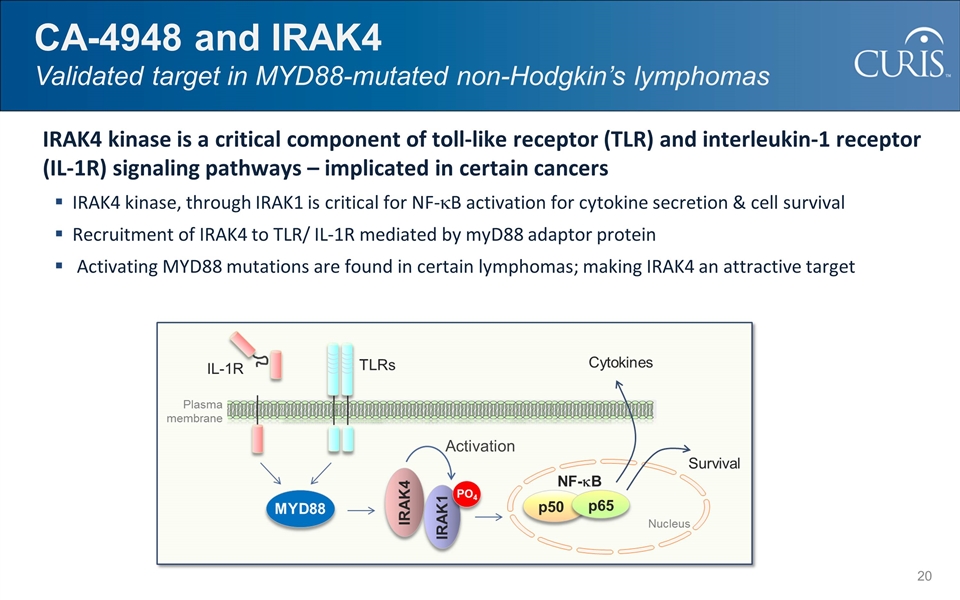
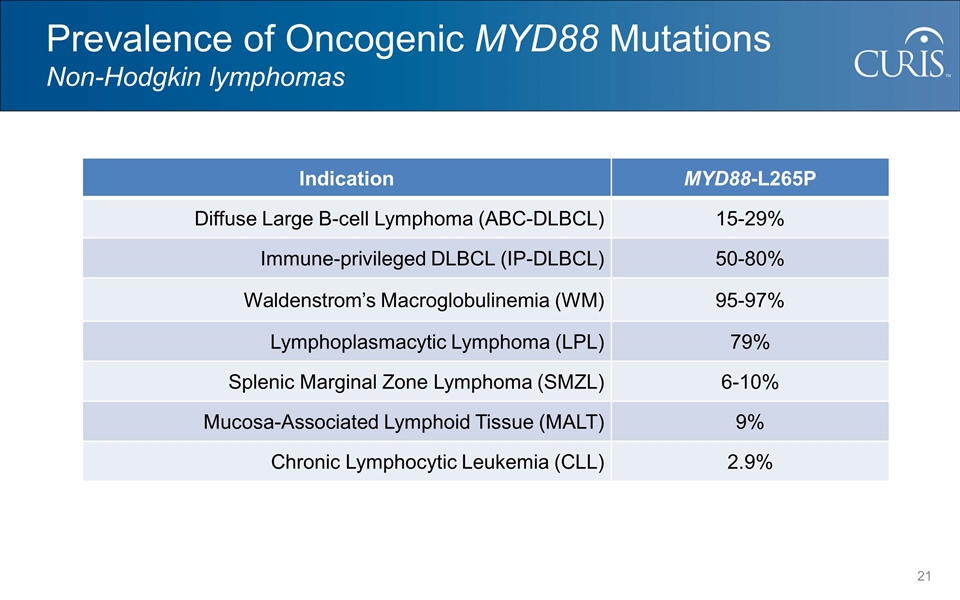
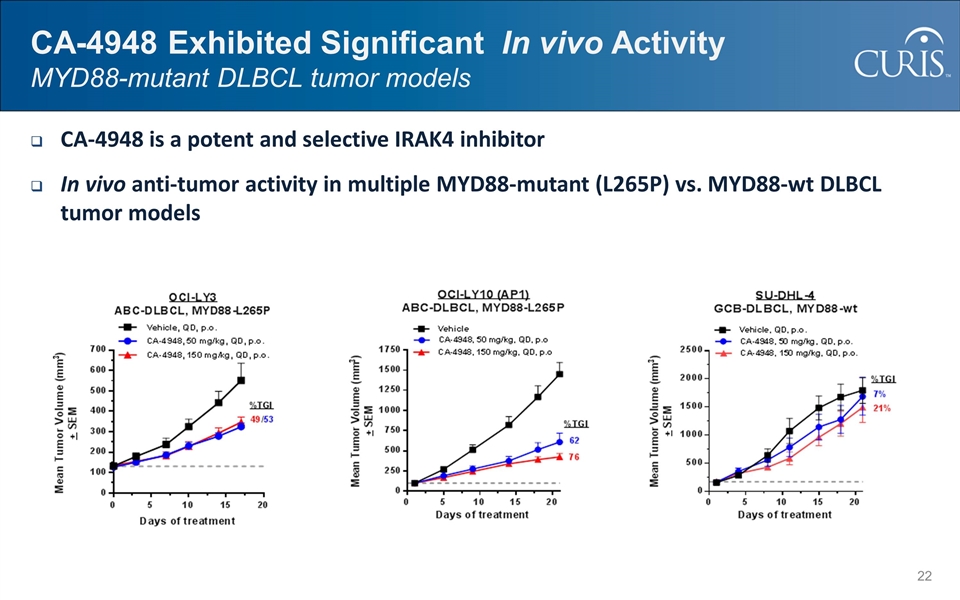
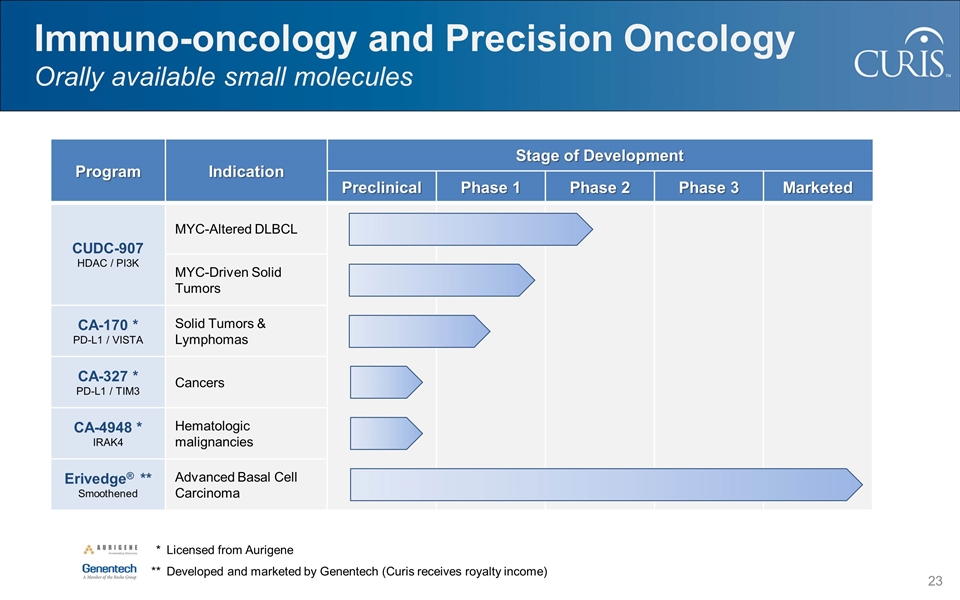
@:OIM]I&
MNZ1J_B;Q%INB26NHZ5J5M;:A87MHM_Y\MO=VT4L:J'9=A4GZV_X**?"'X9^#
M_P!O7XO?L_\ [*OPXU2T\*^!=>T#X;>%_ WAEO%/CC7]:\6Z/X3TBX\87%C#
M=W.O^(M3O[SQ#-J[S6-L\T5I'9.MM;P01.!^@_[%/QN\4?\ !2#_ (*K_L??
M$?XA?#SPYH?C3X'_ R.K_$CQ/H4]Q<7_P 6=0^"_A74YM"\?^+8KBUM+.R\
M3W_B74M"M!':I]DMX?LEFDPM[.TBA]%^/37_ .P_\ ?BS^V+^SKXT\ _&3]J
M7]H/]IGXK?"+]H;]ISPE??\ "20_LK:QXFFO_%US\*_@J\UJUE#KVI6UZ_AW
MQ!\6?,EDEU"SBM]#A2!])%CXN.XSS6CQ7E&78K"1P^>5^&Z>#I9'#,:D\ECG
MF?9S:EBLPS'#QG0>#PN#X?\ ;X:4H5,7-9E#!X;#O%XF<8^QEW >38C@S.M&M+&XO%\0TJ6)ITJL,+3E@
M*N+Q>(6"PBJ2_GHG^%WQ*M;SQE8W/P\\^-=9\?7(UR._CU;7O#]YK6O:OI-WJ@O/+\:6&D:K/Y\UK\S_V//CE\8/V
MB?V'O^"J?@[]ICXB>-?C#\,_ _[/FC_%CPQJ_P 4O$.I^--0\&?&6/6M9E\,
MWWAW7_$5Q>W^EWNM7VG6TXT^VNTA>>Q!M+>+[=>K=>O7XSSW#U!A. N'<32RC SS7-J6?<0<-9KQ'@L.L%AZF#RVC@UFN*P-#,JWMJ=6L\QR[
M+/:N>%PU-826(IU)>TI\T8_BY\/?AO\ $+XJ:\GA/X8> _&GQ&\4SQFXB\.>
M!/#&N>+-:^S*VQKJ73]"LKZXAM0_R&YG2.W$A"F0'BK/Q#^&/Q)^$?B%O"7Q
M5^'OC?X:^)UMUN_^$>\>>&-=\)ZPUJS%%NHK#7+&QGN;0N"@NK9)K?S 4\W>
M"H_8[]IGX@^,O^">_P"Q]^R/^S-^SWKVI?##X@?M(?!W1OVH?VH/BKX-N[C1
M/B#XRN/')EB\#^ +?Q=I[6VN:1X3\.:='?V[Z?I5]:BX2SM3NB&I:VNI9>N>
M._'/[0G_ 19^(WCG]H'6M;^(?B_X!_M;>"_"'[/?Q'\:7ESKWC7^S/%V@Z-
M)XU\!0^*=2DFUK6=%@L]0N[G['=WEZ\,D%A!(=FBV?V;JH\:9I6>7YLLMP$>
