 Exhibit 99.1 |
 Forward Looking Statements
•
This presentation contains forward-looking statements within the meaning of The
Private Securities Litigation Reform Act of 1995. •
These statements involve risks and uncertainties that could cause actual results to
be materially different from historical results or from any future results
expressed or implied by such forward-looking statements.
•
Such
forward-looking
statements
include
statements
regarding:
the
therapeutic
potential
of
Infinity’s
Hedgehog
pathway,
Hsp90
and
PI3K
inhibitors;
the
potential
of
saridegib
and
Hedgehog
pathway
inhibition
in
addressing
chondrosarcoma,
pancreatic
cancer
and
myelofibrosis;
the
potential
of
IPI-145
and
PI3K
inhibition
in
addressing hematologic malignancies and inflammation; the potential of combination
therapy based on retaspimycin HCl in addressing non-small cell lung cancer
and the expectation that Infinity: will provide an update on the
Phase 1b portion of the trial of saridegib in combination with gemcitabine in
patients with pancreatic cancer at ASCO GI in January 2012; will report data
in the second half of 2012 from the Phase 2 portion of the trial of saridegib in combination with gemcitabine
in patients with pancreatic cancer, the Phase 2 trial of saridegib in patients with
myelofibrosis, the dose-escalation portion of the Phase 1b/2 trial of retaspimycin
HCl
in
combination
with
everolimus
in
patients
with
NSCLC,
and
each
of
the
Phase
1
trials
of
IPI-145;
and
will
complete
enrollment
in
the
second
half
of
2012
in
the Phase 2 trial of saridegib in patients with chondrosarcoma and the Phase 2
trial of retaspimycin HCl in combination with docetaxel in patients with NSCLC.
Such forward-looking statements also include estimates of 2012 financial
performance (including revenue, net operating cash burn, and year-end cash and
investments balance) and the expectation that Infinity will have
a cash runway to support its current operating plan through key
inflection points.
•
Such statements are subject to numerous factors, risks and uncertainties that may
cause actual events or results to differ materially from the company’s current
expectations. For example, there can be no guarantee that Infinity’s strategic
alliance with Mundipharma will continue for its expected term or that it will fund
Infinity’s programs as agreed, that any product candidate Infinity is
developing will successfully complete necessary preclinical and clinical development phases,
or that development of any of Infinity’s product candidates will continue.
Further, there can be no guarantee that any positive developments in Infinity’s product
portfolio
will
result
in
stock
price
appreciation.
Management’s
expectations
could
also
be
affected
by
risks
and
uncertainties
relating
to:
Infinity’s
results
of
clinical
trials
and
preclinical
studies,
including
subsequent
analysis
of
existing
data
and
new
data
received
from
ongoing
and
future
studies;
the
content
and
timing
of
decisions
made
by
the
U.S.
Food
and
Drug
Administration
and
other
regulatory
authorities,
investigational
review
boards
at
clinical
trial
sites
and
publication
review bodies; Infinity’s ability to enroll patients in its clinical trials;
unplanned cash requirements and expenditures, including in connection with business
development activities; development of agents by Infinity’s competitors for
diseases in which Infinity is currently developing its product candidates; and Infinity’s
ability to obtain, maintain and enforce patent and other intellectual property
protection for any product candidates it is developing. •
These
and
other
risks
which
may
impact
management's
expectations
are
described
in
greater
detail
under
the
caption
"Risk
Factors"
included
in
Infinity's
quarterly report on Form 10-Q for the quarter ended September 30, 2011 filed
with the U.S. Securities and Exchange Commission on November 8, 2011.
•
Further, any forward-looking statements contained in this presentation speak
only as of the date hereof, and Infinity expressly disclaims any obligation to update
any forward-looking statements, whether as a result of new information, future
events or otherwise. •
All trademarks used in this presentation are the property of their respective
owners. •
Our Internet website is http://www.infi.com. We regularly use our website to post
information regarding our business, product development programs and
governance.
We
encourage
investors
to
use
www.infi.com,
particularly
the
information
in
the
section
entitled
“Investors/Media,”
as
a
source
of
information
about
Infinity. References to www.infi.com in this presentation are not intended to, nor
shall they be deemed to, incorporate information on www.infi.com into this
presentation by reference.
