|
13
|
13
|
||||
|
Impaired financial assets transferred to the consolidated income statement
|
102
|
|
102
|
||||
|
Deferred tax on above items 1
|
– 13
|
– 1
|
– 14
|
||||
|
Fair value adjustments during the year
|
38
|
12
|
50
|
||||
|
Fair value adjustments at December 31, 2017
|
386
|
9
|
395
|
||||
|
1 Included in 2017 is a USD 18 million impact related to the revaluation of deferred tax liabilities on available-for-sale financial investments held in the US that were previously recognized through other comprehensive income. This revaluation resulted from the US tax reform legislation enacted on December 22, 2017, refer to Note 11 for additional disclosures.
|
|||||||
|
(USD millions) |
Fair value
adjustments on marketable securities |
Fair value
adjustments on deferred cash flow hedges |
Total |
||||
|
Fair value adjustments at January 1, 2016
|
461
|
– 18
|
443
|
||||
|
Changes in fair value:
|
|
|
|
||||
|
– Available-for-sale marketable securities
|
1
|
|
1
|
||||
|
– Available-for-sale financial investments
|
– 87
|
|
– 87
|
||||
|
Realized net gains transferred to the consolidated income statement:
|
|
|
|
||||
|
– Marketable securities sold
|
– 1
|
|
– 1
|
||||
|
– Other financial assets sold
|
– 154
|
|
– 154
|
||||
|
Amortized net losses on cash flow hedges transferred to the consolidated income statement
|
|
16
|
16
|
||||
|
Impaired financial assets transferred to the consolidated income statement
|
131
|
|
131
|
||||
|
Deferred tax on above items
|
– 3
|
– 1
|
– 4
|
||||
|
Fair value adjustments during the year
|
– 113
|
15
|
– 98
|
||||
|
Fair value adjustments at December 31, 2016
|
348
|
– 3
|
345
|
||||
|
(USD millions) |
Fair value
adjustments on marketable securities |
Fair value
adjustments on deferred cash flow hedges |
Total |
||||
|
Fair value adjustments at January 1, 2015
|
433
|
– 38
|
395
|
||||
|
Changes in fair value:
|
|
|
|
||||
|
– Available-for-sale marketable securities
|
– 130
|
|
– 130
|
||||
|
– Available-for-sale financial investments
|
80
|
|
80
|
||||
|
– Associated companies’ movements in comprehensive income
|
– 8
|
|
– 8
|
||||
|
Realized net gains transferred to the consolidated income statement:
|
|
|
|
||||
|
– Marketable securities sold
|
– 1
|
|
– 1
|
||||
|
– Other financial assets sold
|
– 103
|
|
– 103
|
||||
|
Amortized net losses on cash flow hedges transferred to the consolidated income statement
|
|
21
|
21
|
||||
|
Impaired financial assets transferred to the consolidated income statement
|
194
|
|
194
|
||||
|
Deferred tax on above items
|
– 4
|
– 1
|
– 5
|
||||
|
Fair value adjustments during the year
|
28
|
20
|
48
|
||||
|
Fair value adjustments at December 31, 2015
|
461
|
– 18
|
443
|
||||
|
(USD millions)
|
2017
|
2016
|
2015
|
||||
|
Defined benefit pension plans before tax
|
1 367
|
– 667
|
– 252
|
||||
|
Other post-employment benefit plans before tax
|
76
|
12
|
168
|
||||
|
Taxation on above items 1
|
– 592
|
140
|
– 63
|
||||
|
Total after tax
|
851
|
– 515
|
– 147
|
||||
|
Attributable to:
|
|
|
|
||||
|
Shareholders of Novartis AG
|
851
|
– 514
|
– 147
|
||||
|
Non-controlling interests
|
|
– 1
|
|
||||
|
|
|||||||
|
1 Included in 2017 is a USD -272 million impact related to the revaluation of deferred tax assets on US post-employment benefits that were previously recognized through other comprehensive income. This revaluation resulted from the US tax reform legislation enacted on December 22, 2017, refer to Note 11 for additional disclosures.
|
|||||||
|
(USD millions) |
Land |
Buildings |
Construction in progress |
Machinery
& other equipment |
Total |
||||||
|
Cost
|
|
|
|
|
|
||||||
|
January 1, 2017
|
687
|
13 113
|
2 680
|
14 816
|
31 296
|
||||||
|
Reclassifications 1
|
5
|
508
|
– 1 617
|
1 104
|
|
||||||
|
Additions
|
13
|
104
|
1 186
|
425
|
1 728
|
||||||
|
Disposals and derecognitions 2
|
– 23
|
– 324
|
– 71
|
– 593
|
– 1 011
|
||||||
|
Currency translation effects
|
38
|
663
|
190
|
1 106
|
1 997
|
||||||
|
December 31, 2017
|
720
|
14 064
|
2 368
|
16 858
|
34 010
|
||||||
|
|
|
|
|
|
|||||||
|
Accumulated depreciation
|
|
|
|
|
|
||||||
|
January 1, 2017
|
– 40
|
– 5 436
|
– 15
|
– 10 164
|
– 15 655
|
||||||
|
Depreciation charge
|
– 3
|
– 510
|
|
– 1 007
|
– 1 520
|
||||||
|
Accumulated depreciation on disposals and derecognitions 2
|
6
|
275
|
34
|
534
|
849
|
||||||
|
Impairment charge
|
|
– 25
|
– 58
|
– 106
|
– 189
|
||||||
|
Reversal of impairment charge
|
|
|
2
|
30
|
32
|
||||||
|
Currency translation effects
|
– 3
|
– 287
|
– 1
|
– 772
|
– 1 063
|
||||||
|
December 31, 2017
|
– 40
|
– 5 983
|
– 38
|
– 11 485
|
– 17 546
|
||||||
|
Net book value at December 31, 2017
|
680
|
8 081
|
2 330
|
5 373
|
16 464
|
||||||
|
Net book value of property, plant & equipment under finance lease contracts
|
|
|
|
|
78
|
||||||
|
Commitments for purchases of property, plant & equipment
|
|
|
|
|
318
|
||||||
|
Capitalized borrowing costs
|
|
|
|
|
9
|
||||||
|
1 Reclassifications between various asset categories due to completion of plant and other equipment under construction.
|
|||||||||||
|
2 Derecognition of assets that are no longer used and are not considered to have a significant disposal value or other alternative use.
|
|||||||||||
|
(USD millions) |
Land |
Buildings |
Construction in progress |
Machinery
& other equipment |
Total |
||||||
|
Cost
|
|
|
|
|
|
||||||
|
January 1, 2016
|
688
|
12 857
|
2 810
|
15 093
|
31 448
|
||||||
|
Reclassifications 1
|
4
|
630
|
– 1 226
|
592
|
|
||||||
|
Additions
|
24
|
176
|
1 226
|
409
|
1 835
|
||||||
|
Disposals and derecognitions 2
|
– 8
|
– 178
|
– 19
|
– 656
|
– 861
|
||||||
|
Currency translation effects
|
– 21
|
– 372
|
– 111
|
– 622
|
– 1 126
|
||||||
|
December 31, 2016
|
687
|
13 113
|
2 680
|
14 816
|
31 296
|
||||||
|
|
|
|
|
|
|||||||
|
Accumulated depreciation
|
|
|
|
|
|
||||||
|
January 1, 2016
|
– 40
|
– 5 188
|
– 7
|
– 10 231
|
– 15 466
|
||||||
|
Depreciation charge
|
– 3
|
– 530
|
|
– 956
|
– 1 489
|
||||||
|
Accumulated depreciation on disposals and derecognitions 2
|
5
|
157
|
1
|
630
|
793
|
||||||
|
Impairment charge
|
– 3
|
– 47
|
– 11
|
– 61
|
– 122
|
||||||
|
Reversal of impairment charge
|
|
6
|
1
|
13
|
20
|
||||||
|
Currency translation effects
|
1
|
166
|
1
|
441
|
609
|
||||||
|
December 31, 2016
|
– 40
|
– 5 436
|
– 15
|
– 10 164
|
– 15 655
|
||||||
|
Net book value at December 31, 2016
|
647
|
7 677
|
2 665
|
4 652
|
15 641
|
||||||
|
Net book value of property, plant & equipment under finance lease contracts
|
|
|
|
|
81
|
||||||
|
Commitments for purchases of property, plant & equipment
|
|
|
|
|
223
|
||||||
|
Capitalized borrowing costs
|
|
|
|
|
9
|
||||||
|
1 Reclassifications between various asset categories due to completion of plant and other equipment under construction.
|
|||||||||||
|
2 Derecognition of assets that are no longer used and are not considered to have a significant disposal value or other alternative use.
|
|||||||||||
|
Goodwill
|
Intangible Assets other than Goodwill
|
||||||||||||||||
|
(USD millions) |
Total |
Acquired
research & development |
Alcon brand name |
Technologies |
Currently
marketed products |
Marketing know-how |
Other
intangible assets |
Total |
|||||||||
|
Cost
|
|
|
|
|
|
|
|
|
|||||||||
|
January 1, 2017
|
31 381
|
5 150
|
2 980
|
6 548
|
33 007
|
5 960
|
1 492
|
55 137
|
|||||||||
|
Impact of business combinations
|
94
|
1 223
|
|
|
|
|
|
1 223
|
|||||||||
|
Reclassifications 1
|
|
– 389
|
|
|
175
|
|
214
|
|
|||||||||
|
Additions
|
|
697
|
|
5
|
282
|
|
162
|
1 146
|
|||||||||
|
Disposals and derecognitions 2
|
|
– 353
|
|
– 1
|
– 328
|
|
– 64
|
– 746
|
|||||||||
|
Currency translation effects
|
704
|
134
|
|
86
|
969
|
|
48
|
1 237
|
|||||||||
|
December 31, 2017
|
32 179
|
6 462
|
2 980
|
6 638
|
34 105
|
5 960
|
1 852
|
57 997
|
|||||||||
|
|
|
|
|
|
|
|
|
||||||||||
|
Accumulated amortization
|
|
|
|
|
|
|
|
|
|||||||||
|
January 1, 2017
|
– 401
|
– 886
|
|
– 3 637
|
– 16 863
|
– 1 430
|
– 981
|
– 23 797
|
|||||||||
|
Reclassifications 1
|
|
6
|
|
|
– 6
|
|
|
|
|||||||||
|
Amortization charge
|
|
|
|
– 577
|
|
– 2 571
|
– 238
|
– 304
|
– 3 690
|
||||||||
|
Accumulated impairments on disposals and derecognitions2
|
|
352
|
|
|
317
|
|
61
|
730
|
|||||||||
|
Impairment charge
|
|
– 615
|
|
|
– 92
|
|
– 2
|
– 709
|
|||||||||
|
Currency translation effects
|
– 28
|
– 27
|
|
– 54
|
– 416
|
|
– 37
|
– 534
|
|||||||||
|
December 31, 2017
|
– 429
|
– 1 170
|
|
– 4 268
|
– 19 631
|
– 1 668
|
– 1 263
|
– 28 000
|
|||||||||
|
Net book value at December 31, 2017
|
31 750
|
5 292
|
2 980
|
2 370
|
14 474
|
4 292
|
589
|
29 997
|
|||||||||
|
1 Reclassifications between various asset categories as a result of product launches of acquired In-Process Research & Development and completion of software development.
|
|||||||||||||||||
|
2 Derecognitions of assets that are no longer used or being developed and are not considered to have a significant disposal value or other alternative use.
|
|||||||||||||||||
|
Goodwill
|
Intangible Assets other than Goodwill
|
||||||||||||||||
|
(USD millions) |
Total |
Acquired
research & development |
Alcon brand name |
Technologies |
Currently
marketed products |
Marketing know-how |
Other
intangible assets |
Total |
|||||||||
|
Cost
|
|
|
|
|
|
|
|
|
|||||||||
|
January 1, 2016
|
31 585
|
4 119
|
2 980
|
6 563
|
33 385
|
5 960
|
1 341
|
54 348
|
|||||||||
|
Impact of business combinations
|
56
|
690
|
|
|
451
|
|
|
1 141
|
|||||||||
|
Reclassifications 1
|
|
– 158
|
|
|
6
|
|
152
|
|
|||||||||
|
Additions
|
|
599
|
|
|
223
|
|
156
|
978
|
|||||||||
|
Disposals and derecognitions 2
|
|
– 23
|
|
|
– 464
|
|
– 130
|
– 617
|
|||||||||
|
Currency translation effects
|
– 260
|
– 77
|
|
– 15
|
– 594
|
|
– 27
|
– 713
|
|||||||||
|
December 31, 2016
|
31 381
|
5 150
|
2 980
|
6 548
|
33 007
|
5 960
|
1 492
|
55 137
|
|||||||||
|
|
|
|
|
|
|
|
|
||||||||||
|
Accumulated amortization
|
|
|
|
|
|
|
|
|
|||||||||
|
January 1, 2016
|
– 411
|
– 650
|
|
– 3 070
|
– 14 221
|
– 1 192
|
– 998
|
– 20 131
|
|||||||||
|
Reclassifications 1
|
|
225
|
|
|
– 225
|
|
|
|
|||||||||
|
Amortization charge
|
|
|
|
– 576
|
– 2 926
|
– 238
|
– 121
|
– 3 861
|
|||||||||
|
Accumulated impairments on disposals and derecognitions2
|
|
22
|
|
|
390
|
|
123
|
535
|
|||||||||
|
Impairment charge
|
|
– 490
|
|
|
– 96
|
|
– 5
|
– 591
|
|||||||||
|
Currency translation effects
|
10
|
7
|
|
9
|
215
|
|
20
|
251
|
|||||||||
|
December 31, 2016
|
– 401
|
– 886
|
|
– 3 637
|
– 16 863
|
– 1 430
|
– 981
|
– 23 797
|
|||||||||
|
Net book value at December 31, 2016
|
30 980
|
4 264
|
2 980
|
2 911
|
16 144
|
4 530
|
511
|
31 340
|
|||||||||
|
1 Reclassifications between various asset categories as a result of product launches of acquired In-Process Research & Development and completion of software development.
|
|||||||||||||||||
|
2 Derecognitions of assets that are no longer used or being developed and are not considered to have a significant disposal value or other alternative use.