M&,SXAH\/82!FL5.K&I&6'7
M+7\/\GIO-,D_M3,9<6Y5PS4XEQL5AV^N/$T,%7C">
M-5+V3S&+P<:+IR6)?Y^:U;_#33OV3_AMX?/[/'Q+TSX_^._BQJVO6/[0NNV_
MB"S\$>-_A=9V<^CV/P]^&NF+>C2?$6HVFOSZ7/J^IVFAW%Y%>/-81ZDYN8+=
M?._#7[-G[1'C/7?$_A;PC\!OC+XF\2^"2B^-- T+X:^-=5UGP?)+!'=PP^*=
M,L]%EO-!NKBTFANK:SU.*VN[FVFBF@ADCD5C^_WQ0^%D?B+_ (* ?\$=?V$X
MHI)](_9R^"7P7\1>.-,))B@\07*R_%[QY+\"_ 7XH>+_A)\,=;_:[T;PYX*\&?#'6+SPAHWB")
M?B'8:%XE^(GCRUT22"/Q_P"*?&>EZ9J.KZWJ'B[^UX8=&FMO#]E!;:+IT%J/
M#RWC+.*]&;R; 8.5.OD^;\98S&YUF>85:&!P$\YQU#*Z-*G3C4Q'+CL!@:F)
MIX:E[+#X2,:DH3C",:57W,VX$R.C6H+.,PQ?-A\\R3@? 8'(LHP%#%9ACZ.2
MX'$YO4J2E6H4?:Y?C\RP^'J8FLL16QBE%RIQ>E#\POV7?V9?'G[3'Q_\!?!#
M0M!\6VG]N^.O#OACX@:YI_AC5=4E^&.@:CK\>D:]XH\46QM%@\/0Z'$E\/\
MBI)--M7U2V339Y4F9DK9_;4^">D_L]_M-?%GX4^%?#/C[P]X \)^*=0T'X?7
MGQ'L=2M=?\9>&_#L@\/3>.8KJ_TO1X=6TSQ/KVEZQJ5CJ&CV,>B^3,MIII,%
MLIK][_@;\0I_^'WG_!07XO>$]>U3P]\(?@[X(^-7Q#^*FCZ!JMSI7AGQM-\)
MO"&G>#K>/Q9964T5CKUNWCJ;6O$EM;ZBLT?]M6S:@J?:B\M?S8?%+XR_%+X[
M>*'\>?%[Q[XO^(GBFXMOLMOJWC/7]3\0ZAIFCM>7FI6GA_3[G4[BYELM%TRX
MU*[^P:9 R6MF)I%AB0,17K2X;@_(<57R_VN(G7I9GQ
M'2J8_D=X*G4Q.$6"5*4IR4(82O3<5[:M54/#XJX=X:X9X2>%@\3C>(,1QKQ#
M@:&8NC1IT*N4\-5*. 4Z=Z]6K3H8ZICJE7E@G5EB\-.$W[&A0Y/3?V3+'P'=
M?''PG?\ Q5^#'Q(_: ^%_ANUU_Q'X_\ A9\*[;5)O%6O^']/T6^C2:6YTBYL
M;S2M TO6;K2=0U[51J.G);V-NT+7D9N5!\YU/POK_B636O'/@_X=>*+3X>:Q
M\0+SP[X:ETK1-?UK0-+U77KRXU+PO\.K;Q L%]#J/B>+1I[2"RTAKZZUS488