2 |
 3
Average survival for newly diagnosed,
metastatic pancreatic cancer:
Less than six months. |
 4
Standard treatment for
unresectable chondrosarcoma:
None. |
 5
Historical response rate to docetaxel for
second-line NSCLC:
8%. |
 6
The urgency is real.
The time is NOW. |
 Sustainable model
for value creation
Building a Fully Integrated
Biopharmaceutical Company
7
Breakthrough
Science
Deep,
Diverse
Pipeline
High-value,
Enabling
Partnerships
Full U.S.
Commercial
Rights |
 Promising Pipeline Based on Breakthrough
Science
8
1H’12
1H’13
2H’13
2H’12
Phase 2
Phase 1
Phase 2
Phase 2
Phase
1
Phase 2
Hedgehog: Saridegib
Hsp90: Retaspimycin HCl
PI3K: IPI-145
Pancreatic Cancer
Chondrosarcoma
NSCLC (Heavy Smokers)
Myelofibrosis
NSCLC (mKRAS)
Inflammation
Hematologic Malignancies
2012
2013
Data
Randomized, double-blind, placebo-controlled trial.
Data
Data
Phase 1b/2
Data
Data
Data
Data |
 |
 Saridegib
Clinical
Development
Market
Potential
•
Key features:
–
Potent, oral Smoothened inhibitor
–
Clinically active
–
Well-tolerated as single agent and in combination
•
Lead development program with three Phase 2 trials
–
Key data anticipated in 2012
–
Clear registration paths
•
Robust biomarker strategy
•
First-in-class potential in multiple indications*
–
Pancreatic cancer: ~100,000 patients
–
Myelofibrosis: ~30,000 patients
–
Chondrosarcoma: Ultra-orphan
Saridegib: Establishing a Leadership Position in
Underserved, Life-Threatening Diseases
10
*Estimates based upon G7 countries (US, UK, IT, DE, ES, FR, JP)
|
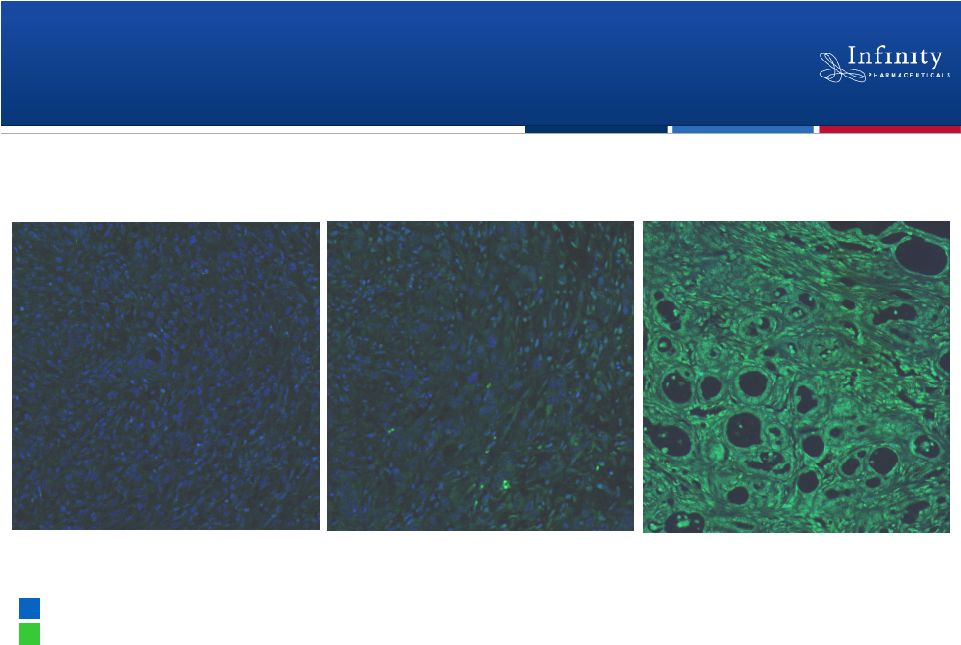 Saridegib in Pancreatic Cancer: Enables
Chemotherapy to Reach Tumor
11
Vehicle
Gemcitabine alone
Saridegib +
gemcitabine
Current standard of care
in pancreatic cancer
Tumor cell nuclei
Fluorescent contrast agent
Saridegib
+
gemcitabine
doubles
median
survival
in
a
mouse
model
of
pancreatic
cancer
(Olive et al. 2009, Science 324: 1457-61.)
|
 New
Scientific Insights: Saridegib’s Effect on Tumor Vasculature Enhances
Tumor Access 12
Control
Bevacizumab
Saridegib
Campbell et al., AACR Tumor Microenvironment Complexity Conference, 2011.