|
|||||||||||||||||
|
Goodwill
|
Intangible Assets other than Goodwill
|
||||||||||||||||
|
(USD millions) |
Total |
Acquired
research & development |
Alcon brand name |
Technologies |
Currently
marketed products |
Marketing know-how |
Other
intangible assets |
Total |
|||||||||
|
Innovative Medicines
|
15 237
|
4 368
|
|
9
|
11 604
|
|
353
|
16 334
|
|||||||||
|
Sandoz
|
8 210
|
625
|
|
539
|
1 589
|
|
30
|
2 783
|
|||||||||
|
Alcon
|
8 295
|
291
|
|
1 822
|
1 281
|
4 292
|
195
|
7 881
|
|||||||||
|
Corporate
|
8
|
8
|
2 980
|
|
|
|
11
|
2 999
|
|||||||||
|
Net book value at December 31, 2017
|
31 750
|
5 292
|
2 980
|
2 370
|
14 474
|
4 292
|
589
|
29 997
|
|||||||||
|
Goodwill
|
Intangible Assets other than Goodwill
|
||||||||||||||||
|
(USD millions) |
Total |
Acquired
research & development |
Alcon brand name |
Technologies |
Currently
marketed products |
Marketing know-how |
Other
intangible assets |
Total |
|||||||||
|
Innovative Medicines
|
15 010
|
3 512
|
|
11
|
12 821
|
|
276
|
16 620
|
|||||||||
|
Sandoz
|
7 669
|
613
|
|
563
|
1 904
|
|
25
|
3 105
|
|||||||||
|
Alcon
|
8 293
|
139
|
|
2 337
|
1 419
|
4 530
|
196
|
8 621
|
|||||||||
|
Corporate
|
8
|
|
2 980
|
|
|
|
14
|
2 994
|
|||||||||
|
Net book value at December 31, 2016
|
30 980
|
4 264
|
2 980
|
2 911
|
16 144
|
4 530
|
511
|
31 340
|
|||||||||
|
(As a percentage) |
Innovative
Medicines |
Sandoz |
Alcon |
Corporate |
|||||
|
Terminal growth rate
|
1.5
|
2.0
|
3.0
|
2.6
|
|||||
|
Discount rate (post-tax)
|
7.0
|
7.0
|
7.0
|
7.0
|
|||||
|
(USD millions)
|
2017
|
2016
|
|||
|
Innovative Medicines
|
– 591
|
– 522
|
|||
|
Sandoz
|
– 61
|
– 65
|
|||
|
Alcon
|
– 57
|
– 4
|
|||
|
Total
|
– 709
|
– 591
|
|||
|
(USD millions) |
Property, plant & equipment |
Intangible assets |
Pensions and
other benefit obligations of associates |
Inventories |
Tax loss carry- forwards |
Other assets, provisions and accruals |
Total |
||||||||
|
Gross deferred tax assets at January 1, 2017
|
224
|
1 331
|
1 839
|
4 160
|
146
|
2 597
|
10 297
|
||||||||
|
Gross deferred tax liabilities at January 1, 2017
|
– 629
|
– 4 019
|
– 358
|
– 511
|
|
– 1 403
|
– 6 920
|
||||||||
|
Net deferred tax balance at January 1, 2017
|
– 405
|
– 2 688
|
1 481
|
3 649
|
146
|
1 194
|
3 377
|
||||||||
|
|
|
|
|
|
|
|
|||||||||
|
At January 1, 2017
|
– 405
|
– 2 688
|
1 481
|
3 649
|
146
|
1 194
|
3 377
|
||||||||
|
Credited/(charged) to income
|
– 30
|
1 279
|
– 90
|
– 304
|
– 49
|
– 46
|
760
|
||||||||
|
Charged to equity
|
|
|
|
|
|
– 101
|
– 101
|
||||||||
|
Charged to other comprehensive income
|
|
|
– 592
|
|
|
– 69
|
– 661
|
||||||||
|
Impact of business combinations
|
|
– 322
|
|
|
5
|
|
– 317
|
||||||||
|
Other movements
|
– 41
|
33
|
37
|
– 14
|
– 14
|
2
|
3
|
||||||||
|
Net deferred tax balance at December 31, 2017
|
– 476
|
– 1 698
|
836
|
3 331
|
88
|
980
|
3 061
|
||||||||
|
|
|
|
|
|
|
|
|||||||||
|
Gross deferred tax assets at December 31, 2017
|
137
|
1 287
|
1 090
|
3 786
|
97
|
1 983
|
8 380
|
||||||||
|
Gross deferred tax liabilities at December 31, 2017
|
– 613
|
– 2 985
|
– 254
|
– 455
|
– 9
|
– 1 003
|
– 5 319
|
||||||||
|
Net deferred tax balance at December 31, 2017
|
– 476
|
– 1 698
|
836
|
3 331
|
88
|
980
|
3 061
|
||||||||
|
|
|
|
|
|
|
|
|||||||||
|
After offsetting the following amount of deferred tax assets and liabilities within the same tax jurisdiction the balance amounts to:
|
|
|
|
|
|
|
|
|
|
|
|
|
151
|
||
|
Deferred tax assets at December 31, 2017
|
|
|
|
|
|
|
8 229
|
||||||||
|
Deferred tax liabilities at December 31, 2017
|
|
|
|
|
|
|
– 5 168
|
||||||||
|
Net deferred tax balance at December 31, 2017
|
|
|
|
|
|
|
3 061
|
||||||||
|
|
|
|
|
|
|
|
|||||||||
|
|
|
|
|
|
|
|
|||||||||
|
Gross deferred tax assets at January 1, 2016
|
216
|
611
|
1 730
|
3 821
|
62
|
2 866
|
9 306
|
||||||||
|
Gross deferred tax liabilities at January 1, 2016
|
– 639
|
– 3 962
|
– 401
|
– 565
|
– 5
|
– 1 132
|
– 6 704
|
||||||||
|
Net deferred tax balance at January 1, 2016
|
– 423
|
– 3 351
|
1 329
|
3 256
|
57
|
1 734
|
2 602
|
||||||||
|
|
|
|
|
|
|
|
|||||||||
|
At January 1, 2016
|
– 423
|
– 3 351
|
1 329
|
3 256
|
57
|
1 734
|
2 602
|
||||||||
|
Credited/(charged) to income
|
– 13
|
1 057
|
53
|
373
|
55
|
– 517
|
1 008
|
||||||||
|
Charged to equity
|
|
|
|
|
|
– 44
|
– 44
|
||||||||
|
Credited/(charged) to other comprehensive income
|
|
|
140
|
|
|
– 2
|
138
|
||||||||
|
Impact of business combinations
|
4
|
– 400
|
|
|
23
|
37
|
– 336
|
||||||||
|
Other movements
|
27
|
6
|
– 41
|
20
|
11
|
– 14
|
9
|
||||||||
|
Net deferred tax balance at December 31, 2016
|
– 405
|
– 2 688
|
1 481
|
3 649
|
146
|
1 194
|
3 377
|
||||||||
|
|
|
|
|
|
|
|
|||||||||
|
Gross deferred tax assets at December 31, 2016
|
224
|
1 331
|
1 839
|
4 160
|
146
|
2 597
|
10 297
|
||||||||
|
Gross deferred tax liabilities at December 31, 2016
|
– 629
|
– 4 019
|
– 358
|
– 511
|
|
– 1 403
|
– 6 920
|
||||||||
|
Net deferred tax balance at December 31, 2016
|
– 405
|
– 2 688
|
1 481
|
3 649
|
146
|
1 194
|
3 377
|
||||||||
|
|
|
|
|
|
|
|
|||||||||
|
After offsetting the following amount of deferred tax assets and liabilities within the same tax jurisdiction the balance amounts to:
|
|
|
|
|
|
|
|
|
|
|
|
|
263
|
||
|
Deferred tax assets at December 31, 2016
|
|
|
|
|
|
|
10 034
|
||||||||
|
Deferred tax liabilities at December 31, 2016
|
|
|
|
|
|
|
– 6 657
|
||||||||
|
Net deferred tax balance at December 31, 2016
|
|
|
|
|
|
|
3 377
|
||||||||
|
(USD billions)
|
2017
|
2016
|
|||
|
Expected to have an impact on current tax payable after more than 12 months
|
|
|
|||
|
– Deferred tax assets
|
3.5
|
4.8
|
|||
|
– Deferred tax liabilities
|
4.4
|
5.9
|
|||
|
(USD billions)
|
2017
|
2016
|
|||
|
Unremitted earnings that have been retained by consolidated entities for reinvestment
|
66
|
63
|
|||
|
(USD billions)
|
2017
|
2016
|
|||
|
Investments in subsidiaries
|
3
|
2
|
|||
|
Goodwill from acquisitions
|
– 29
|
– 28
|
|||
|
(USD millions)
|
Not capitalized
|
Capitalized
|
2017 total
|
||||
|
One year
|
37
|
3
|
40
|
||||
|
Two years
|
64
|
4
|
68
|
||||
|
Three years
|
87
|
5
|
92
|
||||
|
Four years
|
26
|
25
|
51
|
||||
|
Five years
|
67
|
16
|
83
|
||||
|
More than five years
|
654
|
1 671
|
2 325
|
||||
|
Total
|
935
|
1 724
|
2 659
|
||||
|
(USD millions)
|
Not capitalized
|
Capitalized
|
2016 total
|
||||
|
One year
|
21
|
12
|
33
|
||||
|
Two years
|
30
|
5
|
35
|
||||
|
Three years
|
50
|
5
|
55
|
||||
|
Four years
|
75
|
3
|
78
|
||||
|
Five years
|
73
|
25
|
98
|
||||
|
More than five years
|
405
|
1 913
|
2 318
|
||||
|
Total
|
654
|
1 963
|
2 617
|
||||
|
(USD millions)
|
2017
|
2016
|
2015
|
||||
|
Tax losses carried forward that expired
|
1
|
19
|
13
|
||||
|
(USD millions) |
Income
statement |
Equity |
Total |
||||
|
Deferred tax asset and liability revaluation
|
|
|
|
||||
|
Items previously recognized in consolidated income statement
|
– 24
|
|
– 24
|
||||
|
Items previously recognized in other comprehensive income 1
|
|
– 254
|
– 254
|
||||
|
Items previously recognized in retained earnings 2
|
|
– 71
|
– 71
|
||||
|
Total revaluation of deferred tax assets and liabilities
|
– 24
|
– 325
|
– 349
|
||||
|
Total revaluation of current tax payables
|
– 37
|
|
– 37
|
||||
|
Total revaluation of deferred tax assets and liabilities and current income tax liabilities
|
– 61
|
– 325
|
– 386
|
||||
|
1 Related to post-employment benefits and available for sale financial investments.
|
|||||||
|
2 Related to equity based compensation plans.
|
|||||||
|
(USD millions)
|
2017
|
2016
|
|||
|
Available-for-sale long-term financial investments
|
1 275
|
1 096
|
|||
|
Long-term receivables from customers
|
197
|
231
|
|||
|
Minimum lease payments from finance lease agreements
|
122
|
147
|
|||
|
Contingent consideration receivables 1
|
394
|
586
|
|||
|
Long-term loans, advances and security deposits
|
255
|
136
|
|||
|
Total financial assets
|
2 243
|
2 196
|
|||
|
1 Note 28 provides additional disclosures related to contingent considerations.
|
|||||
|
(USD millions)
|
2017
|
2016
|
|||
|
Deferred compensation plans
|
484
|
451
|
|||
|
Prepaid post-employment benefit plans
|
133
|
47
|
|||
|
Other non-current assets
|
201
|
200
|
|||
|
Total other non-current assets
|
818
|
698
|
|||
|
2017
|
2016
|
||||||||||||||||||||
|
(USD millions) |
Total
future payments |
Unearned
finance income |
Present value |
Provision |
Net
book value |
Total
future payments |
Unearned
finance income |
Present value |
Provision |
Net
book value |
|||||||||||
|
Not later than one year 1
|
83
|
– 7
|
76
|
– 3
|
73
|
91
|
– 5
|
86
|
– 2
|
84
|
|||||||||||
|
Between one and five years
|
180
|
– 14
|
166
|
– 59
|
107
|
182
|
– 16
|
166
|
– 37
|
129
|
|||||||||||
|
Later than five years
|
31
|
– 2
|
29
|
– 14
|
15
|
63
|
– 4
|
59
|
– 41
|
18
|
|||||||||||
|
Total
|
294
|
– 23
|
271
|
– 76
|
195
|
336
|
– 25
|
311
|
– 80
|
231
|
|||||||||||
|
1 The current portion of the minimum lease payments is recorded in trade receivables or other current assets (to the extent not yet invoiced).
|
|||||||||||||||||||||
|
(USD millions)
|
2017
|
2016
|
|||
|
Raw material, consumables
|
841
|
705
|
|||
|
Work in progress
|
2 957
|
2 700
|
|||
|
Finished products
|
3 069
|
2 850
|
|||
|
Total inventories
|
6 867
|
6 255
|
|||
|
(USD billions)
|
2017
|
2016
|
2015
|
||||
|
Cost of goods sold
|
– 10.3
|
– 10.3
|
– 10.5
|
||||
|
(USD millions)
|
2017
|
2016
|
2015
|
||||
|
Inventory provisions
|
– 470
|
– 283
|
– 356
|
||||
|
Reversals of inventory provisions
|
189
|
67
|
148
|
||||
|
(USD millions)
|
2017
|
2016
|
|||
|
Total gross trade receivables
|
8 790
|
8 364
|
|||
|
Provisions for doubtful trade receivables
|
– 190
|
– 162
|
|||
|
Total trade receivables, net
|
8 600
|
8 202
|
|||
|
(USD millions)
|
2017
|
2016
|
2015
|
||||
|
January 1
|
– 162
|
– 142
|
– 156
|
||||
|
Impact of divestments
|
12
|
|
|
||||
|
Provisions for doubtful trade receivables charged to the consolidated income statement
|
– 119
|
– 76
|
– 68
|
||||
|
Utilization provisions for doubtful trade receivables
|
12
|
17
|
39
|
||||
|
Reversal of provisions for doubtful trade receivables
|
76
|
37
|
32
|
||||
|
Currency translation effects
|
– 9
|
2
|
11
|
||||
|
December 31
|
– 190
|
– 162
|
– 142
|
||||
|
(USD millions)
|
2017
|
2016
|
|||
|
Not overdue
|
7 758
|
7 386
|
|||
|
Past due for not more than one month
|
279
|
262
|
|||
|
Past due for more than one month but less than three months
|
230
|
223
|
|||
|
Past due for more than three months but less than six months
|
137
|
185
|
|||
|
Past due for more than six months but less than one year
|
137
|
145
|
|||
|
Past due for more than one year
|
249
|
163
|
|||
|
Provisions for doubtful trade receivables
|
– 190
|
– 162
|
|||
|
Total trade receivables, net
|
8 600
|
8 202
|
|||
|
(USD millions)
|
2017
|
2016
|
|||
|
Total balance of gross trade receivables from closely monitored countries
|
1 733
|
1 717
|
|||
|
Past due for more than one year
|
124
|
82
|
|||
|
Provisions
|
95
|
63
|
|||
|
(USD millions)
|
2017
|
2016
|
|||
|
US dollar (USD)
|
3 451
|
3 432
|
|||
|
Euro (EUR)
|
1 533
|
1 366
|
|||
|
Japanese yen (JPY)
|
600
|
567
|
|||
|
Chinese yuan (CNY)
|
312
|
264
|
|||
|
Russian ruble (RUB)
|
268
|
347
|
|||
|
Brazilian real (BRL)
|
237
|
222
|
|||
|
British pound (GBP)
|
208
|
160
|
|||
|
Australian dollar (AUD)
|
165
|
147
|
|||
|
Swiss franc (CHF)
|
127
|
135
|
|||
|
Canadian dollar (CAD)
|
73
|
97
|
|||
|
Other currencies
|
1 626
|
1 465
|
|||
|
Total trade receivables, net
|
8 600
|
8 202
|
|||
|
(USD millions)
|
2017
|
2016
|
|||
|
Debt securities
|
328
|
306
|
|||
|
Fund investments
|
34
|
31
|
|||
|
Total available-for-sale marketable securities
|
362
|
337
|
|||
|
Commodities
|
106
|
94
|
|||
|
Time deposits with original maturity more than 90 days
|
125
|
108
|
|||
|
Derivative financial instruments
|
31
|
230
|
|||
|
Accrued interest on debt securities and time deposits
|
1
|
1
|
|||
|
Total marketable securities, commodities, time deposits and derivative financial instruments
|
625
|
770
|
|||
|
(USD millions)
|
2017
|
2016
|
|||
|
US dollar (USD)
|
303
|
284
|
|||
|
Euro (EUR)
|
14
|
|
12
|
||
|
Japanese yen (JPY)
|
11
|
10
|
|||
|
Total debt securities
|
328
|
306
|
|||
|
(USD millions)
|
2017
|
2016
|
|||
|
Current accounts
|
2 970
|
1 912
|
|||
|
Time deposits and short-term investments with original maturity less than 90 days
|
5 890
|
5 095
|
|||
|
Total cash and cash equivalents
|
8 860
|
7 007
|
|||
|
(USD millions)