M1=+'<^89G_3S_@FS+-\)_P!E?_@I_P#M0^8MI>>&OV('TJXCLWR#]J@CL]%G(1@PC<,=H*,?:]5^-OBS]B#_@D7^QWX:^&,]KX8^-
M'[37Q0^+WQLTWQU':VEQXI^''@^P?_A"_P#A)/!&]-\
M6VD::SH^A:AKYT:ZLKN^%S'SX_BC&X?B3&X?*L!''XVIG.2\)86EB'A2KTL/4R_#UJ"KXFE1G5JPE&G4C-T*,3JR?@W!8CA3 8O.O'#T)WJT
M''V^)E+\:_B#\%/C+\'I=%7XM?";XD?# ^(;>2[T'_A8/@GQ)X/&M01HKROI
M;:[IMBE\84DCDGB@9YH$D1IXXPU?>^N_LO>%K'_@G'^S%X]\-_"O7_&_[5'[
M47[0WQ*B\&W?AF+Q;X@\3R_!GX;V=]I5QH^C^"M%GNK"[M[K7H+"]N=6_L*:
M_ABO3NU"*V&![7\6OBC\1_$7_!$SP%JOQQ\>>+_BCXQ^*G[;VOWOPLU;X@:[
MJ'BOQ!HGA+P!X2U6SU\:;KFLS7>KFQO-=@U:V>$W4L,9U>2&((LJ(?7/^"B7
M[3/Q0_8_^%?[&G[%7P-\6ZC\+=?^'O[*/@76_C!X\\&R#0_B)/>?$""/59_
M-AXOLPFO>&_#ESJ.F7OB;Q-9:+>:?/XBO[C1?[3GFM-,A@D\O$\1\29OB>'L
MKP=#!TLUI\9\0X7%3P^-Q^&RK&8+A7!5J=7$U7*G5QOU1YGC<#1K8.5*:Q%6
MFZ$9*%256E[6#X5X5R7#\2YKC,3CZ^2U>"N'<1@HXG Y?7SG 9GQ;F.&EA,-
M1@Z\<$\;4RG!9E6PV,56FJ&'Q/MI<[I^RK?AE\0/AQ\1/A/K\WA3XI> _&/P
MV\46]M%>R>'O'?AW6O"FL"QFWB&^6PUNSLKB6QE:.18[R%)+9VBE42[HW ^_
M_ WP<^%7QG^/7_!/G]F?0/V>OB/\(?&/B:Z\'Z;^T;KGQ$/B#2M4^,BZSK%G
MK6L^,/#6DW>H/_8?@L>$M$\36VBWVDV&B275O="8)+<6:W ^M_VRM&\9?&_X
M>_\ !$_X!?$?5M:\<_'#XG>!K/6_%GB_Q+>3:MXRO?"/QB^(7A.P\,Z5K.JW
M1DU+4(-/T&"]D6:^EFN&&F23/-++)-*?K'0/'&B>)O\ @M;^U]^T+J%J+_X=
M?L$?L_?$.YL+.&1C##'\)_AUIO@&RTRS= ZPRW6NZYXP6V,:C%RLTL8!#FN+
M,^-L=B\G6,^KU,/F>%X?XSS"+R_,,11PM3&X#,'PIDU2G"+IK$4,TQN(JXK"
M4L8FL)*E3J)5*D85J?H9-P%E^#SRI@9XGZUE6-XAX%RV4F_&3]HOQM\"