Saridegib increases microvessel density, vessel volume
and vessel function to enhance tumor access |
 Saridegib: Rigorous Phase 2 Trial in
Metastatic Pancreatic Cancer
13
•
Randomized, double-blind, placebo-controlled trial
–
Primary endpoint: Overall survival
–
Dose: 160 mg saridegib (QD) and 1000 mg/m
2
gemcitabine (Q3W)
•
Enrollment complete; Data anticipated 2H’12
–
Phase 1b data to be updated at ASCO GI in January 2012
Dose
Escalation
MTD
1:1
Randomization
Saridegib + gemcitabine
(n = 60)
Placebo + gemcitabine
(n = 60)
Phase 1b
Phase 2 |
 Saridegib: Encouraging Activity and
Tolerability in Phase 1b
•
Saridegib + gemcitabine led
to a 31% response rate
–
Overall response rate to
gemcitabine is historically
<10%*
•
Favorable PK and safety
profile
–
No interaction between
saridegib and gemcitabine
–
Most common AEs were
fatigue and nausea
–
Combination did not reveal
unique or more severe AEs
14
Stephenson et al., ASCO 2011.
*Moore, et al. J Clin Oncol 25:1960-6.; Seitz et al. Oncology
18:43-7. |
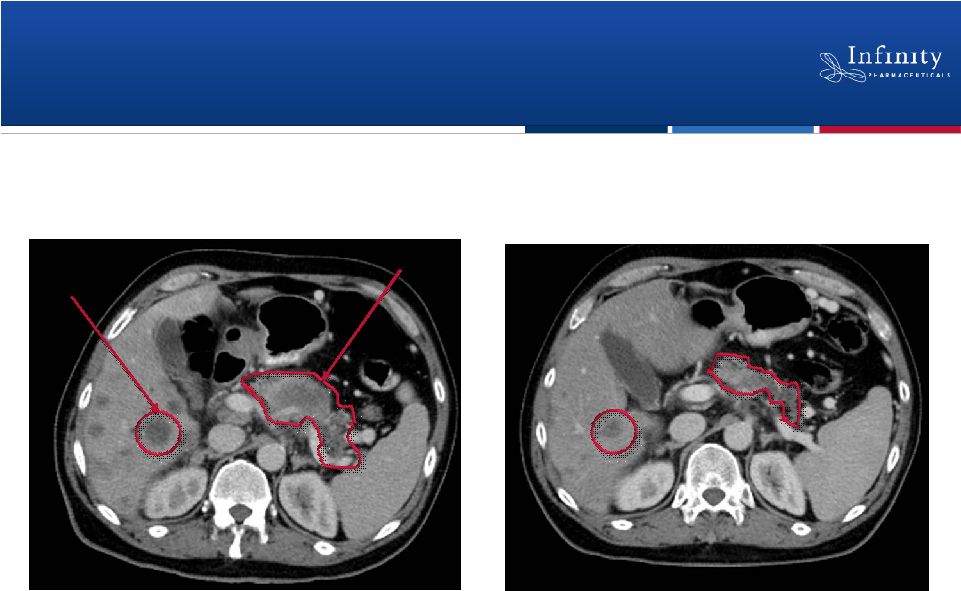     Saridegib: Evidence of Activity in
Pancreatic Cancer
Pre-treatment
After 4 months of treatment
Pancreatic mass and liver metastases
Reduction in primary mass and metastases
Liver
metastasis
Pancreas
15 |
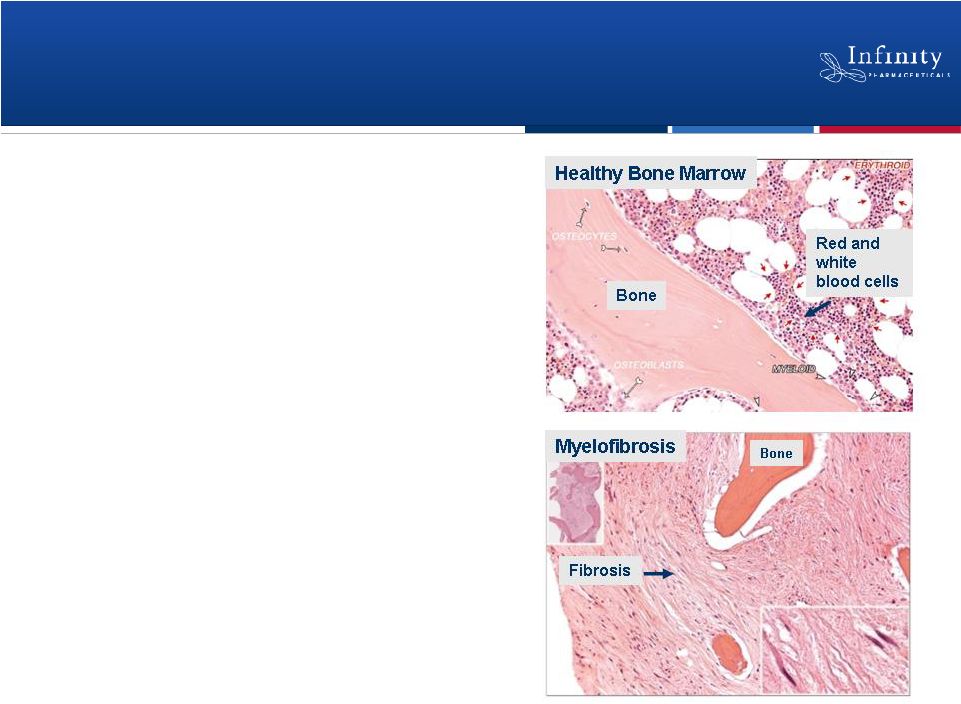 •
Blood cancer characterized by bone
marrow failure and enlarged spleen due
to pathogenic fibrosis
•