|
2017
|
2016
|
|||
|
VAT receivable
|
717
|
521
|
|||
|
Withholding tax recoverable
|
93
|
282
|
|||
|
Prepaid expenses
|
|
|
|||
|
– Third parties
|
753
|
692
|
|||
|
– Associated companies
|
3
|
5
|
|||
|
Receivables from associated companies
|
8
|
7
|
|||
|
Contingent consideration receivable 1
|
450
|
|
|||
|
Other receivables and current assets
|
1 030
|
1 034
|
|||
|
Total other current assets
|
3 054
|
2 541
|
|||
|
1 Note 28 provides additional disclosures related to contingent consideration.
|
|||||
|
(USD millions) |
Jan 1, 2015 |
Movement
in year |
Dec 31, 2015 |
Movement
in year |
Dec 31, 2016 |
Movement
in year |
Dec 31, 2017 |
||||||||
|
Share capital
|
1 001
|
– 10
|
991
|
– 19
|
972
|
– 3
|
969
|
||||||||
|
Treasury shares
|
– 103
|
2
|
– 101
|
25
|
– 76
|
– 24
|
– 100
|
||||||||
|
Outstanding share capital
|
898
|
– 8
|
890
|
6
|
896
|
– 27
|
869
|
||||||||
|
|
2017
|
2016
|
2015
|
||||||||||||||||||
|
Number of outstanding shares (in millions) |
Note |
Total
Novartis shares |
Total
treasury shares |
Total
outstanding shares |
Total
Novartis shares |
Total
treasury shares |
Total
outstanding shares |
Total
Novartis shares |
Total
treasury shares |
Total
outstanding shares |
|||||||||||
|
Balance at beginning of year
|
|
2 627.1
|
– 253.0
|
2 374.1
|
2 677.0
|
– 303.1
|
2 373.9
|
2 706.2
|
– 307.6
|
2 398.6
|
|||||||||||
|
Shares canceled for capital reduction 1
|
|
– 10.3
|
10.3
|
|
– 49.9
|
49.9
|
|
– 29.2
|
29.2
|
|
|||||||||||
|
Shares acquired to be held in Group Treasury 2
|
|
|
|
|
|
|
|
|
– 9.6
|
– 9.6
|
|||||||||||
|
Shares acquired to be canceled 3
|
|
|
– 66.2
|
– 66.2
|
|
– 10.3
|
– 10.3
|
|
– 49.9
|
– 49.9
|
|||||||||||
|
Other share purchases 4
|
|
|
– 3.8
|
– 3.8
|
|
– 2.6
|
– 2.6
|
|
– 4.1
|
– 4.1
|
|||||||||||
|
Exercise of options and employee transactions 5
|
17.6
|
|
4.6
|
4.6
|
|
4.1
|
4.1
|
|
27.0
|
27.0
|
|||||||||||
|
Equity-based compensation 5
|
|
|
8.8
|
8.8
|
|
9.0
|
9.0
|
|
11.9
|
11.9
|
|||||||||||
|
Total movements
|
|
– 10.3
|
– 46.3
|
– 56.6
|
– 49.9
|
50.1
|
0.2
|
– 29.2
|
4.5
|
– 24.7
|
|||||||||||
|
Balance at end of year
|
|
2 616.8
|
– 299.3
|
2 317.5
|
2 627.1
|
– 253.0
|
2 374.1
|
2 677.0
|
– 303.1
|
2 373.9
|
|||||||||||
|
|
|||||||||||||||||||||
|
1 Novartis reduced its share capital by cancelling shares which were repurchased on the SIX Swiss Exchange second trading line during previous years.
|
|||||||||||||||||||||
|
2 Shares repurchased on the SIX Swiss Exchange first trading line
|
|||||||||||||||||||||
|
3 For 2017 and 2016, shares repurchased on the SIX Swiss Exchange second trading line under the CHF 10 billion share buyback authority approved at the 2016 Annual General Meeting (AGM). For 2015, shares repurchased on the SIX Swiss Exchange second trading line under the CHF 10 billion share buyback authority approved at the 2008 Annual General Meeting (AGM).
|
|||||||||||||||||||||
|
4 Shares acquired from employees, which were previously granted to them under the respective programs
|
|||||||||||||||||||||
|
5 Shares delivered as a result of options being exercised and physical share deliveries related to equity-based participation plans
|
|||||||||||||||||||||
|
2017
|
2016
|
2015
|
|||||
|
Dividend per share (in CHF)
|
2.75
|
2.70
|
2.60
|
||||
|
Total dividend payment (in USD billion)
|
6.5
|
6.5
|
6.6
|
||||
|
|
2017
|
2016
|
2015
|
||||||||||||
|
Note |
Number of
outstanding shares (in millions) |
Equity impact USDm |
Number of
outstanding shares (in millions) |
Equity impact USDm |
Number of
outstanding shares (in millions) |
Equity impact USDm |
|||||||||
|
Shares acquired to be held in Group Treasury 1
|
|
|
|
|
|
– 9.6
|
– 897
|
||||||||
|
Shares acquired to be canceled 2
|
|
– 66.2
|
– 5 270
|
– 10.3
|
– 784
|
– 49.9
|
– 4 805
|
||||||||
|
Other share purchases 3
|
|
– 3.8
|
– 304
|
– 2.6
|
– 208
|
– 4.1
|
– 417
|
||||||||
|
Purchase of treasury shares
|
|
– 70.0
|
– 5 574
|
– 12.9
|
– 992
|
– 63.6
|
– 6 119
|
||||||||
|
Exercise of options and employee transactions 4
|
17.6
|
4.6
|
255
|
4.1
|
214
|
27.0
|
1 592
|
||||||||
|
Equity-based compensation 5, 6
|
|
8.8
|
612
|
9.0
|
664
|
11.9
|
815
|
||||||||
|
Total
|
|
– 56.6
|
– 4 707
|
0.2
|
– 114
|
– 24.7
|
– 3 712
|
||||||||
|
|
|||||||||||||||
|
1 Shares repurchased on the SIX Swiss Exchange first trading line
|
|||||||||||||||
|
2 For 2017 and 2016, shares repurchased on the SIX Swiss Exchange second trading line under the CHF 10 billion share buyback authority approved at the 2016 Annual General Meeting (AGM). For 2015, shares repurchased on the SIX Swiss Exchange second trading line under the CHF 10 billion share buyback authority approved at the 2008 Annual General Meeting (AGM).
|
|||||||||||||||
|
3 Shares acquired from employees, which were previously granted to them under the respective programs
|
|||||||||||||||
|
4 Shares delivered as a result of options being exercised related to equity-based participation plans and the delivery of treasury shares. The average share price of the shares delivered was significantly below market price reflecting the strike price of the options exercised.
|
|||||||||||||||
|
5 Equity-settled share-based compensation is expensed in the consolidated income statement in accordance with the vesting period of the share-based compensation plans. The value for the shares and options granted is credited to consolidated equity over the respective vesting period. In addition, tax benefits arising from tax deductible amounts exceeding the expense recognized in the income statement are credited to equity.
|
|||||||||||||||
|
6 Included in 2017 is a USD 71 million impact related to the revaluation of deferred tax assets on equity based compensation that were previously recognized through retained earnings. This revaluation resulted from the US tax reform legislation enacted on December 22, 2017, refer to Note 11 for additional disclosures.
|
|||||||||||||||
|
(USD millions)
|
2017
|
2016
|
|||
|
Straight bonds
|
22 957
|
17 285
|
|||
|
Liabilities to banks and other financial institutions 1
|
539
|
708
|
|||
|
Finance lease obligations
|
87
|
82
|
|||
|
Total, including current portion of non-current financial debt
|
23 583
|
18 075
|
|||
|
Less current portion of non-current financial debt
|
– 359
|
– 178
|
|||
|
Total non-current financial debts
|
23 224
|
17 897
|
|||
|
1 Average interest rate 0.3% (2016: 0.4%)
|
|||||
|
Coupon |
Currency |
Nominal amount |
Issuance year |
Maturity year |
Issuer |
Issue price |
2017
(USD millions) |
2016
(USD millions) |
|||||||||
|
5.125%
|
USD
|
3 000
|
2009
|
2019
|
Novartis Securities Investment Ltd., Hamilton, Bermuda
|
99.822%
|
2 997
|
2 995
|
|||||||||
|
4.400%
|
USD
|
1 000
|
2010
|
2020
|
Novartis Capital Corporation, New York, United States
|
99.237%
|
997
|
996
|
|||||||||
|
2.400%
|
USD
|
1 500
|
2012
|
2022
|
Novartis Capital Corporation, New York, United States
|
99.225%
|
1 491
|
1 490
|
|||||||||
|
3.700%
|
USD
|
500
|
2012
|
2042
|
Novartis Capital Corporation, New York, United States
|
98.325%
|
489
|
489
|
|||||||||
|
3.400%
|
USD
|
2 150
|
2014
|
2024
|
Novartis Capital Corporation, New York, United States
|
99.287%
|
2 134
|
2 132
|
|||||||||
|
4.400%
|
USD
|
1 850
|
2014
|
2044
|
Novartis Capital Corporation, New York, United States
|
99.196%
|
1 824
|
1 823
|
|||||||||
|
0.750%
|
EUR
|
600
|
2014
|
2021
|
Novartis Finance S.A., Luxembourg, Luxembourg
|
99.134%
|
713
|
625
|
|||||||||
|
1.625%
|
EUR
|
600
|
2014
|
2026
|
Novartis Finance S.A., Luxembourg, Luxembourg
|
99.697%
|
714
|
627
|
|||||||||
|
0.250%
|
CHF
|
500
|
2015
|
2025
|
Novartis AG, Basel, Switzerland
|
100.640%
|
513
|
491
|
|||||||||
|
0.625%
|
CHF
|
550
|
2015
|
2029
|
Novartis AG, Basel, Switzerland
|
100.502%
|
564
|
539
|
|||||||||
|
1.050%
|
CHF
|
325
|
2015
|
2035
|
Novartis AG, Basel, Switzerland
|
100.479%
|
333
|
318
|
|||||||||
|
3.000%
|
USD
|
1 750
|
2015
|
2025
|
Novartis Capital Corporation, New York, United States
|
99.010%
|
1 730
|
1 728
|
|||||||||
|
4.000%
|
USD
|
1 250
|
2015
|
2045
|
Novartis Capital Corporation, New York, United States
|
98.029%
|
1 218
|
1 217
|
|||||||||
|
0.125%
|
EUR
|
1 250
|
2016
|
2023
|
Novartis Finance S.A., Luxembourg, Luxembourg
|
99.127%
|
1 480
|
1 299
|
|||||||||
|
0.625%
|
EUR
|
500
|
2016
|
2028
|
Novartis Finance S.A., Luxembourg, Luxembourg
|
98.480%
|
588
|
516
|
|||||||||
|
1.800%
|
USD
|
1 000
|
2017
|
2020
|
Novartis Capital Corporation, New York, United States
|
99.609%
|
996
|
|
|||||||||
|
2.400%
|
USD
|
1 000
|
2017
|
2022
|
Novartis Capital Corporation, New York, United States
|
99.449%
|
993
|
|
|||||||||
|
3.100%
|
USD
|
1 000
|
2017
|
2027
|
Novartis Capital Corporation, New York, United States
|
99.109%
|
988
|
|
|||||||||
|
0.000%
|
EUR
|
1 250
|
2017
|
2021
|
Novartis Finance S.A., Luxembourg, Luxembourg
|
99.133%
|
1 480
|
|
|||||||||
|
1.125%
|
EUR
|
600
|
2017
|
2027
|
Novartis Finance S.A., Luxembourg, Luxembourg
|
99.874%
|
715
|
|
|||||||||
|
Total straight bonds
|
|
|
|
|
|
|
22 957
|
17 285
|
|||||||||
|
(USD millions)
|
2017
|
2016
|
|||
|
2017
|
|
178
|
|||
|
2018
|
359
|
345
|
|||
|
2019
|
3 173
|
3 168
|
|||
|
2020
|
1 997
|
1 000
|
|||
|
2021
|
2 194
|
628
|
|||
|
2022
|
2 485
|
2 442
|
|||
|
After 2022
|
13 375
|
10 314
|
|||
|
Total
|
23 583
|
18 075
|
|||
|
(USD millions)
|
2017
|
2016
|
|||
|
US dollar (USD)
|
15 945
|
12 952
|
|||
|
Euro (EUR)
|
5 695
|
3 092
|
|||
|
Japanese yen (JPY)
|
533
|
683
|
|||
|
Swiss franc (CHF)
|
1 410
|
1 348
|
|||
|
Total
|
23 583
|
18 075
|
|||
|
(USD millions) |
2017
Balance sheet |
2017
Fair values |
2016
Balance sheet |
2016
Fair values |
|||||
|
Straight bonds
|
22 957
|
23 835
|
17 285
|
17 943
|
|||||
|
Others
|
626
|
626
|
790
|
790
|
|||||
|
Total
|
23 583
|
24 461
|
18 075
|
18 733
|
|||||
|
(USD millions)
|
2017
|
2016
|
|||
|
Total net book value of property, plant & equipment pledged as collateral for non-current financial debts
|
84
|
94
|
|||
|
(USD millions)
|
2017
|
2016
|
|||
|
Accrued liability for employee benefits:
|
|
|
|||
|
Defined benefit pension plans 1
|
3 157
|
4 490
|
|||
|
Other long-term employee benefits and deferred compensation
|
625
|
545
|
|||
|
Other post-employment benefits 1
|
953
|
1 005
|
|||
|
Environmental remediation provisions
|
706
|
708
|
|||
|
Provisions for product liabilities, governmental investigations and other legal matters
|
230
|
264
|
|||
|
Contingent consideration 2
|
809
|
840
|
|||
|
Other non-current liabilities
|
577
|
618
|
|||
|
Total provisions and other non-current liabilities
|
7 057
|
8 470
|
|||
|
|
|||||
|
1 Note 24 provides additional disclosures related to post-employment benefits.
|
|||||
|
2 Note 28 provides additional disclosures related to contingent consideration.
|
|||||
|
(USD millions)
|
2017
|
2016
|
2015
|
||||
|
January 1
|
773
|
871
|
923
|
||||
|
Cash payments
|
– 46
|
– 75
|
– 52
|
||||
|
Releases
|
– 153
|
|
– 5
|
||||
|
Additions
|
154
|
1
|
6
|
||||
|
Currency translation effects
|
33
|
– 24
|
– 1
|
||||
|
December 31
|
761
|
773
|
871
|
||||
|
Less current provision
|
– 55
|
– 65
|
– 80
|
||||
|
Non-current environmental remediation provisions at December 31
|
706
|
708
|
791
|
||||
|
(USD millions) |
Expected
cash outflows |
||
|
Due within two years
|
164
|
||
|
Due later than two years, but within five years
|
241
|
||
|
Due later than five years, but within ten years
|
315
|
||
|
Due after ten years
|
41
|
||
|
Total environmental remediation liability provisions
|
761
|
||
|
(USD millions)
|
2017
|
2016
|
2015
|
||||
|
January 1
|
395
|
1 194
|
849
|
||||
|
Cash payments
|
– 69
|
– 811
|
– 256
|
||||
|
Releases of provisions
|
– 70
|
– 239
|
– 223
|
||||
|
Additions to provisions
|
93
|
243
|
832
|
||||
|
Currency translation effects
|
2
|
8
|
– 8
|
||||
|
December 31
|
351
|
395
|
1 194
|
||||
|
Less current portion
|
– 121
|
– 131
|
– 743
|
||||
|
Non-current product liabilities, governmental investigations and other legal matters provisions at December 31
|
230
|
264
|
451
|
||||
|
(USD millions)
|
2017
|
2016
|
|||
|
Interest-bearing accounts of associates payable on demand 1
|
1 822
|
1 601
|
|||
|
Bank and other financial debt 2
|
692
|
836
|
|||
|
Commercial paper
|
2 328
|
3 174
|
|||
|
Current portion of non-current financial debt
|
359
|
178
|
|||
|
Fair value of derivative financial instruments
|
107
|
116
|
|||
|
Total current financial debt and derivative financial instruments
|
5 308
|
5 905
|
|||
|
1 Weighted average interest rate 0.5% (2016: 0.5%)
|
|||||
|
2 Weighted average interest rate 7.0% (2016: 6.7%)
|
|||||
|
(USD millions)
|
2017
|
2016
|
|||
|
Taxes other than income taxes
|
660
|
547
|
|||
|
Restructuring provisions
|
153
|
222
|
|||
|
Accrued expenses for goods and services received but not invoiced
|
977
|
880
|
|||
|
Accruals for royalties
|
586
|
550
|
|||
|
Provisions for deductions from revenue
|
4 672
|
4 183
|
|||
|
Accruals for compensation and benefits including social security
|
2 327
|
1 993
|
|||
|
Environmental remediation liabilities
|
55
|
65
|
|||
|
Deferred income
|
305
|
287
|
|||
|
Provisions for product liabilities, governmental investigations and other legal matters 1