M/@!\2=#_ &4O _Q7\9>$/!_B_2?"/CO7?AYI6A^"]2;PQ=7!\=:I#JL5U:2Z
MKIE_>W.HW6LW5M;/[T^XT#PIH.K7-IJ6M:L^HV<4:010O
M(Z2O$_Z6_P#!./\ ;D_:^^.O_!1CX1Z;XK^)GBOQ3X)^-/B+Q-X;\??!FXNG
MG^$&F_#*]\+>)=5O?#NC?#P*?"VAZ/X4M+:(Z?=66FP7LHMY/[1N[Q]3U W<
M/P&TSP_\!_V9O^"S_P 9O!\BV&A7?B%OV/\ X2W%NY2,Z/XY^).O6NI6&G3(
MS;[:W\+:AHC!8G(:UMHB08ER.S$9]G&5X'$\+9SAJ-7,%E7"V%P&(RW-\Q^L
M8E<09G_JW*&)S"5.CBZ&8X>K0Q.+>.P[C[5PG.$*#A=\N%X>R3-LPPW%N1XR
MM2RZ><\78O'8;,\IRYX;#SX:RNGQ2OJF ]MB<%6RROAZ]/!K XJ,72G&G2G[
M>E6C./SO\./V%]9\3_L4_M _M;:_\.OBZOB&R\8^$O"G[/\ \._#WA?Q!_9F
MHZ-XIN-+\1:U\4+U?[(N]=\1>!O"_@ZZO+32=3MY8?#DTUO)JFIZM>F%(Z^(
M/AQ\)/BO\8]0NM)^$7PQ^(?Q2U*QBCFOK/X>^#_$7BZ33XI1NBDU!M#T^]BL
M!.OS0?:WA,R@M&&7)K]@OVB_CA\?/A1_P30_X)T_";0/C-\3-,\=_'JT^)GC
M7Q#(O@_\./@E\.? ^M_%76/A[JU]X3\5
M_&#XS>-]$M?$?B#Q/XR\3:)+8ZY?6UI;S:?_ &?I8OUL[>6X>W=);73-(@LL
M1<,8BGD6-Q>)2@JE>%;'T<.\'
M1H*-;%5\=B,56K4L/&*GT9UPQPOAL-A,3*IFN"P/#_#_ S#.HT<-@)9AFG$
M?%F'J\08'"4)>V5&A4H9=7Q'U[$5Y/#8/#Y?0PU"C7K2E*/XY>*O"GBGP+KV
MH>%O''ACQ'X+\3Z0ZQZMX<\6Z+JGAS7=-9EW(;W2M7MK2]MED4%XI)85CE12
M\3,GS5WWASX#_'+Q5J\/A[PQ\&/BOXAUZZ\+VOCFUT71OA[XLU'4[CP5?"3[
M!XOAL[?2GGD\,ZBT$R:;K:H=.U%X94L;BX=&4?J9^TO#XQ_:C_X)Y?\ !.GX
MB?%2>\\3?M#^./CYX]_9W\.>/]6C$WC/X@_"R76KS2-$E\0ZHZB]\0#1=9CT
MZVM=2NS-(\S7%W),UQJ5T]QW_P#P5%_;C^+'PN_:9\1_LZ_LR^/M:^#_ ()^
M"7A3P!\+/&&M_#Z<:'XE^)/B+PIX*M=.^R>*M?C1]0NO#7@W2=5;P_X?\+P3
MPZ/9ZC-XAUB:TEU34C<0^IA^,\]S/$9?E.5Y5EDLVJ3XFI9I5Q.,Q?\ 96"?