In myelofibrosis, Hedgehog ligand
expressed in bone marrow
•
Current treatments target symptoms,
not underlying fibrosis
Saridegib in Primary Myelofibrosis: Hedgehog Pathway
Plays Key Role in Pathogenic Fibrosis
16
Tkachuk and Hirshmann, Wintrobe’s Atlas of Clinical
Hematology, 2007. |
  Saridegib 160 mg (QD)
(N = up to 45)
Saridegib 160 mg (QD)
(N = up to 45)
Saridegib 160 mg (QD)
(N = 12)
17
Saridegib: Phase 2 Trial in Myelofibrosis
•
Open-label, exploratory trial with option for expansion
–
Primary endpoint: Response rate according to International Working Group
Criteria
–
No spontaneous remissions in this disease
•
Data anticipated in 2H’12
Expansion |
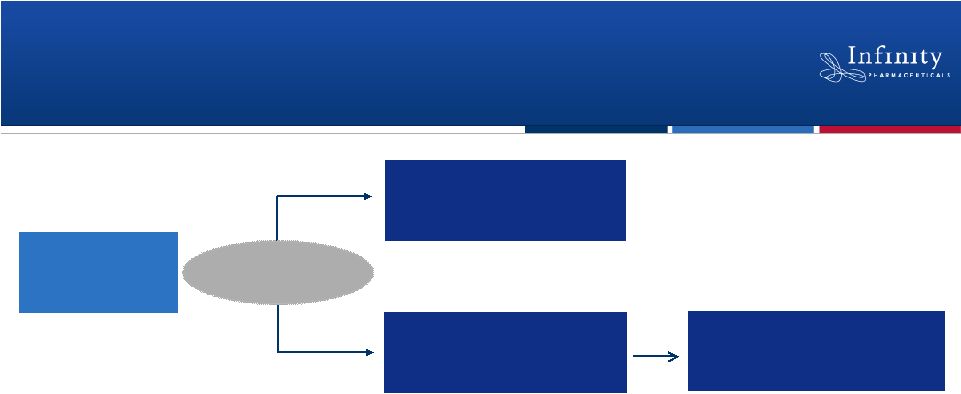 Saridegib: Global, Randomized Phase 2 Trial in
Chondrosarcoma
•
Randomized, double-blind, placebo-controlled trial
–
Enrolling patients with metastatic or locally advanced, unresectable
chondrosarcoma
–
Primary endpoint: Progression-free survival
•
Strong preclinical rationale: Saridegib inactivates Hedgehog signaling in
chondrosarcoma, killing tumor cells*
•
Enrollment completion anticipated 2H’12
~140 Patients
Saridegib 160 mg
(QD)
(N = ~94)
Placebo
(N = ~46)
Progression -
crossover to saridegib
18
2:1
Randomization
*Read, AACR 2011. |
 Retaspimycin HCl (IPI-504)
Targeting Non-Small Cell Lung Cancer Through
Hsp90 Inhibition |
 •
Key features:
–
Selective and potent Hsp90 inhibitor in Phase 2 development