|
121
|
131
|
|||
|
Accrued share-based payments
|
261
|
199
|
|||
|
Contingent considerations 2
|
44
|
49
|
|||
|
Other payables
|
1 042
|
722
|
|||
|
Total provisions and other current liabilities
|
11 203
|
9 828
|
|||
|
|
|||||
|
1 Note 19 provides additional disclosures related to legal provisions.
|
|||||
|
2 Note 28 provides additional disclosures related to contingent considerations.
|
|||||
|
(USD millions)
|
2017
|
2016
|
2015
|
||||
|
January 1
|
4 183
|
3 790
|
3 533
|
||||
|
Impact of business combinations
|
|
|
3
|
||||
|
Additions
|
17 997
|
16 622
|
15 603
|
||||
|
Payments/utilizations
|
– 17 452
|
– 16 189
|
– 15 218
|
||||
|
Changes in offset against gross trade receivables
|
– 252
|
10
|
50
|
||||
|
Currency translation effects
|
196
|
– 50
|
– 181
|
||||
|
December 31
|
4 672
|
4 183
|
3 790
|
||||
|
(USD millions)
|
2017
|
2016
|
2015
|
||||
|
January 1
|
222
|
260
|
333
|
||||
|
Additions
|
194
|
343
|
399
|
||||
|
Cash payments
|
– 200
|
– 260
|
– 435
|
||||
|
Releases
|
– 64
|
– 66
|
– 36
|
||||
|
Transfers
|
– 7
|
– 76
|
|
||||
|
Currency translation effects
|
8
|
21
|
– 1
|
||||
|
December 31
|
153
|
222
|
260
|
||||
|
(USD millions)
|
2017
|
2016
|
2015
|
||||
|
Taxes
|
1 296
|
1 119
|
1 106
|
||||
|
Depreciation, amortization and impairments on:
|
|
|
|
||||
|
Property, plant & equipment
|
1 677
|
1 591
|
1 550
|
||||
|
Intangible assets
|
4 399
|
4 452
|
3 921
|
||||
|
Financial assets 1
|
256
|
132
|
104
|
||||
|
Income from associated companies
|
– 1 108
|
– 703
|
– 266
|
||||
|
Gains on disposal and other adjustments on property, plant & equipment, intangible, financial and other non-current assets, net
|
– 1 043
|
– 935
|
– 869
|
||||
|
Equity-settled compensation expense
|
683
|
671
|
773
|
||||
|
Change in provisions and other non-current liabilities
|
160
|
956
|
1 642
|
||||
|
Net financial expense
|
738
|
1 154
|
1 109
|
||||
|
Total
|
7 058
|
8 437
|
9 070
|
||||
|
1 Including unrealized fair value gains
|
|||||||
|
(USD millions)
|
2017
|
2016
|
2015
|
||||
|
(Increase) in inventories
|
– 247
|
– 235
|
– 482
|
||||
|
(Increase) in trade receivables
|
– 204
|
– 229
|
– 513
|
||||
|
Increase/(Decrease) in trade payables
|
58
|
– 587
|
378
|
||||
|
Change in other net current assets and other operating cash flow items
|
637
|
974
|
– 246
|
||||
|
Total
|
244
|
– 77
|
– 863
|
||||
|
(USD millions) |
2017
Acquisitions |
2017
Divestments |
2016
Acquisitions |
2016
Divestments |
2015
Acquisitions |
2015
Divestments |
|||||||
|
Property, plant & equipment
|
|
25
|
|
|
|
1 000
|
|||||||
|
Currently marketed products
|
|
1
|
– 451
|
|
– 12 970
|
646
|
|||||||
|
(Acquired)/divested research & development
|
– 1 223
|
|
– 690
|
|
– 730
|
13
|
|||||||
|
Technologies
|
|
|
|
|
|
113
|
|||||||
|
Other intangible assets
|
|
3
|
|
|
– 15
|
86
|
|||||||
|
Financial and other assets including deferred tax assets
|
– 8
|
|
– 39
|
|
– 555
|
40
|
|||||||
|
Inventories
|
|
|
– 4
|
|
|
893
|
|||||||
|
Trade receivables and other current assets
|
|
34
|
– 1
|
|
– 3
|
529
|
|||||||
|
Cash and cash equivalents
|
– 20
|
|
– 1
|
|
– 25
|
311
|
|||||||
|
Current and non-current financial debts
|
|
|
|
|
|
– 601
|
|||||||
|
Trade payables and other liabilities including deferred tax liabilities
|
326
|
– 15
|
372
|
|
212
|
– 841
|
|||||||
|
Net identifiable assets (acquired) or divested
|
– 925
|
|
48
|
– 814
|
|
– 14 086
|
2 189
|
||||||
|
Currency translation effects
|
|
|
|
|
|
98
|
|||||||
|
Acquired/(divested) liquidity
|
20
|
|
1
|
|
25
|
– 479
|
|||||||
|
Fair value of previously held equity interests
|
|
|
|
|
64
|
|
|
|
|
|
|
|
|
|
Subtotal
|
– 905
|
|
48
|
– 749
|
|
– 14 061
|
1 808
|
||||||
|
Refinancing of intercompany financial debt, net
|
|
|
|
|
|
578
|
|||||||
|
Goodwill
|
– 94
|
|
– 56
|
|
– 2 438
|
1 042
|
|||||||
|
Divestment gain
|
|
|
|
|
|
7 401
|
|||||||
|
Taxes paid and other portfolio transformation related cash flows
|
|
– 140
|
|
– 748
|
|
– 1 337
|
|||||||
|
Receivables and payables contingent consideration, net 1
|
206
|
|
84
|
|
– 8
|
– 519
|
|||||||
|
Other payments and deferred consideration, net
|
– 36
|
– 3
|
– 44
|
|
|
|
|||||||
|
Deferred portion of sales price 2
|
|
|
|
|
|
– 49
|
|||||||
|
Net cash flows
|
– 829
|
|
– 95
|
– 765
|
– 748
|
– 16 507
|
8 924
|
||||||
|
Of which:
|
|
|
|
|
|
|
|||||||
|
Net cash flows used in/from discontinued operations
|
|
– 140
|
|
– 748
|
|
8 924
|
|||||||
|
Net cash flows used in/from continuing operations
|
– 829
|
45
|
– 765
|
|
– 16 507
|
|
|||||||
|
|
|||||||||||||
|
1 The contingent consideration of the 2016 Transcend Medical, Inc. acquisition amounted to USD 92 million. Of this amount, USD 60 million has been paid in 2016.
|
|||||||||||||
|
2 Divestments include USD 49 million proceeds for the divestment of the Animal Health business received in 2014.
|
|||||||||||||
|
(USD millions)
|
2017
|
2016
|
2015
|
||||
|
Cash flows used in operating activities
|
|
|
– 188
|
||||
|
Purchase of property, plant & equipment
|
|
|
– 41
|
||||
|
Proceeds from sales of property, plant & equipment
|
|
|
1
|
||||
|
Purchase of financial and other non-current assets, net
|
|
|
– 2
|
||||
|
Divestments of businesses 1
|
– 140
|
– 748
|
8 924
|
||||
|
Cash flows used in/from investing activities
|
– 140
|
– 748
|
8 882
|
||||
|
Total net cash flows used in/from discontinued operations
|
– 140
|
|
– 748
|
|
8 694
|
||
|
|
|||||||
|
1 2017 includes payments related to the 2015 portfolio transformation transaction. 2016 includes mainly payments for capital gains taxes and other payments related to the 2015 portfolio transformation transaction. 2015 includes proceeds of USD 10 925 million reduced by USD 2 001 million, for payments of capital gains taxes, transaction-related costs and purchase price adjustments, related to the 2015 portfolio transformation transaction. See Note 2 for a description of the 2015 porfolio transformation transaction.
|
|||||||
|
(USD millions) |
Non-current financial debts |
|
Current
financial debts and derivative financial instruments |
|
Total |
||
|
January 1, 2017
|
17 897
|
5 905
|
23 802
|
||||
|
Increase in non-current financial debts
|
4 933
|
|
4 933
|
||||
|
Repayment of non-current financial debts
|
– 1
|
– 187
|
– 188
|
||||
|
Change in current financial debts
|
|
– 755
|
– 755
|
||||
|
Changes in fair values and other changes
|
– 6
|
– 140
|
– 146
|
||||
|
Amortization of bonds discount
|
16
|
|
16
|
||||
|
Currency translation effects
|
744
|
126
|
870
|
||||
|
Current portion of non-current financial debt
|
– 359
|
359
|
|
||||
|
December 31, 2017
|
23 224
|
5 308
|
28 532
|
||||
|
|
|||||||
|
|
|||||||
|
(USD millions)
|
2017
|
2016
|
2015
|
||||
|
Currently marketed products
|
|
451
|
12 970
|
||||
|
Acquired research & development
|
1 223
|
690
|
730
|
||||
|
Other intangible assets
|
|
|
15
|
||||
|
Deferred tax assets
|
8
|
39
|
555
|
||||
|
Inventories
|
|
4
|
|
||||
|
Trade receivables and other current assets
|
|
1
|
3
|
||||
|
Cash and cash equivalents
|
20
|
1
|
25
|
||||
|
Payables and other liabilities including deferred tax liabilities
|
– 326
|
– 372
|
– 212
|
||||
|
Net identifiable assets acquired
|
925
|
814
|
14 086
|
||||
|
Acquired liquidity
|
– 20
|
– 1
|
– 25
|
||||
|
Goodwill
|
94
|
56
|
2 438
|
||||
|
Net assets recognized as a result of business combinations
|
999
|
869
|
16 499
|
||||
|
Pension plans
|
Other post-employment benefit plans
|
||||||||
|
(USD millions)
|
2017
|
2016
|
2017
|
2016
|
|||||
|
Benefit obligation at January 1
|
23 614
|
23 402
|
1 158
|
1 132
|
|||||
|
Current service cost
|
422
|
437
|
34
|
35
|
|||||
|
Interest cost
|
330
|
390
|
44
|
48
|
|||||
|
Past service costs and settlements
|
– 1 226
|
– 73
|
– 10
|
|
|||||
|
Administrative expenses
|
27
|
29
|
|
|
|||||
|
Remeasurement losses arising from changes in financial assumptions
|
11
|
1 299
|
32
|
46
|
|||||
|
Remeasurement (gains) arising from changes in demographic assumptions
|
– 26
|
– 7
|
– 9
|
– 26
|
|||||
|
Experience-related remeasurement losses/(gains)
|
47
|
117
|
– 87
|
– 33
|
|||||
|
Currency translation effects
|
1 138
|
– 896
|
5
|
7
|
|||||
|
Benefit payments
|
– 1 300
|
– 1 250
|
– 51
|
– 51
|
|||||
|
Contributions of associates
|
207
|
207
|
|
|
|||||
|
Effect of acquisitions, divestments or transfers
|
– 34
|
– 41
|
– 1
|
|
|||||
|
Benefit obligation at December 31
|
23 210
|
23 614
|
1 115
|
1 158
|
|||||
|
Fair value of plan assets at January 1
|
19 225
|
19 536
|
153
|
172
|
|||||
|
Interest income
|
236
|
293
|
5
|
6
|
|||||
|
Return on plan assets excluding interest income
|
1 429
|
742
|
12
|
– 1
|
|||||
|
Currency translation effects
|
909
|
– 757
|
|
|
|||||
|
Novartis Group contributions
|
579
|
542
|
43
|
27
|
|||||
|
Contributions of associates
|
207
|
207
|
|
|
|||||
|
Settlements
|
– 995
|
– 77
|
|
|
|||||
|
Benefit payments
|
– 1 300
|
– 1 250
|
– 51
|
– 51
|
|||||
|
Effect of acquisitions, divestments or transfers
|
– 15
|
– 11
|
|
|
|||||
|
Fair value of plan assets at December 31
|
20 275
|
19 225
|
162
|
153
|
|||||
|
Funded status
|
– 2 935
|
– 4 389
|
– 953
|
– 1 005
|
|||||
|
Limitation on recognition of fund surplus at January 1
|
– 54
|
– 50
|
|
|
|||||
|
Change in limitation on recognition of fund surplus (incl. exchange rate differences)
|
– 30
|
|
|
|
|||||
|
Interest income on limitation of fund surplus
|
– 5
|
– 4
|
|
|
|||||
|
Limitation on recognition of fund surplus at December 31
|
– 89
|
– 54
|
|
|
|||||
|
Net liability in the balance sheet at December 31
|
– 3 024
|
– 4 443
|
– 953
|
– 1 005
|
|||||
|
Pension plans
|
Other post-employment benefit plans
|
||||||||
|
(USD millions)
|
2017
|
2016
|
2017
|
2016
|
|||||
|
Net liability at January 1
|
– 4 443
|
– 3 916
|
– 1 005
|
– 960
|
|||||
|
Current service cost
|
– 422
|
– 437
|
– 34
|
– 35
|
|||||
|
Net interest expense
|
– 99
|
– 101
|
– 39
|
– 42
|
|||||
|
Administrative expenses
|
– 27
|
– 29
|
|
|
|||||
|
Past service costs and settlements
|
231
|
– 4
|
10
|
|
|||||
|
Remeasurements
|
1 397
|
– 667
|
76
|
12
|
|||||
|
Currency translation effects
|
– 229
|
139
|
– 5
|
– 7
|
|||||
|
Novartis Group contributions
|
579
|
542
|
43
|
27
|
|||||
|
Effect of acquisitions, divestments or transfers
|
19
|
30
|
1
|
|
|||||
|
Change in limitation on recognition of fund surplus
|
– 30
|
|
|
|
|||||
|
Net liability at December 31
|
– 3 024
|
– 4 443
|
– 953
|
– 1 005
|
|||||
|
|
|
|
|
||||||
|
Amounts recognized in the consolidated balance sheet
|
|
||||||||
|
Prepaid benefit cost
|
133
|
47
|
|
|
|||||
|
Accrued benefit liability
|
– 3 157
|
– 4 490
|
– 953
|
– 1 005
|
|||||
|
2017
|
2016
|
||||||||||||||||
|
(USD millions) |
Switzerland |
United States |
Rest of
the world |
Total |
Switzerland |
United States |
Rest of
the world |
Total |
|||||||||
|
Benefit obligation at December 31
|
14 606
|
3 788
|
4 816
|
23 210
|
15 436
|
3 783
|
4 395
|
23 614
|
|||||||||
|
Thereof unfunded
|
|
728
|
499
|
1 227
|
|
739
|
497
|
1 236
|
|||||||||
|
By type of member
|
|
|
|
|
|
|
|
|
|||||||||
|
Active
|
5 627
|
796
|
1 646
|
8 069
|
6 426
|
891
|
1 460
|
8 777
|
|||||||||
|
Deferred pensioners
|
|
1 258
|
1 646
|
2 904
|
|
831
|
1 515
|
2 346
|
|||||||||
|
Pensioners
|
8 979
|
1 734
|
1 524
|
12 237
|
9 010
|
2 061
|
1 420
|
12 491
|
|||||||||
|
Fair value of plan assets at December 31
|