M#>9X/*9XJA5I8>6*Q6'QU?$/ZO2=.%6$XSHU9THTJF(7!B."LCRK#9CFV9YI
MFSR>E1X6KY3'"X7!K-"Q$*N)6#PE? 4,*UB,1!U*-2DXUH*3K
M8>D_PG"O'(\;JR21L\(H_"G@307%I&Z32=/86K
M6_VR6"8O>7%M$5+3;A]Z_P##LSQ*.O[9O_!._P"H_:GTC'_J.&OJ%KU:,:T:7-3SIU$ESPFK-W]UZ6LW\QDW#F=9Q"KB<
MKR^KB\/3J2H2J1J4(*-5*G4Y'[6K"3DH3A)\JDM5K<_-RG"188KB5^$B1I&(
MSPJ1EFZ>P[\#WYKZ^^/?['6L? 'P3:>-[_\ :#_92^*D%WXBT_PX/#7P0^--
ME\0_&,$FH6FH7:ZO^U(SE;:ZN["$QM]I!'A'P:\!77Q
M5^+GPO\ AA91M+<_$3XD>!_!:QJ"28?$?B+3=+NWP.<0V=S<3.V"%6,L< $U
M6&SG+L5@JN9X7$QKX/#1KU*M:,9P26%I.O7TJQIOW*<7)MI1LMQXG)LRPF84
MI4DHKE;DGTN?L/\9_#'[#?[$WP
MR_9*\)_%?]C&/X]_&;XJ?L[>&OBW\2O$,OQL^(/@"32]4\13!K6VGTK2+B^T
M]GNF:^@1+.TTZ&V@TJ/]U-).THY;P_\ #']B;]M?X!_M1>)/@%^SSXQ_96^-
M'[,WPS3XRVX?XL:Y\2_ /CKPK9/?'5M#U)/$2)-IFH21:9=PV&_@[>:!\)O!
M?@:_\!:5:>"-/\RWL- U#7!)?7$N[5ICJDLS1PIJIN8(HXXX%#?#7Q,_;*_9
MI^%'[.7Q*_9G_85^%WQ,\+Z/\:&LHOCA\=?C9JVC7WQ*\;>%M)+RP^#]$TCP
M\\VG>']&N-T\%P8YK.*'3[[5(8=+GO\ 5)M4C_%\C6;YIE&1XG 8?C:/$V88
MS 9A7S7&YAF^&R##8?%YG1QN*=2AC,P_LS&8%97.IA,/A!R[^T
ML+CZF;4Z>.Q.,QF84:-!JK"O.HY1H3]%^!OPZ_9&^"W_ 3W\,_M8?M-?LYW
M?[0'B[XL?M">(OA[\/\ 0(/B3XH^'5Q;>$-!T>9+F]BNM'NC83VMIJN@:]//
MY^FW%W*+'6RMW9&\L[2>6%K."UN6MH+M[+4WOK
M>#3[_G?^"E,3?"G]FO\ X)J?LO K%>>#OV?+SXR>+K-?D>/Q1\5;NSG#72#G
M[0DJ>(U&\;@)6^;DY\P_X)!_#S6/'/[>WP/==ES'IOA
M_P +^'O#>K6"W&I71*QVR7VK:K8V,!E91(&N9%#I;3;>MTIXCA3B3C66=9Y@
ML;&KQ'FN2U:.655J.+6&P]&I1J823Q/UQ1ISC5J
M4Y+DC5IX;BSAC@2&1Y!CL"Z'"V4YU3K9#EDL=B<7C\OP>(SG$_VM'#0S>C6P
M3Q5>K2Q%/&P6%^IN4^:C"I R/^"=/[+_ (>^-/[;^D_!CXY^&VO?!/P]M/BK
MK7QB\.RZAJ.G1+!\-=.O]+O=.N-4TFZT_4;>"'Q9-IH-Q:7=J\T<('F+%*X/
MPEXLN]"OO%OBR^\+:='I'A6\\4^([GPMI$4]Q=1:5X9FUJ^D\/Z='D-9P">ZGFN)=GF32R2,SM^UO[*WCO3H=&_P""RW["?A
MS>L/+)U+XY^.?$-WISV3$#$\]LWA:ZPG[P)?<08G$5:U.A@,#PYE?U%5:CPU',*F EG>:^B/V=OV9/C;^U+XLN?!GP4\&R^);W2
M;:#4?%&MWM_::)X2\$Z-.O&WQ9T"_O9/"E]X0T.
MTT?1],\.>!+".^DTZ3PG!KVHW$EKXAN+:36=]>YM[&:'3;;\U]#UKQF
MNF:CX(\,>(O$>FV'CV\TG2=5\/Z/K6J:;IGB>^:YDL-"M]DF-=$_9@_9K^#GPAMK>
MV&VWM]5701XAUH1HGRK+)'J.DBXP!\\2CH..?,IYS4XQXSIQKU)5L2IJET97')*
M?!'$F._LRK4SB-7*Z?X&\*:WXHN+&&0$Q2WZZ/97:V*2A7,+7;0B4(WE[@IQY^K*D1=CM52S
M,S9P% !).?3GWQ^%?N-HWA3]J7X;?LI?!_X3ZQ^T'^S3_P $Z/"6J07GQ'F@
MU7XH^+_"7[07QT7Q*8YM,\7_ !+TOPAIWB#Q/IUE# /(TC1[>XTJV-J+:ROK
M"+^RX+2W]3B//)9+1P2H+!/%8[%K#TECZM54HTH4I5:]>&$P-.OF682I*-*+
MPV H2G%UZ=2M5H45S/@X4R.GGE3,'B7CUAPE2HTJ]62BOQK\7>!O&_PYUN?PS\0O!OBKP'XD@C2
M>;0?&7A_5O#6KI;N65+C^S]8M+.X:W9E94N(T>%V#!9"5(&_H?PA^+7B/6O#
M7A[P_P#"SXCZYKWC'13XD\(Z-I/@CQ+?ZGXG\-[C'_PD>@V5MILD^J^'C(#'
M_;EDDVE!P4-V&XK].?B)\2KWP5\
M;;:TU\WVM?#O4)8-"T?1['4?%MG8^,-5T2+6M:T*QM;_ %FWB?5)-+AO8%:W
M%O,]S_@J+^U'\1?@G\4_"_[*WP(\6:M\,-'^!7P0^%G@#QUXH\ 7TWA[QKXT
MU?2O#,&I:7X8U#Q;I;0:[#X2\*V.H0W-CX:L+^TTVY\0ZSJ^JZC!=W/V8V_S
M&7<99MFRR7#9=E^"GCLQK\2TZ]:O6Q5'+J&'X?Q^'RQ8^-.K@X9D\/B\7B*<
M*>&K4:6)C*].=6DHNM'ZS'<&Y5E$\ZQ.89CCH8#+J7#4Z-&AAL+7S'$8GB+
M5\S675)4\;/+H8C!8+#U:D\1AJL\-5CR58TJC:H2_&7Q5X2\6>!->OO"WCCP
MOXB\&>)]+,8U+P[XKT34O#VMV/G()86N=,U6VM;R*.:(B2"5H?*F3YHG=?%B#X4_$JZ^%]C'+/=?$2V\#^)YO!4$$0/FW+^)(M-;2A9Q
MD$27WVDV4; B2==IK]YOC'\(+#]I3]N[_@G/\!?C3J-QXKUCP?\ LB^#?&O[
M16M:I+YFL^+X-(TW5_'5YH>OWJ!+F\DU";3!I]_*[&XFL]U+Q39^#X?