–
Well-defined and manageable safety profile with QW dosing
–
Clinical activity in combination with docetaxel in NSCLC
•
Two trials ongoing in NSCLC
–
In combination with docetaxel in heavy smokers
–
In combination with everolimus in mKRAS
•
Robust biomarker strategy
Retaspimycin HCl: Leading the Field in Hsp90
20
*Estimates based upon G7 countries (US, UK, IT, DE, ES, FR, JP)
•
Targeting unmet need in NSCLC patient subpopulations*
Heavy smokers: ~180,000 patients
Squamous cell carcinoma: ~145,000 patients
mKRAS: ~125,000 patients
Retaspimycin
HCl
Clinical
Development
Market
Potential |
 Retaspimycin HCl:
Phase 2 Trial in NSCLC Patients with a Smoking History
•
Randomized, double-blind, placebo-controlled trial
21
~200 smokers w/
2
nd
-
or 3
rd
-line
NSCLC
(docetaxel naïve)
Follow-up for OS
Follow-up for OS
Docetaxel +
Retaspimycin HCl
(N = ~100)
Docetaxel +
placebo
(N = ~100)
R
Primary endpoint: Overall survival
Secondary endpoints: Predictive biomarkers, progression free survival,
overall response rate
Dose: 450 mg/m
2
retaspimycin HCl (QW) and 75 mg/m
2
docetaxel (Q3W)
•
Enrollment completion anticipated 2H’12 |
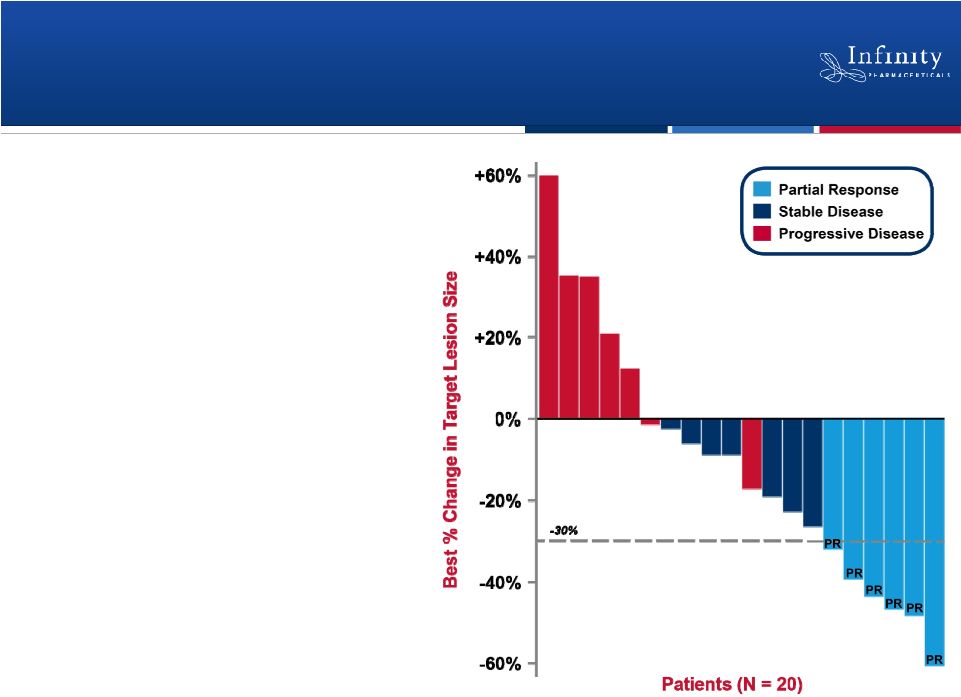 Retaspimycin HCl Phase 1b Trial: Clinically
Active in Combination with Docetaxel
22
Encouraging Phase 1b data
•
Partial response in 6 patients
(ORR = 26%)
•
Stable disease in 7 patients
•
Well-tolerated
–