14 445
|
2 400
|
3 430
|
20 275
|
13 958
|
2 282
|
2 985
|
19 225
|
|||||||||
|
Funded status
|
– 161
|
– 1 388
|
– 1 386
|
– 2 935
|
– 1 478
|
– 1 501
|
– 1 410
|
– 4 389
|
|||||||||
|
Pension plans
|
Other post-employment benefit plans
|
||||||||||||
|
2017
|
2016
|
2015
|
2017
|
2016
|
2015
|
||||||||
|
Weighted average assumptions used to determine benefit obligations at December 31
|
|
|
|
|
|
|
|||||||
|
Discount rate
|
1.5%
|
1.4%
|
1.8%
|
3.7%
|
4.2%
|
4.4%
|
|||||||
|
Expected rate of pension increase
|
0.5%
|
0.4%
|
0.4%
|
|
|
|
|||||||
|
Expected rate of salary increase
|
2.8%
|
2.2%
|
2.9%
|
|
|
|
|||||||
|
Interest on savings account
|
0.6%
|
0.5%
|
0.8%
|
|
|
|
|||||||
|
Current average life expectancy for a 65-year-old male in years
|
22
|
22
|
21
|
21
|
21
|
21
|
|||||||
|
Current average life expectancy for a 65-year-old female in years
|
24
|
24
|
24
|
23
|
23
|
23
|
|||||||
|
(USD millions) |
Change in 2017 year-end
defined benefit pension obligation |
||
|
25 basis point increase in discount rate
|
– 753
|
||
|
25 basis point decrease in discount rate
|
803
|
||
|
1 year increase in life expectancy
|
840
|
||
|
25 basis point increase in rate of pension increase
|
533
|
||
|
25 basis point decrease in rate of pension increase
|
– 138
|
||
|
25 basis point increase of interest on savings account
|
56
|
||
|
25 basis point decrease of interest on savings account
|
– 54
|
||
|
25 basis point increase in rate of salary increase
|
49
|
||
|
25 basis point decrease in rate of salary increase
|
– 50
|
||
|
2017
|
2016
|
2015
|
|||||
|
Healthcare cost trend rate assumed for next year
|
6.5%
|
7.0%
|
7.5%
|
||||
|
Rate to which the cost trend rate is assumed to decline
|
4.5%
|
5.0%
|
5.0%
|
||||
|
Year that the rate reaches the ultimate trend rate
|
2025
|
2022
|
2022
|
||||
|
Pension plans
|
|||||||||
|
(as a percentage) |
Long-term
target minimum |
Long-term
target maximum |
2017 |
2016 |
|||||
|
Equity securities
|
15
|
40
|
31
|
31
|
|||||
|
Debt securities
|
20
|
60
|
35
|
35
|
|||||
|
Real estate
|
5
|
20
|
15
|
15
|
|||||
|
Alternative investments
|
0
|
20
|
15
|
15
|
|||||
|
Cash and other investments
|
0
|
15
|
4
|
4
|
|||||
|
Total
|
|
|
100
|
100
|
|||||
|
December 31,
2017 |
December 31,
2016 |
||||
|
Investment in shares of Novartis AG
|
|
|
|||
|
Number of shares (in millions)
|
11.0
|
11.0
|
|||
|
Market Value (in USD billions)
|
0.9
|
0.8
|
|||
|
(USD millions) |
Pension plans |
Other post-
employment benefit plans |
|||
|
Novartis Group contributions
|
|
|
|||
|
2018 (estimated)
|
395
|
62
|
|||
|
Expected future benefit payments
|
|
|
|||
|
2018
|
1 226
|
63
|
|||
|
2019
|
1 166
|
65
|
|||
|
2020
|
1 163
|
67
|
|||
|
2021
|
1 147
|
68
|
|||
|
2022
|
1 133
|
69
|
|||
|
2023–2027
|
5 534
|
344
|
|||
|
(USD millions)
|
2017
|
2016
|
2015
|
||||
|
Contributions for defined contribution plans continuing operations
|
406
|
338
|
359
|
||||
|
Contributions for defined contribution plans discontinued operations
|
|
|
1
|
||||
|
(USD millions)
|
2017
|
2016
|
2015
|
||||
|
Expense related to equity-based participation plans
|
924
|
846
|
968
|
||||
|
of which continuing operations
|
924
|
846
|
903
|
||||
|
of which discontinued operations
|
|
|
65
|
||||
|
Liabilities arising from equity-based payment transactions
|
261
|
199
|
209
|
||||
|
2017
|
2016
|
||||||||
|
Options (millions) |
Weighted
average exercise price (USD) |
Options (millions) |
Weighted
average exercise price (USD) |
||||||
|
Options outstanding at January 1
|
9.5
|
59.4
|
11.7
|
59.9
|
|||||
|
Sold or exercised
|
– 2.1
|
59.2
|
– 2.2
|
61.8
|
|||||
|
Forfeited or expired
|
|
|
|
|
|||||
|
Outstanding at December 31
|
7.4
|
59.5
|
9.5
|
59.4
|
|||||
|
Exercisable at December 31
|
7.4
|
59.5
|
9.5
|
59.4
|
|||||
|
Options outstanding
|
|||||||
|
Range of exercise prices (USD) |
Number outstanding (millions) |
Average
remaining contractual life (years) |
Weighted
average exercise price (USD) |
||||
|
45–49
|
0.7
|
1.0
|
46.7
|
||||
|
50–54
|
1.1
|
2.0
|
54.6
|
||||
|
55–59
|
2.7
|
3.3
|
57.6
|
||||
|
65–70
|
2.9
|
5.0
|
66.1
|
||||
|
Total
|
7.4
|
3.6
|
59.5
|
||||
|
2017
|
2016
|
||||||||
|
ADR options (millions) |
Weighted
average exercise price (USD) |
ADR options (millions) |
Weighted
average exercise price (USD) |
||||||
|
Options outstanding at January 1
|
25.9
|
59.9
|
31.9
|
60.2
|
|||||
|
Sold or exercised
|
– 5.6
|
59.9
|
– 6.0
|
61.7
|
|||||
|
Forfeited or expired
|
|
|
|
|
|||||
|
Outstanding at December 31
|
20.3
|
59.9
|
25.9
|
59.9
|
|||||
|
Exercisable at December 31
|
20.3
|
59.9
|
25.9
|
59.9
|
|||||
|
ADR options outstanding
|
|||||||
|
Range of exercise prices (USD) |
Number outstanding (millions) |
Average
remaining contractual life (years) |
Weighted
average exercise price (USD) |
||||
|
45–49
|
1.8
|
1.0
|
46.4
|
||||
|
50–54
|
2.1
|
2.0
|
53.7
|
||||
|
55–59
|
8.0
|
3.5
|
58.0
|
||||
|
65–69
|
8.4
|
5.0
|
66.1
|
||||
|
Total
|
20.3
|
3.7
|
59.9
|
||||
|
2017
|
2016
|
||||||||||||
|
Number of shares in millions |
Weighted
average fair value at grant date in USD |
Fair value at grant date in USD millions |
Number of shares in millions |
Weighted
average fair value at grant date in USD |
Fair value at grant date in USD millions |
||||||||
|
Non-vested shares at January 1
|
21.0
|
89.5
|
1 880
|
20.1
|
87.1
|
1 751
|
|||||||
|
Granted
|
|
|
|
|
|
|
|||||||
|
– Annual incentive
|
1.3
|
69.3
|
90
|
0.1
|
73.8
|
7
|
|||||||
|
– Share savings plans
|
4.5
|
69.4
|
312
|
4.4
|
78.1
|
344
|
|||||||
|
– Select North America
|
4.5
|
64.1
|
288
|
4.8
|
72.4
|
348
|
|||||||
|
– Select outside North America
|
2.0
|
65.3
|
131
|
1.6
|
74.4
|
119
|
|||||||
|
– Long-Term Performance Plan
|
1.4
|
71.5
|
100
|
1.2
|
79.2
|
95
|
|||||||
|
– Long-Term Relative Performance Plan
|
0.4
|
47.7
|
19
|
0.3
|
58.5
|
18
|
|||||||
|
– Other share awards
|
1.3
|
67.8
|
88
|
0.7
|
65.8
|
46
|
|||||||
|
Vested
|
– 10.7
|
78.2
|
– 837
|
– 10.4
|
68.8
|
– 716
|
|||||||
|
Forfeited
|
– 1.8
|
80.7
|
– 145
|
– 1.8
|
73.1
|
– 132
|
|||||||
|
Non-vested shares at December 31
|
23.9
|
80.6
|
1 926
|
21.0
|
89.5
|
1 880
|
|||||||
|
Number of options (millions) |
Weighted
average exercise price (USD) |
Number of SSARs (millions) |
Weighted
average exercise price (USD) |
||||||
|
Outstanding at January 1, 2016
|
0.2
|
36.8
|
1.8
|
36.6
|
|||||
|
Exercised
|
– 0.1
|
37.6
|
– 0.4
|
38.9
|
|||||
|
Outstanding at December 31, 2016
|
0.1
|
36.0
|
1.4
|
35.9
|
|||||
|
Exercisable at December 31, 2016
|
0.1
|
36.0
|
1.4
|
35.9
|
|||||
|
Outstanding at January 1, 2017
|
0.1
|
36.0
|
1.4
|
35.9
|
|||||
|
Exercised
|
|
|
– 0.6
|
39.8
|
|||||
|
Outstanding at December 31, 2017
|
0.1
|
33.7
|
0.8
|
33.0
|
|||||
|
Exercisable at December 31, 2017
|
0.1
|
33.7
|
0.8
|
33.0
|
|||||
|
Executive Officers
|
Non-Executive Directors
|
Total
|
|||||||||||||||||
|
(USD millions)
|
2017
|
2016
|
2015
|
2017
|
2016
|
2015
|
2017
|
2016
|
2015
|
||||||||||
|
Cash and other compensation
|
18.4
|
20.8
|
17.1
|
4.0
|
4.0
|
4.7
|
22.4
|
24.8
|
21.8
|
||||||||||
|
Post-employment benefits
|
2.0
|
2.2
|
1.9
|
|
|
|
2.0
|
2.2
|
1.9
|
||||||||||
|
Equity-based compensation
|
49.9
|
46.2
|
52.9
|
4.8
|
4.6
|
4.4
|
54.7
|
50.8
|
57.3
|
||||||||||
|
Total
|
70.3
|
69.2
|
71.9
|
8.8
|
8.6
|
9.1
|
79.1
|
77.8
|
81.0
|
||||||||||
|
Currency
|
2017
|
2016
|
2015
|
||||||
|
Prof. Dr. Brody
|
CHF
|
0
|
25 000
|
100 000
|
|||||
|
Prof. Dr. Zinkernagel
|
CHF
|
0
|
50 000
|
200 000
|
|||||
|
Dr. Krauer
|
CHF
|
60 000
|
60 000
|
60 000
|
|||||
|
Dr. Vasella
|
CHF
|
26 279
|
0
|
0
|
|||||
|
USD
|
0
|
250 000
|
250 000
|
||||||
|
(USD millions)
|
2017
|
||
|
2018
|
309
|
||
|
2019
|
224
|
||
|
2020
|
161
|
||
|
2021
|
131
|
||
|
2022
|
123
|
||
|
Thereafter
|
2 221
|
||
|
Total
|
3 169
|
||
|
Expense of current year
|
337
|
||
|
(USD millions) |
Research & Development commitments |
Other
intangible asset purchase commitments |
Total |
||||
|
2018
|
780
|
130
|
910
|
||||
|
2019
|
671
|
|
671
|
||||
|
2020
|
864
|
|
864
|
||||
|
2021
|
801
|
|
801
|
||||
|
2022
|
353
|
|
353
|
||||
|
Thereafter
|
837
|
|
837
|
||||
|
Total
|
4 306
|
130
|
4 436
|
||||
|
(USD millions)
|
Note
|
2017 1
|
2016 1
|
||||
|
Cash and cash equivalents
|
15
|
8 860
|
7 007
|
||||
|
Financial assets - measured at fair value through other comprehensive income
|
|
|
|
||||
|
Available-for-sale marketable securities
|
|
|
|
||||
|
Debt securities
|
15
|
328
|
306
|
||||
|
Fund investments
|
15
|
34
|
31
|
||||
|
Total available-for-sale marketable securities
|
|
362
|
337
|
||||
|
Available-for-sale long-term financial investments
|
|
|
|
||||
|
Equity securities
|
12
|
1 109
|
989
|
||||
|
Fund investments
|
12
|
166
|
107
|
||||
|
Total available-for-sale long-term financial investments
|
|
1 275
|
1 096
|
||||
|
|
|
|
|||||
|
Total financial assets - measured at fair value through other comprehensive income
|
|
1 637
|
1 433
|
||||
|
|
|
|
|||||
|
Financial assets - measured at amortized costs
|
|
|
|
||||
|
Trade receivables, income tax receivables, and other current assets (excluding contingent consideration receivables and pre-payments)
|
14/16
|
10 650
|
10 202
|
||||
|
Accrued interest on debt securities and time deposits
|
15
|
1
|
1
|
||||
|
Time deposits with original maturity more than 90 days
|
15
|
125
|
108
|
||||
|
Long-term loans and receivables from customers and finance lease, advances, security deposits
|
12
|
574
|
514
|
||||
|
Total financial assets - measured at amortized costs
|
|
11 350
|
10 825
|
||||
|
|
|
|
|||||
|
Financial assets - measured at fair value through the consolidated income statement
|
|
|
|
||||
|
Associated companies at fair value through profit and loss
|
|
216
|
188
|
||||
|
Derivative financial instruments
|
15
|
31
|
230
|
||||
|
Contingent consideration receivables
|
12/16
|
844
|
586
|
||||
|
Total financial assets - measured at fair value through the consolidated income statement
|
|
1 091
|
1 004
|
||||
|
|
|
|
|||||
|
Total financial assets
|
|
22 938
|
20 269
|
||||
|
|
|
||||||
|
Financial liabilities - measured at amortized costs
|
|
|
|
||||
|
Current financial debt
|
|
|
|
||||
|
Interest-bearing accounts of associates payable on demand
|
20
|
1 822
|
1 601
|
||||
|
Bank and other financial debt
|
20
|
692
|
836
|
||||
|
Commercial paper
|
20
|
2 328
|
3 174
|
||||
|
Current portion of non-current debt
|
20
|
359
|
178
|
||||
|
Total current financial debt
|
|
5 201
|
5 789
|
||||
|
Non-current financial debt
|
|
|
|
||||
|
Straight bonds
|
18
|
22 957
|
17 285
|
||||
|
Liabilities to banks and other financial institutions
|
18
|
539
|
708
|
||||
|
Finance lease obligations
|
18
|
87
|
82
|
||||
|
Current portion of non-current debt
|
18
|
– 359
|
– 178
|
||||
|
Total non-current financial debt
|
|
23 224
|
17 897
|
||||
|
|
|
|
|||||
|
Trade payables
|
|
5 169
|
4 873
|
||||
|
|
|
|
|||||
|
Total financial liabilities - measured at amortized costs
|
|
33 594
|
28 559
|
||||
|
|
|
||||||
|
Financial liabilities - measured at fair value through the consolidated income statement
|
|
|
|
||||
|
Contingent consideration (see Note 19/21) and other financial liabilities
|
|
924
|
1 018
|
||||
|
Derivative financial instruments
|
20
|
107
|
116
|
||||
|
Total financial liabilities - measured at fair value through the consolidated income statement