M&H?#R!%\,ZK::MX2F8:W+?:;-=FZFDFMYK:.TM([C
MD\\[S:>,QE:G@J6&6+QN%P=#!5*5"55U+KT:8'!U:.
M&J0^NXIUG"=",+U/+?VT?@G\-_@+\._V+?"WA[PS'I7Q7\;?L[V7Q=^-NO?V
MIK%W=ZWJ_CB^AE\+VES87M_<:;I2Z786NIQ1PZ59:>LT9C:Y6650Q\L_8E\,
M_#+QQ^UG\!_ ?QD\/0^*OAKX[\^"_%GAKQ?:R1EU=9O#.MV.MIM88P2;';GW/OGTLBHYAF7 E".
M(Q.,AF6DL-B:&2UJ& Q,IT>6,9O&2P=:M6E.#=
M6K6E4FI.6OHW[0GPX'P=^//QH^%:Q206_P /?B?XU\+Z?%,SN\>C:=KMX-"#
MRRLTDF=$?3V\Z5V>0,))'9F+'ZRU_P""'PJ^ W[!&A_%7XM>%DU[]HG]J37)
M+WX$:=?ZIK-@_P ,/@UX;$3ZO\2+G1K#4;&VO[_Q1YBIHZ:]9W]DL6L:'/!"
M#:WZR?;O[3/[(^E_M#_\%6[]+F1-(^"'COX9?#_]JGXK^*9&%OIVC_"JR\,6
ML/BV>>^4&.WD\0WOA]-&MYF)*?VI=7REA9R8_/W]H#XN7_[>G[9N@+HEH^G^
M!O%'CCP)\$_@UX5BB\BU\,?"BVU^T\.Z!:VUE'\EK+J%C<7OB348XU7RI[\V
MK#RK*)4\# Y[6X@H\-4Z>)K8>A@LFRSB;BG$TJTJ,TJ6%OA-@>YCLAP^08GB.=7#T:U?&9OF/#?"F&JT858-U,3%8K-
M(TIPE'DP.!Q.'PV!J.#7UW%QE2DJF"DEUG[:_P !OAQ\"?AO^P_X/\*^%;?2
MOC)\1O@;9_$SXPZ_+JVLSS:WJ_C2[TN#PM:7=E?7]SI>CP64AU:,C2K"Q#10
MI)/YI3+?0GQR\/?L5?L!>*]'_9_\6?LM7G[57Q@L/!7A3Q/\3?B-\0_B3XD\
M$^%8]2\5V+:@FF> ?#?AN&:-=,M8ED6*^FB,BNL<$]_?WD%X8>!_X*8:;XS_
M &A?^"@_Q+^&?P@\'>(O'MW\+O"7A7X;:'X5\)V$NIZA;Z+\/_#%G?\ B.ZA
MM+8@Q6>EZGKMXM]<(46V*+NVD+F7PA^WK\'_ (T^#/"OPI_X*#_ L?%^S\):
M5!X6\+_M#_#VY_X1[X[^%-&MMD,']JR1RV:^*ELHU_?^3J%N=0DC-Q?Z%J^H
MSSS7'ET89WC,EX6QE66:9Q0G@\3G7$.5Y7G-7!YY)\03CCLJK\T4UZ>,PV368U,!B/9Q]K[:=*6+A->>_M+_"K]F#Q?^SGX
M/_:S_9>AOOA.=1\>WGPQ^)_[.OBSQG:^*]4\*^(1:27EEXH\$7%[=-X@U+PM
M(#:)F_8(U67Q9XR.G^&
M]?\ B-??'SQE+&]YH_ARXU#Q+XTNO#VF_8(@ER=*OM2.E6=Q9PPM<1VL4D<:
MJ:^3/VTOV2O"_P"SG??"KX@?"CX@'XK? +X_>%+KQ?\ "+QO?6,6G^(XK&S>
MS.I>'/$<$4%M')>V$>I6,L5]#::>URLL]M?:98ZA8W,;_07_ 2-M;7PW\9/
MC?\ M :FJ+I?[./[-/Q+\="XE&$BUO5K,:7I2JS859Y;*UUJ.''SDL5&.^^9
M0PM3@+'9MA\ZSS'0PM+-*V25Y9CFV6X_#8C$S>!PN69C*ABZ&*QU?+L>WA5_
M:/M*O/'DJQJ>_*J96\1#CO!Y9B.;;3]2O\ 0+[4="M/$8T5I;F6.R.HVHOO[.:_G=X(
M_):Y)DW#$_X)]?LS?"+Q=HGB;]H;]JK2)=7^".E>,/!7P0\#>'/[3U70/^%B
M_&;XD^(]%T*".SO]*O-/U!]-\$V.IC4]02UO4C:2:1K@R)H]U%7P%\*_AUX[
M^/OQ0\'_ W\(VQU/Q]\4O%4.G6F]9&AAU'6[B6_U?6=192=FG:1;MJ&MZM.