No unexpected or overlapping
toxicities
–
No dose reductions or
discontinuations due to liver
function tests
Riely et al., ASCO 2011. |
 Retaspimycin HCl: Responses Observed in
Patients with Historically Poor Prognoses
23
*Hanna et al, J Clin Oncol, 22:1589-97. |
 Retaspimycin HCl: Phase 1b/2 Trial in
NSCLC Patients with mKRAS
Retaspimycin HCl
+ everolimus
(N = 12)
Determine
recommended
Phase 2 dose in
mKRAS NSCLC
•
Small, open-label exploratory trial with option for expansion
–
Primary efficacy endpoint: Response rate
–
Neither drug active as single agent in these patients
•
Strong preclinical rationale: Evidence of substantial tumor regression in a
NSCLC model*
•
Data anticipated 2H’12
24
Retaspimycin HCl
+ everolimus
(N = up to 45)
Expansion
Phase 1b
Phase 2
*De Raedt et al., 2011; Cancer Cell 13;20(3):400-13.
|
 |
 IPI-145
Clinical
Development
Market
Potential
•
Key features:
–
Oral
–
Potent PI3K-
inhibitor
–
Active in preclinical models of inflammation
•
Dual development path:
–
Inflammation
–
Hematologic malignancies
•
Robust biomarker strategy
IPI-145: Only PI3K-
,
Inhibitor in the Clinic
26
*Estimates based upon G7 countries (US, UK, IT, DE, ES, FR, JP)
|
 The
Power of PI3K- ,
Inhibition
27 |
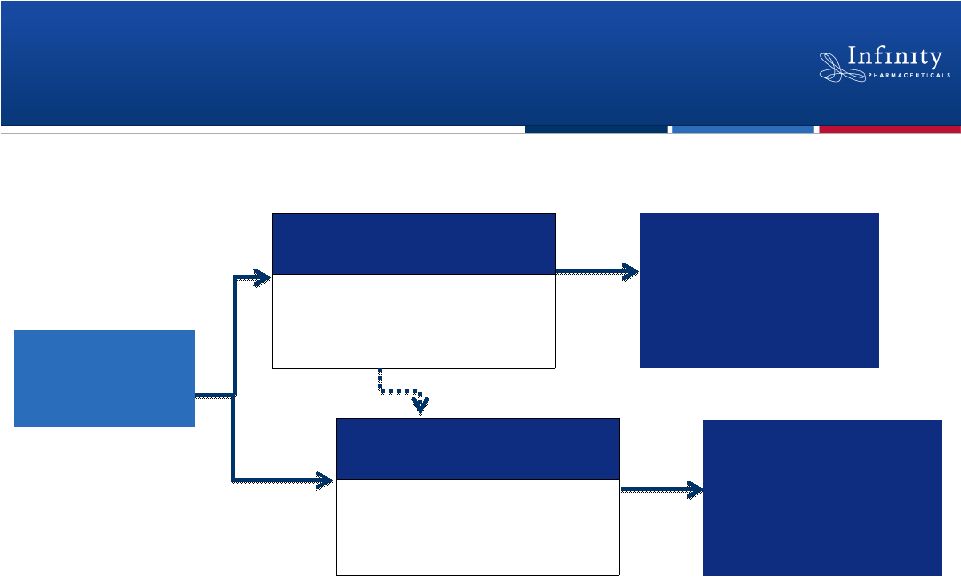           IPI-145: Dual Clinical Development Paths in
Inflammation and Hematological Malignancies
IPI-145
Phase 1 in Hematologic
Malignancies
•
Dose Escalation