|
|
1 031
|
1 134
|
||||
|
|
|
|
|||||
|
Total financial liabilities
|
|
34 625
|
29 693
|
||||
|
|
|||||||
|
1 Except for straight bonds (see Note 18), the carrying amount is a reasonable approximation of fair value.
|
|||||||
|
Contract or underlying principal amount
|
Positive fair values
|
Negative fair values
|
|||||||||||
|
(USD millions)
|
2017
|
2016
|
2017
|
2016
|
2017
|
2016
|
|||||||
|
Currency-related instruments
|
|
|
|
|
|
|
|||||||
|
Forward foreign exchange rate contracts
|
8 410
|
8 220
|
31
|
230
|
– 107
|
– 116
|
|||||||
|
Total derivative financial instruments included in marketable securities and in current financial debts
|
8 410
|
8 220
|
31
|
230
|
– 107
|
– 116
|
|||||||
|
2017
|
|||||||||||||||||
|
(USD millions)
|
EUR
|
USD
|
Other
|
Total
|
|||||||||||||
|
Currency-related instruments
|
|
|
|
|
|||||||||||||
|
Forward foreign exchange rate contracts
|
2 768
|
4 361
|
1 281
|
8 410
|
|||||||||||||
|
Total derivative financial instruments
|
2 768
|
4 361
|
1 281
|
8 410
|
|||||||||||||
|
2016
|
|||||||||||
|
(USD millions)
|
EUR
|
USD
|
JPY
|
Other
|
Total
|
||||||
|
Currency-related instruments
|
|
|
|
|
|
||||||
|
Forward foreign exchange rate contracts
|
3 623
|
3 427
|
43
|
1 127
|
8 220
|
||||||
|
Total derivative financial instruments
|
3 623
|
3 427
|
43
|
1 127
|
8 220
|
||||||
|
2017
|
|||||||||||
|
(USD millions) |
Level 1 |
Level 2 |
Level 3 |
Valued at
amortized cost |
Total |
||||||
|
Financial assets
|
|
|
|
|
|
||||||
|
Debt securities
|
303
|
25
|
|
|
328
|
||||||
|
Fund investments
|
34
|
|
|
|
34
|
||||||
|
Total available-for-sale marketable securities
|
337
|
25
|
|
|
362
|
||||||
|
Time deposits with original maturity more than 90 days
|
|
|
|
125
|
125
|
||||||
|
Derivative financial instruments
|
|
31
|
|
|
31
|
||||||
|
Accrued interest on debt securities
|
|
|
|
1
|
1
|
||||||
|
Total marketable securities, time deposits and derivative financial instruments
|
337
|
56
|
|
126
|
519
|
||||||
|
Available-for-sale financial investments
|
672
|
|
437
|
|
1 109
|
||||||
|
Fund investments
|
|
|
166
|
|
166
|
||||||
|
Contingent consideration receivables
|
|
|
394
|
|
394
|
||||||
|
Long-term loans and receivables from customers and finance lease, advances, security deposits
|
|
|
|
574
|
574
|
||||||
|
Financial investments and long-term loans
|
672
|
|
997
|
574
|
2 243
|
||||||
|
Associated companies at fair value through profit and loss
|
28
|
|
188
|
|
216
|
||||||
|
Contingent consideration receivables short-term
|
|
|
450
|
|
450
|
||||||
|
|
|
|
|
|
|||||||
|
Financial liabilities
|
|
|
|
|
|
||||||
|
Contingent consideration payables
|
|
|
– 852
|
|
– 852
|
||||||
|
Other financial liabilities
|
|
|
– 72
|
|
– 72
|
||||||
|
Derivative financial instruments
|
|
– 107
|
|
|
– 107
|
||||||
|
Total financial liabilities at fair value
|
|
– 107
|
– 924
|
|
– 1 031
|
||||||
|
2016
|
|||||||||||
|
(USD millions) |
Level 1 |
Level 2 |
Level 3 |
Valued at
amortized cost |
Total |
||||||
|
Financial assets
|
|
|
|
|
|
||||||
|
Debt securities
|
284
|
22
|
|
|
306
|
||||||
|
Fund investments
|
31
|
|
|
|
31
|
||||||
|
Total available-for-sale marketable securities
|
315
|
22
|
|
|
337
|
||||||
|
Time deposits with original maturity more than 90 days
|
|
|
|
108
|
108
|
||||||
|
Derivative financial instruments
|
|
230
|
|
|
230
|
||||||
|
Accrued interest on debt securities
|
|
|
|
1
|
1
|
||||||
|
Total marketable securities, time deposits and derivative financial instruments
|
315
|
252
|
|
109
|
676
|
||||||
|
Available-for-sale financial investments
|
513
|
|
476
|
|
989
|
||||||
|
Fund investments
|
|
|
107
|
|
107
|
||||||
|
Contingent consideration receivables
|
|
|
586
|
|
586
|
||||||
|
Long-term loans and receivables from customers and finance lease, advances, security deposits
|
|
|
|
514
|
514
|
||||||
|
Financial investments and long-term loans
|
513
|
|
1 169
|
514
|
2 196
|
||||||
|
Associated companies at fair value through profit and loss
|
|
|
188
|
|
188
|
||||||
|
|
|
|
|
|
|||||||
|
Financial liabilities
|
|
|
|
|
|
||||||
|
Contingent consideration payables
|
|
|
– 889
|
|
– 889
|
||||||
|
Other financial liabilities
|
|
|
– 129
|
|
– 129
|
||||||
|
Derivative financial instruments
|
|
– 116
|
|
|
– 116
|
||||||
|
Total financial liabilities at fair value
|
|
– 116
|
– 1 018
|
|
– 1 134
|
||||||
|
2017
|
||||||||||||||
|
(USD millions) |
Associated
companies at fair value through profit and loss |
Fund investments |
Available-
for-sale financial investments |
Contingent consideration receivables |
Contingent consideration payables |
Other financial liabilities |
||||||||
|
January 1
|
188
|
107
|
476
|
586
|
– 889
|
– 129
|
||||||||
|
Fair value gains and other adjustments, including from divestments recognized in the consolidated income statement
|
45
|
|
32
|
278
|
362
|
|
||||||||
|
Fair value losses (including impairments and amortizations) and other adjustments recognized in the consolidated income statement
|
– 34
|
|
– 45
|
|
– 193
|
– 37
|
||||||||
|
Fair value adjustments recognized in the consolidated statement of comprehensive income
|
|
45
|
– 40
|
|
|
|
||||||||
|
Purchases
|
37
|
28
|
113
|
|
– 238
|
|
||||||||
|
Cash receipts and payments
|
|
|
|
– 20
|
106
|
94
|
||||||||
|
Disposals
|
– 19
|
– 18
|
– 52
|
|
|
|
||||||||
|
Reclassification
|
– 29
|
4
|
– 47
|
|
|
|
||||||||
|
December 31
|
188
|
166
|
437
|
844
|
– 852
|
– 72
|
||||||||
|
|
|
|
|
|
|
|||||||||
|
Total of fair value gains and losses recognized in the consolidated income statement for assets and liabilities held at December 31, 2017
|
11
|
0
|
– 13
|
278
|
169
|
– 37
|
||||||||
|
2016
|
||||||||||||||
|
(USD millions) |
Associated
companies at fair value through profit and loss |
Fund investments |
Available-
for-sale financial investments |
Contingent consideration receivables |
Contingent consideration payables |
Other financial liabilities |
||||||||
|
January 1
|
181
|
94
|
473
|
550
|
– 790
|
– 315
|
||||||||
|
Fair value gains and other adjustments, including from divestments recognized in the consolidated income statement
|
26
|
|
1
|
51
|
|
3
|
||||||||
|
Fair value losses (including impairments and amortizations) and other adjustments recognized in the consolidated income statement
|
– 28
|
– 1
|
– 24
|
|
– 156
|
|
||||||||
|
Fair value adjustments recognized in the consolidated statement of comprehensive income
|
|
14
|
– 8
|
|
|
|
||||||||
|
Purchases
|
41
|
5
|
122
|
|
– 172
|
|
||||||||
|
Cash receipts and payments
|
|
|
|
– 15
|
229
|
183
|
||||||||
|
Disposals
|
– 3
|
– 5
|
– 18
|
|
|
|
||||||||
|
Reclassification
|
– 29
|
|
– 70
|
|
|
|
||||||||
|
December 31
|
188
|
107
|
476
|
586
|
– 889
|
– 129
|
||||||||
|
|
|
|
|
|
|
|||||||||
|
Total of fair value gains and losses recognized in the consolidated income statement for assets and liabilities held at December 31, 2016
|
– 2
|
– 1
|
– 23
|
51
|
– 156
|
3
|
||||||||
|
2017
|
|||||||||||||
|
(USD millions) |
Due within one month |
Due later than
one month but less than three months |
Due later than
three months but less than one year |
Due later than
one year but less than five years |
Due after five years |
Total |
|||||||
|
Current assets
|
|
|
|
|
|
|
|||||||
|
Marketable securities and time deposits
|
71
|
72
|
105
|
181
|
58
|
487
|
|||||||
|
Commodities
|
|
|
|
|
106
|
106
|
|||||||
|
Derivative financial instruments and accrued interest
|
7
|
19
|
6
|
|
|
32
|
|||||||
|
Cash and cash equivalents
|
4 260
|
4 600
|
|
|
|
8 860
|
|||||||
|
Total current financial assets
|
4 338
|
4 691
|
111
|
181
|
164
|
9 485
|
|||||||
|
|
|
|
|
|
|
||||||||
|
Non-current liabilities
|
|
|
|
|
|
|
|||||||
|
Financial debt
|
|
|
|
– 9 849
|
– 13 375
|
– 23 224
|
|||||||
|
Financial debt - undiscounted
|
|
|
|
– 9 893
|
– 13 519
|
– 23 412
|
|||||||
|
Total non-current financial debt
|
|
|
|
– 9 849
|
– 13 375
|
– 23 224
|
|||||||
|
|
|
|
|
|
|
||||||||
|
Current liabilities
|
|
|
|
|
|
|
|||||||
|
Financial debt
|
– 4 576
|
– 169
|
– 456
|
|
|
– 5 201
|
|||||||
|
Financial debt - undiscounted
|
– 4 576
|
– 169
|
– 456
|
|
|
– 5 201
|
|||||||
|
Derivative financial instruments
|
– 31
|
– 48
|
– 28
|
|
|
– 107
|
|||||||
|
Total current financial debt
|
– 4 607
|
– 217
|
– 484
|
|
|
– 5 308
|
|||||||
|
|
|
|
|
|
|
||||||||
|
Net debt
|
– 269
|
4 474
|
– 373
|
– 9 668
|
– 13 211
|
– 19 047
|
|||||||
|
2016
|
|||||||||||||
|
(USD millions) |
Due within one month |
Due later than
one month but less than three months |
Due later than
three months but less than one year |
Due later than
one year but less than five years |
Due after five years |
Total |
|||||||
|
Current assets
|
|
|
|
|
|
|
|||||||
|
Marketable securities and time deposits
|
32
|
126
|
110
|
124
|
53
|
445
|
|||||||
|
Commodities
|
|
|
|
|
94
|
94
|
|||||||
|
Derivative financial instruments and accrued interest
|
38
|
102
|
91
|
|
|
231
|
|||||||
|
Cash and cash equivalents
|
5 907
|
1 100
|
|
|
|
7 007
|
|||||||
|
Total current financial assets
|
5 977
|
1 328
|
201
|
124
|
147
|
7 777
|
|||||||
|
|
|
|
|
|
|
||||||||
|
Non-current liabilities
|
|
|
|
|
|
|
|||||||
|
Financial debt
|
|
|
|
– 5 141
|
– 12 756
|
– 17 897
|
|||||||
|
Financial debt - undiscounted
|
|
|
|
– 5 155
|
– 12 901
|
– 18 056
|
|||||||
|
Total non-current financial debt
|
|
|
|
– 5 141
|
– 12 756
|
– 17 897
|
|||||||
|
|
|
|
|
|
|
||||||||
|
Current liabilities
|
|
|
|
|
|
|
|||||||
|
Financial debt
|
– 5 099
|
– 250
|
– 440
|
|
|
– 5 789
|
|||||||
|
Financial debt - undiscounted
|
– 5 099
|
– 250
|
– 440
|
|
|
– 5 789
|
|||||||
|
Derivative financial instruments
|
– 15
|
– 72
|
– 29
|
|
|
– 116
|
|||||||
|
Total current financial debt
|
– 5 114
|
– 322
|
– 469
|
|
|
– 5 905
|
|||||||
|
|
|
|
|
|
|
||||||||
|
Net debt
|
863
|
1 006
|
– 268
|
– 5 017
|
– 12 609
|
– 16 025
|
|||||||
|
2017
|
|||||||||
|
(USD millions) |
Due within one month |
Due later than
one month but less than three months |
Due later than
three months but less than one year |
|
Total |
||||
|
Derivative financial instruments and accrued interest on derivative financial instruments
|
|
|
|
||||||
|
Potential outflows in various currencies - from financial derivative liabilities
|
– 953
|
– 972
|
– 2 824
|
– 4 749
|
|||||
|
Potential inflows in various currencies - from financial derivative assets
|
928
|
948
|
2 778
|
4 654
|
|||||
|
2016
|
|||||||||
|
(USD millions) |
Due within one month |
Due later than
one month but less than three months |
Due later than
three months but less than one year |
|
Total |
||||
|
Derivative financial instruments and accrued interest on derivative financial instruments
|
|
|
|
|
|||||
|
Potential outflows in various currencies - from financial derivative liabilities
|
– 1 087
|
– 1 246
|
– 2 027
|
– 4 360
|
|||||
|
Potential inflows in various currencies - from financial derivative assets
|
1 109
|
1 287
|
2 051
|
4 447
|
|||||
|
2017
|
|||||||||||
|
(USD millions) |
Due later than
one month but less than three months |
Due later than
three months but less than one year |
Due later than
one year but less than five years |
Due after five years |
Total |
||||||
|
Contractual interest on non-current liabilities
|
– 113
|
– 507
|
– 1 765
|
– 3 859
|
– 6 244
|
||||||
|
Trade payables
|
– 5 169
|
|
|
|
– 5 169
|
||||||
|
2016
|
|||||||||||
|
(USD millions) |
Due later than
one month but less than three months |
Due later than
three months but less than one year |
Due later than
one year but less than five years |
Due after five years |
Total |
||||||
|
Contractual interest on non-current liabilities
|
– 104
|
– 433
|
– 1 694
|
– 4 015
|
– 6 246
|
||||||
|
Trade payables
|
– 4 873
|
|
|
|
– 4 873
|
||||||
|
(USD millions)
|
2017
|
2016
|
|||
|
All financial instruments
|
498
|
541
|
|||
|
Analyzed by components:
|
|
|
|||
|
Instruments sensitive to foreign currency exchange rates
|
184
|
222
|
|||
|
Instruments sensitive to equity market movements
|
27
|
26
|
|||
|
Instruments sensitive to interest rates
|
242
|
328
|
|||
|
2017
|
|||||||
|
(USD millions)
|
Average
|
High
|
Low
|
||||
|
All financial instruments
|
521
|
560
|
466
|
||||
|
Analyzed by components:
|
|
|
|
||||
|
Instruments sensitive to foreign currency exchange rates
|
277
|
352
|
184
|
||||
|
Instruments sensitive to equity market movements
|
28
|
35
|
21
|
||||
|
Instruments sensitive to interest rates
|
282
|
338
|
219
|
||||
|
2016
|
|||||||
|
(USD millions)
|
Average
|
High
|
Low
|
||||
|
All financial instruments
|
402
|
541
|
316
|
||||
|
Analyzed by components:
|
|
|
|
||||
|
Instruments sensitive to foreign currency exchange rates
|
203
|
245
|
147
|
||||
|
Instruments sensitive to equity market movements
|
50
|
99
|
26
|
||||
|
Instruments sensitive to interest rates
|
308
|
407
|
234
|
||||
|
(USD millions)
|
2017
|
2016
|
|||
|
All financial instruments
|
7
|
6
|
|||
|
Analyzed by components:
|
|
|
|||
|
Instruments sensitive to foreign currency exchange rates
|
|
|
|||
|
Instruments sensitive to equity market movements
|
|
|
|||
|
Instruments sensitive to interest rates
|
7
|
6
|
|||
|
2015
|
|||||||||||
|
(USD millions) |
Vaccines |
Consumer Health1 |
Corporate
(including eliminations) |
Total
discontinued operations |
|||||||
|
Net sales to third parties of discontinued operations
|
145
|
456
|
|
601
|
|||||||
|
Sales to continuing segments
|
18
|
1
|
|
19
|
|||||||
|
Net sales of discontinued operations
|
163
|
457
|
|
620
|
|||||||
|
Other revenues
|
18
|
5
|
|
23
|
|||||||
|
Cost of goods sold
|
– 192
|
– 184
|
|
– 376
|
|||||||
|
Gross profit of discontinued operations
|
– 11
|
278
|
|
267
|
|||||||
|
Marketing & Sales
|
– 57
|
– 187
|
|
– 244
|
|||||||
|
Research & Development
|
– 151
|
– 30
|
|
– 181
|
|||||||
|
General & Administration
|
– 26
|
– 32
|
|
– 58
|
|||||||
|
Other income
|
2 870
|
10 558
|
– 8
|
13 420
|
|||||||
|
Other expense
|
– 57
|
– 14
|
– 656
|
– 727
|
|||||||
|
Operating income of discontinued operations
|
2 568
|
10 573
|
– 664
|
12 477
|
|||||||
|
Income from associated companies
|
2
|
|
|
2
|
|||||||
|
Income before taxes of discontinued operations
|
|
|
|
12 479
|
|||||||
|
Taxes
|
|
|
|
– 1 713
|
|||||||
|
Net income of discontinued operations
|
|
|
|
10 766
|
|||||||
|
1 Consumer Health is the aggregation of the former OTC and Animal Health divisions.
|
|||||||||||
|
(USD millions)
|
2015
|
||
|
Impairment charges on property, plant & equipment, net
|
83
|
||
|
Additions to restructuring provisions
|
– 1
|
||
|
Equity-based compensation of Novartis equity plans
|
– 65
|
||
|
As at December 31, 2017 |
Share
capital 1 |
Equity
interest |
|||||||
|
Algeria
|
|||||||||
|
Société par actions SANDOZ
|
Algiers
|
DZD
|
650.0
|
m
|
100%
|
||||
|
Argentina
|
|||||||||
|
Novartis Argentina S.A.