M<".QL[ES\V*_=W]KW]E#XQZG#^SW\"OV9A6KX++\!"V(JX&MC<57^IXO,\6XX7#/#.Q\X>QHXVC@L+0
M^N87*L'S8K%/$J2YOJE&5EBW?\;_ -K;X7Z;\%/VGOCQ\*=#LVT_P]X(^)7B
M#2_#5D\UQ<&U\,W$D>J^';<7-U)-=7"P:-J%E"L]S--<2J@>>625F8_/:?>'
MX_R-?J+_ ,%A_"S:'^VKK7B81P+#\4/AE\-/'GF6LT5S:W&H'1I?"^JRVMS!
MF"Z@:Y\.*\5S [0SQR1RQGRW1F_+I/O#\?Y&ONN%]W<^,XDP$^>:+5F+[O0]3W;^@Q
M5:K,7W>IZGLW]#BN2M\?R0$F?8_F_P#A0.C\'[X[M_<'MG\_PH_$_D_^- Z/
MR?OCLW]P>^?S_"LC6&WS_1%E/NC_ .OZ^].IJ?='_P!?U]Z=0:+=>J_,ECZ/
M]!_.GTR/H_T'\Z?0;CTZ_A_45+42=?P_J*EH ])^$/C^7X4?%/X8?%"&&2X?
MX=^/_"GC%[>+)DGM=#UFVO+^",$$>;+8)?!OA.WOO%6E@7&G:5XGL-.6/09+Z: L+2#Q5X<:SCLIK@
MKC5]*OM/DVW+11/^,D?*#ZGLQ_D<5]S_ +,O[>'Q&_9Z\(ZU\(/$OA7PU\=/
MV=_$\=U%KOP9^(2M+IUI%?MYE^WA/57M[UM$^UREKB73KFSOM)%[_P 3"V@L
M;T_:3\EGN5YC'--G2JUJ%+$S]RAB:->
ME"OAY57"C*7-"I4IQ;9]?D6:Y?++WX5K:;H>K:O:Z[?:
M;8SW=GX9TI=I6-D@7=))/SZS;^!/VP/@XUVS2R>$/"&M_#WQWX9L)'.XV^D7WB>==8@LX>5
MACFGE^7@)&/EKG/B3\8_V8M%^"OB7X-_LT_"[XI:5J/Q!U_PIJ'Q$^+7QB\0
M:#J'BG6= \'7LVKZ5X0T;1/#D9T_1]'GUMK?4[I87MS+):H;D7DBP-;]#SC%
M5WAJ5+),VHUJU6E"L\70IT:&#HN&(E&HZ=-S<*>$>(G4FHJ/NRYEG
M/*,/1A7JU,XRJM3I4:LZ4<+B:U2OBJJC^XHT:,L-3G"4YN'/+%+#TZ<'*4I>
MZT_LA]_CO_@B+##H@%W=_!K]H-KOQ;;0_O)=/L'\77L_VVY0%GBMQ9^,]-NC
M*^Q/)\R0ML5L?C3'RX^I'5NN#V(KZT_93_:Y\4_LP:CXRTE_#&D?$_X/_%/3
M#H7Q7^$'B:5X-%\5ZA6H8B,Y4JLH2H5Z-16JQG3G$]+,I83.\'DV(HX_!X7&8'+,-E.-
MP>-G/#V>"E)1G34HUJ%2"O"<:D9'OW['OC;Q-\"OV*?VY
MOC7X:UJZ\)^(]HZI#8+?6]S9W4EEHNK2R
MW<,EO<((''-4TWXC:)%)/H_AW71X?L-&L-8D2??>^'(K^)CJ4,VK>'F=KN?2I(OS\^-
M_P"T.WQ,\-^"_A1X \$V/PB^ 7PTN+Z_\%_#33M3N=J*R:MX]^(?B
MBYC@N/%_CG5$9T>^>WM[#2X)9+73+98V9S\[VL]S:3VUY97-S8WUC=6]]87]
MG-);7MA?6(I9=7RS"X.I+&Y7E^ 6 QN&J.<,)FJQ5;%8C,Y1ISM4I*I]? GXD^*OA-\2](?1O&/A"_:TOHPK_8=3LI,R:7XA
MT2X< 7NA:Y9E+[3;M"._%+]MGX7_M1_"SPEX9_:K^$GBS5/C1X T_^RO#G[0?PEUK
MPYI7B'5-/5<"V\7>'/$5K_9^IP7:XN8Y;.D?M=_
ML]:9^Q7K'[&$OP_^-MSI>M^(I_&-S\3$U'P!#K$'B9_$=GX@MIX/#1G>QDT^
MV73[736MI=3$TUNLDWGI*RA.+,I9[F6$X?CC,DQ$%SBA/ 9CD&:8;
M/$QKT\51Q&,ITE1P6,IQP[IQJTY.4'7IREAYPIJHZG--GYRW.DZA:6FDWU]8