•Expansion Cohorts
Phase 1 in
Healthy Subjects
•
Single Ascending Dose
•
Multi Ascending Dose
Phase 2 in
Inflammation
Phase 2 in
Hematologic
Malignancies
28 |
 CONFIDENTIAL
29
Large Addressable Patient Populations |
 CONFIDENTIAL
30
Large Addressable Patient Populations |
 CONFIDENTIAL
31
Large Addressable Patient Populations |
 CONFIDENTIAL
32
Large Addressable Patient Populations |
 CONFIDENTIAL
33
Large Addressable Patient Populations |
 CONFIDENTIAL
34 |
 CONFIDENTIAL
35
Large Addressable Patient Populations |
 |
 2012 Financial Guidance:
Cash Runway Through Key Inflection Points
•
Over $323M in current and committed capital
–
Approximately $115 million in cash as of December 31, 2011
(unaudited) –
Over $208 million in committed R&D funding through 2013 from
Mundipharma
•
2012 Financial Guidance
–
Revenue: ~$114 million
–
Net operating cash burn: $30 -
$40 million
–
Year-end cash and investments balance: $75 -
$85 million
37 |
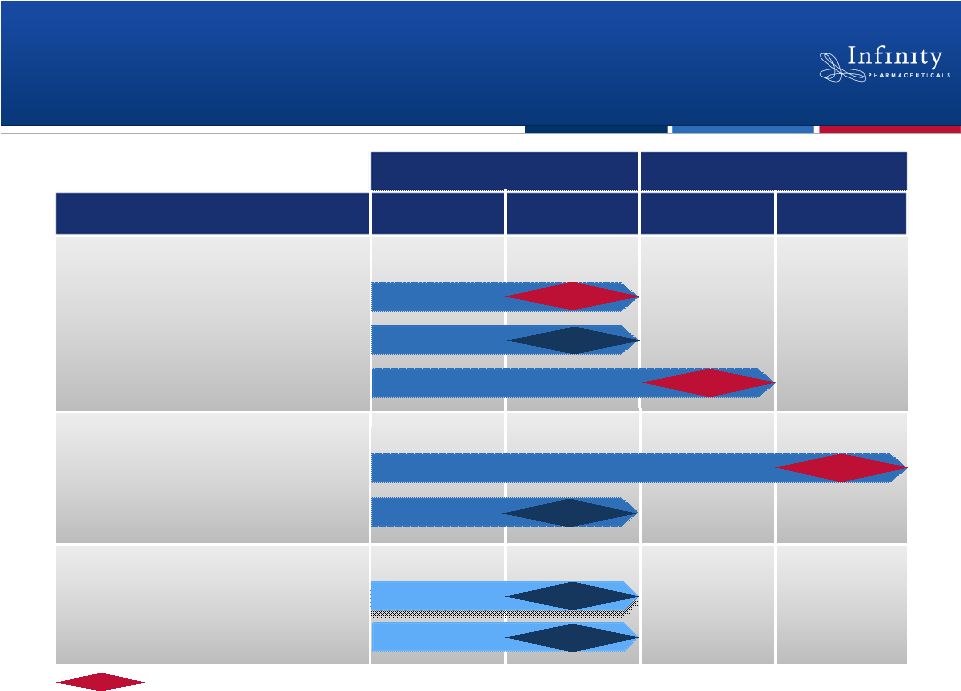 Promising Pipeline Based on Breakthrough
Science
38
1H’12
1H’13
2H’13
2H’12
Phase 2
Phase 1
Phase 2
Phase 2
Phase
1
Phase 2
Hedgehog: Saridegib
Hsp90: Retaspimycin HCl
PI3K: IPI-145
Pancreatic Cancer
Chondrosarcoma
NSCLC (Heavy Smokers)
Myelofibrosis
NSCLC (mKRAS)
Inflammation
Hematologic Malignancies
2012
2013
Data
Randomized, double-blind, placebo-controlled trial.
Data
Data
Phase 1b/2
Data
Data
Data
Data |
 |