|
Buenos Aires
|
ARS
|
906.1
|
m
|
100%
|
||||
|
Alcon Laboratorios S.A.
|
Buenos Aires
|
ARS
|
83.9
|
m
|
100%
|
||||
|
Australia
|
|||||||||
|
Novartis Australia Pty Ltd
|
North Ryde, NSW
|
AUD
|
2
|
|
100%
|
||||
|
Novartis Pharmaceuticals Australia Pty Ltd
|
North Ryde, NSW
|
AUD
|
3.8
|
m
|
100%
|
||||
|
Sandoz Pty Ltd
|
North Ryde, NSW
|
AUD
|
11.6
|
m
|
100%
|
||||
|
Alcon Laboratories (Australia) Pty Ltd
|
Frenchs Forest, NSW
|
AUD
|
2.6
|
m
|
100%
|
||||
|
Austria
|
|||||||||
|
Novartis Austria GmbH
|
Vienna
|
EUR
|
1.0
|
m
|
100%
|
||||
|
Novartis Pharma GmbH
|
Vienna
|
EUR
|
1.1
|
m
|
100%
|
||||
|
Sandoz GmbH
|
Kundl
|
EUR
|
32.7
|
m
|
100%
|
||||
|
EBEWE Pharma Ges.m.b.H Nfg. KG
|
Unterach am Attersee
|
EUR
|
1.0
|
m
|
100%
|
||||
|
Bangladesh
|
|||||||||
|
Novartis (Bangladesh) Limited
|
Gazipur
|
BDT
|
162.5
|
m
|
60%
|
||||
|
Belgium
|
|||||||||
|
N.V. Novartis Pharma S.A.
|
Vilvoorde
|
EUR
|
7.1
|
m
|
100%
|
||||
|
N.V. Sandoz S.A.
|
Vilvoorde
|
EUR
|
19.2
|
m
|
100%
|
||||
|
S.A. Alcon-Couvreur N.V.
|
Puurs
|
EUR
|
110.6
|
m
|
100%
|
||||
|
N.V. Alcon S.A.
|
Vilvoorde
|
EUR
|
141 856
|
|
100%
|
||||
|
Bermuda
|
|||||||||
|
Novartis Investment Ltd.
|
Hamilton
|
USD
|
12 000
|
|
100%
|
||||
|
Novartis Securities Investment Ltd.
|
Hamilton
|
CHF
|
30 000
|
|
100%
|
||||
|
Novartis Finance Services Ltd.
|
Hamilton
|
CHF
|
20 000
|
|
100%
|
||||
|
Novartis B2 Ltd.
|
Hamilton
|
USD
|
12 000
|
|
100%
|
||||
|
Novartis B3 Ltd.
|
Hamilton
|
USD
|
106 400
|
|
100%
|
||||
|
Triangle International Reinsurance Limited
|
Hamilton
|
CHF
|
1.0
|
m
|
100%
|
||||
|
Trinity River Insurance Co Ltd.
|
Hamilton
|
USD
|
370 000
|
|
100%
|
||||
|
Brazil
|
|||||||||
|
Novartis Biociências S.A.
|
São Paulo
|
BRL
|
265.0
|
m
|
100%
|
||||
|
Sandoz do Brasil Indústria Farmacêutica Ltda.
|
Cambé, PR
|
BRL
|
190.0
|
m
|
100%
|
||||
|
Canada
|
|||||||||
|
Novartis Pharmaceuticals Canada Inc.
|
Dorval, Quebec
|
CAD
|
13.0
|
m
|
100%
|
||||
|
Sandoz Canada Inc.
|
Boucherville, Quebec
|
CAD
|
80.8
|
m
|
100%
|
||||
|
Alcon Canada Inc.
|
Mississauga, Ontario
|
CAD
|
2 500
|
|
100%
|
||||
|
CIBA Vision Canada Inc.
|
Mississauga, Ontario
|
CAD
|
82 886
|
|
100%
|
||||
|
Chile
|
|||||||||
|
Novartis Chile S.A.
|
Santiago de Chile
|
CLP
|
2.0
|
bn
|
100%
|
||||
|
Alcon Laboratorios Chile Ltd.
|
Santiago de Chile
|
CLP
|
2.0
|
bn
|
100%
|
||||
|
China
|
|||||||||
|
Beijing Novartis Pharma Co., Ltd.
|
Beijing
|
USD
|
30.0
|
m
|
100%
|
||||
|
Novartis Pharmaceuticals (HK) Limited
|
Hong Kong
|
HKD
|
200
|
|
100%
|
||||
|
China Novartis Institutes for BioMedical Research Co., Ltd.
|
Shanghai
|
USD
|
320.0
|
m
|
100%
|
||||
|
Suzhou Novartis Pharma Technology Co., Ltd.
|
Changshu
|
USD
|
103.4
|
m
|
100%
|
||||
|
Shanghai Novartis Trading Ltd.
|
Shanghai
|
USD
|
3.2
|
m
|
100%
|
||||
|
Sandoz (China) Pharmaceutical Co., Ltd.
|
Zhongshan
|
USD
|
36.5
|
m
|
100%
|
||||
|
Alcon Hong Kong Limited
|
Hong Kong
|
HKD
|
77 000
|
|
100%
|
||||
|
Alcon (China) Ophthalmic Product Co., Ltd.
|
Beijing
|
USD
|
60.0
|
m
|
100%
|
||||
|
Colombia
|
|||||||||
|
Novartis de Colombia S.A.
|
Santafé de Bogotá
|
COP
|
7.9
|
bn
|
100%
|
||||
|
Laboratorios Alcon de Colombia S.A.
|
Santafé de Bogotá
|
COP
|
20.9
|
m
|
100%
|
||||
|
Croatia
|
|||||||||
|
Sandoz d.o.o. farmaceutska industrija
|
Zagreb
|
HRK
|
25.6
|
m
|
100%
|
||||
|
Czech Republic
|
|||||||||
|
Novartis s.r.o.
|
Prague
|
CZK
|
51.5
|
m
|
100%
|
||||
|
Sandoz s.r.o.
|
Prague
|
CZK
|
44.7
|
m
|
100%
|
||||
|
Alcon Pharmaceuticals (Czech Republic) s.r.o.
|
Prague
|
CZK
|
31.0
|
m
|
100%
|
||||
|
Denmark
|
|||||||||
|
Novartis Healthcare A/S
|
Copenhagen
|
DKK
|
14.0
|
m
|
100%
|
||||
|
Sandoz A/S
|
Copenhagen
|
DKK
|
12.0
|
m
|
100%
|
||||
|
Alcon Nordic A/S
|
Copenhagen
|
DKK
|
0.5
|
m
|
100%
|
||||
|
Ecuador
|
|||||||||
|
Novartis Ecuador S.A.
|
Quito
|
USD
|
4.0
|
m
|
100%
|
||||
|
Egypt
|
|||||||||
|
Novartis Pharma S.A.E.
|
Cairo
|
EGP
|
193.8
|
m
|
99.77%
|
||||
|
Sandoz Egypt Pharma S.A.E.
|
New Cairo City
|
EGP
|
250 000
|
|
100%
|
||||
|
Finland
|
|||||||||
|
Novartis Finland Oy
|
Espoo
|
EUR
|
459 000
|
|
100%
|
||||
|
As at December 31, 2017 |
Share
capital 1 |
Equity
interest |
|||||||
|
France
|
|||||||||
|
Novartis Groupe France S.A.
|
Rueil-Malmaison
|
EUR
|
103.0
|
m
|
100%
|
||||
|
Novartis Pharma S.A.S.
|
Rueil-Malmaison
|
EUR
|
43.4
|
m
|
100%
|
||||
|
Sandoz S.A.S.
|
Levallois-Perret
|
EUR
|
5.4
|
m
|
100%
|
||||
|
Laboratoires Alcon S.A.S.
|
Rueil-Malmaison
|
EUR
|
12.9
|
m
|
100%
|
||||
|
Germany
|
|||||||||
|
Novartis Deutschland GmbH
|
Wehr
|
EUR
|
155.5
|
m
|
100%
|
||||
|
Novartis Business Services GmbH
|
Wehr
|
EUR
|
25 000
|
|
100%
|
||||
|
Novartis Pharma GmbH
|
Nuremberg
|
EUR
|
25.6
|
m
|
100%
|
||||
|
Novartis Pharma Produktions GmbH
|
Wehr
|
EUR
|
2.0
|
m
|
100%
|
||||
|
Sandoz International GmbH
|
Holzkirchen
|
EUR
|
100 000
|
|
100%
|
||||
|
1 A Pharma GmbH
|
Oberhaching
|
EUR
|
26 000
|
|
100%
|
||||
|
HEXAL AG
|
Holzkirchen
|
EUR
|
93.7
|
m
|
100%
|
||||
|
Salutas Pharma GmbH
|
Barleben
|
EUR
|
42.1
|
m
|
100%
|
||||
|
Aeropharm GmbH
|
Rudolstadt
|
EUR
|
26 000
|
|
100%
|
||||
|
Alcon Pharma GmbH
|
Freiburg im Breisgau
|
EUR
|
512 000
|
|
100%
|
||||
|
CIBA Vision GmbH
|
Grosswallstadt
|
EUR
|
15.4
|
m
|
100%
|
||||
|
WaveLight GmbH
|
Erlangen
|
EUR
|
6.6
|
m
|
100%
|
||||
|
Gibraltar
|
|||||||||
|
Novista Insurance Limited
|
Gibraltar City
|
CHF
|
130.0
|
m
|
100%
|
||||
|
Greece
|
|||||||||
|
Novartis (Hellas) S.A.C.I.
|
Metamorphosis/Athens
|
EUR
|
23.4
|
m
|
100%
|
||||
|
Alcon Laboratories Hellas- Commercial and Industrial S.A.
|
Maroussi, Athens
|
EUR
|
5.7
|
m
|
100%
|
||||
|
Hungary
|
|||||||||
|
Novartis Hungary Healthcare Limited Liability Company
|
Budapest
|
HUF
|
545.6
|
m
|
100%
|
||||
|
Sandoz Hungary Limited Liability Company
|
Budapest
|
HUF
|
883.0
|
m
|
100%
|
||||
|
India
|
|||||||||
|
Novartis India Limited
|
Mumbai
|
INR
|
140.7
|
m
|
73.4%
|
||||
|
Novartis Healthcare Private Limited
|
Mumbai
|
INR
|
60.0
|
m
|
100%
|
||||
|
Sandoz Private Limited
|
Mumbai
|
INR
|
32.0
|
m
|
100%
|
||||
|
Alcon Laboratories (India) Private Limited
|
Bangalore
|
INR
|
1.1
|
bn
|
100%
|
||||
|
Indonesia
|
|||||||||
|
PT. Novartis Indonesia
|
Jakarta
|
IDR
|
7.7
|
bn
|
100%
|
||||
|
PT. CIBA Vision Batam
|
Batam
|
IDR
|
11.9
|
bn
|
100%
|
||||
|
Ireland
|
|||||||||
|
Novartis Ireland Limited
|
Dublin
|
EUR
|
25 000
|
|
100%
|
||||
|
Novartis Ringaskiddy Limited
|
Ringaskiddy, County Cork
|
EUR
|
2.0
|
m
|
100%
|
||||
|
Alcon Laboratories Ireland Limited
|
Cork City
|
EUR
|
541 251
|
|
100%
|
||||
|
Israel
|
|||||||||
|
Novartis Israel Ltd.
|
Petach Tikva
|
ILS
|
1 000
|
|
100%
|
||||
|
Optonol Ltd.
|
Neve-Ilan
|
ILS
|
752 545
|
|
100%
|
||||
|
Italy
|
|||||||||
|
Novartis Farma S.p.A.
|
Origgio
|
EUR
|
18.2
|
m
|
100%
|
||||
|
Sandoz S.p.A.
|
Origgio
|
EUR
|
1.7
|
m
|
100%
|
||||
|
Sandoz Industrial Products S.p.A.
|
Rovereto
|
EUR
|
2.6
|
m
|
100%
|
||||
|
Alcon Italia S.p.A.
|
Milan
|
EUR
|
3.7
|
m
|
100%
|
||||
|
Japan
|
|||||||||
|
Novartis Holding Japan K.K.
|
Tokyo
|
JPY
|
10.0
|
m
|
100%
|
||||
|
Novartis Pharma K.K.
|
Tokyo
|
JPY
|
6.0
|
bn
|
100%
|
||||
|
Ciba-Geigy Japan Limited
|
Tokyo
|
JPY
|
8.5
|
m
|
100%
|
||||
|
Sandoz K.K.
|
Tokyo
|
JPY
|
100.0
|
m
|
100%
|
||||
|
Alcon Japan Ltd.
|
Tokyo
|
JPY
|
500.0
|
m
|
100%
|
||||
|
Luxembourg
|
|||||||||
|
Novartis Investments S.à r.l.
|
Luxembourg-Ville
|
USD
|
100.0
|
m
|
100%
|
||||
|
Novartis Finance S.A.
|
Luxembourg-Ville
|
USD
|
100 000
|
|
100%
|
||||
|
Malaysia
|
|||||||||
|
Novartis Corporation (Malaysia) Sdn. Bhd.
|
Kuala Lumpur
|
MYR
|
3.3
|
m
|
100%
|
||||
|
Alcon Laboratories (Malaysia) Sdn. Bhd.
|
Petaling Jaya
|
MYR
|
1.0
|
m
|
100%
|
||||
|
CIBA Vision Johor Sdn. Bhd.
|
Kuala Lumpur
|
MYR
|
10.0
|
m
|
100%
|
||||
|
Mexico
|
|||||||||
|
Novartis Farmacéutica, S.A. de C.V.
|
Mexico City
|
MXN
|
205.0
|
m
|
100%
|
||||
|
Sandoz, S.A. de C.V.
|
Mexico City
|
MXN
|
468.2
|
m
|
100%
|
||||
|
Alcon Laboratorios, S.A. de C.V.
|
Mexico City
|
MXN
|
5.9
|
m
|
100%
|
||||
|
Morocco
|
|||||||||
|
Novartis Pharma Maroc SA
|
Casablanca
|
MAD
|
80.0
|
m
|
100%
|
||||
|
Netherlands
|
|||||||||
|
Novartis Netherlands B.V.
|
Arnhem
|
EUR
|
1.4
|
m
|
100%
|
||||
|
Novartis Pharma B.V.
|
Arnhem
|
EUR
|
4.5
|
m
|
100%
|
||||
|
Sandoz B.V.
|
Almere
|
EUR
|
907 560
|
|
100%
|
||||
|
Alcon Nederland B.V.
|
Arnhem
|
EUR
|
18 151
|
|
100%
|
||||
|
New Zealand
|
|||||||||
|
Novartis New Zealand Ltd
|
Auckland
|
NZD
|
820 000
|
|
100%
|
||||
|
As at December 31, 2017 |
Share
capital 1 |
Equity
interest |
|||||||
|
Norway
|
|||||||||
|
Novartis Norge AS
|
Oslo
|
NOK
|
1.5
|
m
|
100%
|
||||
|
Pakistan
|
|||||||||
|
Novartis Pharma (Pakistan) Limited
|
Karachi
|
PKR
|
3.9
|
bn
|
99.99%
|
||||
|
Panama
|
|||||||||
|
Novartis Pharma (Logistics), Inc.
|
Panama City
|
USD
|
10 000
|
|
100%
|
||||
|
Alcon Centroamerica S.A.
|
Panama City
|
PAB
|
1 000
|
|
100%
|
||||
|
Philippines
|
|||||||||
|
Novartis Healthcare Philippines, Inc.
|
Manila
|
PHP
|
298.8
|
m
|
100%
|
||||
|
Sandoz Philippines Corporation
|
Manila
|
PHP
|
30.0
|
m
|
100%
|
||||
|
Poland
|
|||||||||
|
Novartis Poland Sp. z o.o.
|
Warszawa
|
PLN
|
44.2
|
m
|
100%
|
||||
|
Sandoz Polska Sp. z o.o.
|
Warszawa
|
PLN
|
25.6
|
m
|
100%
|
||||
|
Lek S.A.
|
Strykow
|
PLN
|
11.4
|
m
|
100%
|
||||
|
Alcon Polska Sp. z o.o.
|
Warszawa
|
PLN
|
750 000
|
|
100%
|
||||
|
Portugal
|
|||||||||
|
Novartis Portugal SGPS Lda.
|
Porto Salvo
|
EUR
|
500 000
|
|
100%
|
||||
|
Novartis Farma - Produtos Farmacêuticos S.A.
|
Porto Salvo
|
EUR
|
2.4
|
m
|
100%
|
||||
|
Sandoz Farmacêutica Lda.
|
Porto Salvo
|
EUR
|
499 900
|
|
100%
|
||||
|
Alcon Portugal-Produtos e Equipamentos Oftalmológicos Lda.
|
Porto Salvo
|
EUR
|
4.5
|
m
|
100%
|
||||
|
Romania
|
|||||||||
|
Novartis Pharma Services Romania S.R.L.
|
Bucharest
|
RON
|
3.0
|
m
|
100%
|
||||
|
Sandoz S.R.L.
|
Targu-Mures
|
RON
|
105.2
|
m
|
100%
|
||||
|
Alcon Romania S.R.L.
|
Bucharest
|
RON
|
10.8
|
m
|
100%
|
||||
|
Russian Federation
|
|||||||||
|
Novartis Pharma LLC
|
Moscow
|
RUB
|
20.0
|
m
|
100%
|
||||
|
Novartis Neva LLC
|
St. Petersburg
|
RUB
|
1.3
|
bn
|
100%
|
||||
|
ZAO Sandoz
|
Moscow
|
RUB
|
57.4
|
m
|
100%
|
||||
|
Alcon Farmacevtika LLC
|
Moscow
|
RUB
|
44.1
|
m
|
100%
|
||||
|
Saudi Arabia
|
|||||||||
|
Saudi Pharmaceutical Distribution Co. Ltd.
|
Riyadh
|
SAR
|
26.8
|
m
|
75%
|
||||
|
Singapore
|
|||||||||
|
Novartis (Singapore) Pte Ltd.
|
Singapore
|
SGD
|
100 000
|
|
100%
|
||||
|
Novartis Singapore Pharmaceutical Manufacturing Pte Ltd
|
Singapore
|
SGD
|
45.0
|
m
|
100%
|
||||
|
Novartis Asia Pacific Pharmaceuticals Pte Ltd
|
Singapore
|
SGD
|
39.0
|
m
|
100%
|
||||
|
Novartis Institute for Tropical Diseases Pte Ltd
|
Singapore
|
SGD
|
2 004
|
|
100%
|
||||
|
Alcon Pte Ltd
|
Singapore
|
SGD
|
164 000
|
|
100%
|
||||
|
Alcon Singapore Manufacturing Pte Ltd
|
Singapore
|
SGD
|
101 000
|
|
100%
|
||||
|
CIBA Vision Asian Manufacturing and Logistics Pte Ltd.
|
Singapore
|
SGD
|
1.0
|
m
|
100%
|
||||
|
Slovakia
|
|||||||||
|
Novartis Slovakia s.r.o.
|
Bratislava
|
EUR
|
2.0
|
m
|
100%
|
||||
|
Slovenia
|
|||||||||
|
Lek Pharmaceuticals d.d.
|
Ljubjana
|
EUR
|
48.4
|
m
|
100%
|
||||
|
Sandoz Pharmaceuticals d.d.
|
Ljubjana
|
EUR
|
1.5
|
m
|
100%
|
||||
|
South Africa
|
|||||||||
|
Novartis South Africa (Pty) Ltd
|
Midrand
|
ZAR
|
86.3
|
m
|
100%
|
||||
|
Sandoz South Africa (Pty) Ltd
|
Kempton Park
|
ZAR
|
3.0
|
m
|
100%
|
||||
|
Alcon Laboratories (South Africa) (Pty) Ltd.
|
Midrand
|
ZAR
|
201 820
|
|
100%
|
||||
|
South Korea
|
|||||||||
|
Novartis Korea Ltd.
|
Seoul
|
KRW
|
24.5
|
bn
|
98.55%
|
||||
|
Sandoz Korea Ltd.
|
Seoul
|
KRW
|
17.8
|
bn
|
100%
|
||||
|
Alcon Korea Ltd.
|
Seoul
|
KRW
|
33.8
|
bn
|
100%
|
||||
|
Spain
|
|||||||||
|
Novartis Farmacéutica S.A.
|
Barcelona
|
EUR
|
63.0
|
m
|
100%
|
||||
|
Sandoz Farmacéutica S.A.
|
Madrid
|
EUR
|
270 450
|
|
100%
|
||||
|
Sandoz Industrial Products S.A.
|
Les Franqueses del Vallés / Barcelona
|
EUR
|
9.3
|
m
|
100%
|
||||
|
Alcon Cusi S.A.
|
Barcelona
|
EUR
|
11.6
|
m
|
100%
|
||||
|
Abadia Retuerta S.A.
|
Sardón de Duero/Valladolid
|
EUR
|
6.0
|
m
|
100%
|
||||
|
Sweden
|
|||||||||
|
Novartis Sverige AB
|
Täby / Stockholm
|
SEK
|
5.0
|
m
|
100%
|
||||
|
Switzerland
|
|||||||||
|
Novartis International AG
|
Basel
|
CHF
|
10.0
|
m
|
100%
|
||||
|
Novartis Holding AG
|
Basel
|
CHF
|
100.2
|
m
|
100%
|
||||
|
Novartis International Pharmaceutical Investment AG
|
Basel
|
CHF
|
100 000
|
|
100%
|
||||
|
Novartis Bioventures AG
|
Basel
|
CHF
|
100 000
|
|
100%
|
||||
|
Novartis Forschungsstiftung
|
Basel
|
--
|
--
|
|
100%
|
||||
|
Novartis Stiftung für Kaderausbildung
|
Basel
|
--
|
--
|
|
100%
|
||||
|
Novartis Mitarbeiterbeteiligungsstiftung
|
Basel
|
--
|
--
|
|
100%
|
||||
|
Novartis Stiftung für Mensch und Umwelt
|
Basel
|
--
|
--
|
|
100%
|
||||
|
Stiftung der Novartis AG für Erziehung, Ausbildung und Bildung
|
Basel
|
--
|
--
|
|
100%
|
||||
|
Novartis Pharma AG
|
Basel
|
CHF
|
350.0
|
m
|
100%
|
||||
|
Novartis International Pharmaceutical AG
|
Basel
|
CHF
|
100 000
|
|
100%
|
||||
|
Novartis Pharma Services AG
|
Basel
|
CHF
|
20.0
|
m
|
100%
|
||||
|
Novartis Pharma Schweizerhalle AG
|
Muttenz
|
CHF
|
18.9
|
m
|
100%
|
||||
|
Novartis Pharma Stein AG
|
Stein
|
CHF
|
251 000
|
|
100%
|
||||
|
Novartis Pharma Schweiz AG
|
Risch
|
CHF
|
5.0
|
m
|
100%
|
||||
|
Sandoz AG
|
Basel
|
CHF
|
5.0
|
m
|
100%
|
||||
|
Sandoz Pharmaceuticals AG
|
Risch
|
CHF
|
100 000
|
|
100%
|
||||
|
Alcon Switzerland SA
|
Risch
|
CHF
|
100 000
|
|
100%
|
||||
|
Alcon Pharmaceuticals Ltd.
|
Fribourg
|
CHF
|
200 000
|
|
100%
|
||||
|
Roche Holding AG
|
Basel
|
CHF
|
160.0
|
m
|
33/6 2
|
||||
|
As at December 31, 2017 |
Share
capital 1 |
Equity
interest |
|||||||
|
Taiwan
|
|||||||||
|
Novartis (Taiwan) Co., Ltd.
|
Taipei
|
TWD
|
170.0
|
m
|
100%
|
||||
|
Thailand
|
|||||||||
|
Novartis (Thailand) Limited
|
Bangkok
|
THB
|
302.0
|
m
|
100%
|
||||
|
Alcon Laboratories (Thailand) Limited
|
Bangkok
|
THB
|
228.1
|
m
|
100%
|
||||
|
Turkey
|
|||||||||
|
Novartis Saglik, Gida ve Tarim Ürünleri Sanayi ve Ticaret A.S.
|
Istanbul
|
TRY
|
98.0
|
m
|
100%
|
||||
|
Farmanova Saglik Hizmetleri Ltd. Sti.
|
Istanbul
|
TRY
|
6.7
|
m
|
100%
|
||||
|
Sandoz Ilaç Sanayi ve Ticaret A.S.
|
Istanbul
|
TRY
|
165.2
|
m
|
99.99%
|
||||
|
Sandoz Syntek Ilaç Hammaddeleri Sanayi ve Ticaret A.S.
|
Istanbul
|
TRY
|
46.0
|
m
|
100%
|
||||
|
Sandoz Grup Saglik Ürünleri Ilaçlari Sanayi ve Ticaret A.S.
|
Gebze - Kocaeli
|
TRY
|
50.0
|
m
|
100%
|
||||
|
Alcon Laboratuvarlari Ticaret A.S.
|
Istanbul
|
TRY
|
25.2
|
m
|
100%
|
||||
|
United Arab Emirates
|
|||||||||
|
Novartis Middle East FZE
|
Dubai
|
AED
|
7.0
|
m
|
100%
|
||||
|
United Kingdom
|
|||||||||
|
Novartis UK Limited
|
Frimley/Camberley
|
GBP
|
25.5
|
m
|
100%
|
||||
|
Novartis Pharmaceuticals UK Limited
|
Frimley/Camberley
|
GBP
|
5.4
|
m
|
100%
|
||||
|
Novartis Grimsby Limited
|
Frimley/Camberley
|
GBP
|
250.0
|
m
|
100%
|
||||
|
Ziarco Group Limited
|
Frimley/Camberley
|
GBP
|
3 904
|
|
100%
|
||||
|
Sandoz Limited
|
Frimley/Camberley
|
GBP
|
2.0
|
m
|
100%
|
||||
|
Alcon Eye Care UK Limited
|
Frimley/Camberley
|
GBP
|
550 000
|
|
100%
|
||||
|
Glaxosmithkline Consumer Healthcare Holdings Limited
|
Brentford, Middlesex
|
GBP
|
100 000
|
|
36.5%
|
||||
|
United States of America
|
|||||||||
|
Novartis Corporation
|
East Hanover, NJ
|
USD
|
72.2
|
m
|
100%
|
||||
|
Novartis Finance Corporation
|
New York, NY
|
USD
|
1 000
|
|
100%
|
||||
|
Novartis Capital Corporation
|
New York, NY
|
USD
|
1
|
|
100%
|
||||
|
Novartis Services, Inc.
|
East Hanover, NJ
|
USD
|
1
|
|
100%
|
||||
|
Novartis US Foundation
|
New York, NY
|
--
|
--
|
|
100%
|
||||
|
Novartis Pharmaceuticals Corporation
|
East Hanover, NJ
|
USD
|
5.2
|
m
|
100%
|
||||
|
Novartis Institutes for BioMedical Research, Inc.
|
Cambridge, MA
|
USD
|
1
|
|
100%
|
||||
|
Corthera, Inc.
|
San Mateo, CA
|
USD
|
1
|
|
100%
|
||||
|
CoStim Pharmaceuticals Inc.
|
Cambridge, MA
|
USD
|
1
|
|
100%
|
||||
|
Encore Vision, Inc.
|
New York, NY
|
USD
|
1
|
|
100%
|
||||
|
Navigate BioPharma Services, Inc.
|
Wilmington, NC
|
USD
|
100
|
|
100%
|
||||
|
Reprixys Pharmaceuticals Corporation
|
Oklahoma City, OK
|
USD
|
1
|
|
100%
|
||||
|
Spinifex Pharmaceuticals, Inc.
|
Wilmington, NC
|
USD
|
1
|
|
100%
|
||||
|
Novartis Institute for Functional Genomics, Inc.
|
San Diego, CA
|
USD
|
1 000
|
|
100%
|
||||
|
Sandoz Inc.
|
Princeton, NJ
|
USD
|
25 000
|
|
100%
|
||||
|
Fougera Pharmaceuticals Inc.
|
Melville, NY
|
USD
|
1
|
|
100%
|
||||
|
Eon Labs, Inc.
|
Princeton, NJ
|
USD
|
1
|
|
100%
|
||||
|
Alcon Laboratories, Inc.
|
Fort Worth, TX
|
USD
|
1 000
|
|
100%
|
||||
|
Alcon Refractivehorizons, LLC
|
Fort Worth, TX
|
USD
|
10
|
|
100%
|
||||
|
Alcon Research, Ltd.
|
Fort Worth, TX
|
USD
|
12.5
|
|
100%
|
||||
|
Alcon Lensx, Inc.
|
Aliso Viejo, CA
|
USD
|
1
|
|
100%
|
||||
|
Alcon Laboratories Holding Corporation
|
Fort Worth, TX
|
USD
|
10
|
|
100%
|
||||
|
Novartis Vaccines and Diagnostics, Inc.
|
Cambridge, MA
|
USD
|
3
|
|
100%
|
||||
|
ClarVista Medical, Inc.
|
Aliso Viejo, CA
|
USD
|
1
|
|
100%
|
||||
|
Transcend Medical, Inc.
|
Menlo Park, CA
|
USD
|
1
|
|
100%
|
||||
|
Venezuela
|
|||||||||
|
Novartis de Venezuela, S.A.
|
Caracas
|
VEF
|
1.4
|
m
|
100%
|
||||
|
Alcon Pharmaceutical, C.A.
|
Caracas
|
VEF
|
5.5
|
m
|
100%
|
||||
|
In addition, the Group is represented by subsidiaries and associated companies in the following countries: Bosnia/Herzegovina, Bulgaria, Dominican Republic, Guatemala, Kenya, Latvia, the Former Yugoslav Republic of Macedonia, Nigeria, Peru, Puerto Rico, Ukraine and Uruguay
|
|||||||||
|
1 Share capital may not reflect the taxable share capital and does not include any paid-in surplus
|
|||||||||
|
2 Approximately 33% of voting shares; approximately 6% of total net income and equity attributable to Novartis
|
|||||||||
|
m = million; bn = billion
|
|||||||||