MW%O8Z[;3ZAHEU*FR'4[2PU&;3KB[LI VYTM-3M)[5I5V-'=6[["&0-7[@?LF
M?'?5/VPOV5?VE?V4/VAIQXPN/A7\&-6^*/PS^(NL,)_$>D6OA."<:='JFJ2A
MYKC4O"^JIIQTS6Y'%]=Z)>7FDZC+V=HT\MXNK/<
M-7X@P\<$LHQM#-<'FU*ME>:5:5&E0P,K4J
M+#U*=H2J1G)JU'):T<4LRPF(R_$Y=4IYA@*E:M\'!X)GL-<\*ZX?&\7A^_AT[5]*N+A-.?Q(TFIII]SJ\4^I
MM%<:4EM-I<[16-G:R17UI*KI/:2NC>.7F?L%_GD_V;J.3SU^P7&3^=?9>U=2
M.(3HU*7L^>*=114:BY7[U-QD[P\VETND[I?,J'LW0_>4ZCFJ"<:B
M<5RROVE)->B/PI\.?\BUX_#?QV^&]KI.MW&GV6I^&O&/@KQ$)&\
M-?$3P!XBBC@\2^"==\J.:2"VU)(+2\L=1C@N'TO6-.TV_-K>0P3V5S][^(?%
MO_!"+QMKT_Q8N/AU^WI\,KO4;N?6]8_9F\"W?PRO/A^FK32/>3:'X;^(&I:B
M=:T3PG/".VOK.YT^S(CTZVTJ%(+>V_%RD/3\1_,5\QFG"F!S+,/[5IXS
M-LIS&>&A@\5BLFQ]3 SQV$I2G.C0QD.6I2J^PE5JNA74(XJ@JDHT:].,I*7T
M>4\5X[+< LJJ8/*LUR^&*EC<-AZOHDGNK[5]%OAUX4@
MTR5;ZST?4=5TRT:ZUVZUJ[MXK6_N%%K-;F1ED+1I\&45KE_"N3956RNM@,//
M#O*,-F.&PD%6J5(/^U*^%Q.-Q->59U*^)QM>MA>>IBZU:=67MJZ;_>2;RS#B
MS.\THYE1QV(IUEFN)P&(Q=3V%*G.V5TL11P&&PZI1A2PN#P]/$SA##8>G3HJ
M-.A%12H4[?M'XU_:J_X)^?M<_ 7]E[2/VMH_VJOAE\:_V4O@_IOP*MT_9_T'
MX<>*?!OQ9\!>&WB?0)[:;QMJEBO@7Q)_P!*\96VM?#W
M5]9MXH=4L_%T^HZ=J'B[XH:UH#:UJGBNYUC1?#^DR^)_$>IQ:=71X!R6A6P-6.(S>5/*L;CLPRK!SS&J\#EV*S"&/CB98?#Q4>:+EF>,J4OK
M,\14PTZEL+5HP^^*OP'_ ."FG_!7J;XC?%#P'\4[
MS]ESQ=##8W?AS2)+3PWXE\#?##X<_"J/2;/Q1XXUO2M=_LSP3X(\'ZK877C'
MQIK%MKTB6NC1R65I)>:EJ,%K-^/7C&Y\,7GC#Q==^![*XTSP3<^*O$=QX+TZ
M\N;F^NK'P?-K-])X7M+N]O&^UW=W;Z&UA%G:E>V%KK^D"[MK_^R=;M[2>&+5M*^W65G?'3=06X
MLFO+.UNF@,UO"R5L'T'_ 'R/_BJ]?)7>W4L5-2<:&[TN:]T^.[EBTR\^T2G3/L%Y]+>%?VJ_P!AS]B_X7_M'Z/^Q9??
MM1?%_P"+W[3'PCU7X(:EXI^/NB>!/A_X!^&OP_\ $3XUB^B\,^$=2U:[\=^.
M?LTLL&EW5\MGI>G3;IX)+>UGOK+4?QE(.#P/R'_Q5*#99;0S)U/KM+"8FZ@XU'
M7KS@\11Q$J%2K.K0=.>K_0+]HK]JGX??$;]C#]AG]E+X::)XIT>/]FW3OB=X
MA^*M]X@LM,L['Q1\4/B1K=OJ#ZAX;:PU34)[_2]+LIM6M!>ZI;Z9=N+N-([,
M+YK4[]H/]JOX?_$']B3]B+]DWX=:'XHTR?\ 9_;XJ^,OBYJFNVFF6>E>)_B=
M\1=:BOHKOPRUAJE_=WVEZ7IEUK-B+S5;72[HB>)4M74LP_/K!]!_WR/_ (JC
M'^R/^^1_\56V'X6RK#1P$80K-9;G.99]AW.JY.>99LLR6+KXC1*K;^UL9]7C
M:$:%Z/(KT8M\%?B[-\3_ &BZD\,GFF2Y9P_B'"A!>SRO*EE:PM##.]Z/-_9>
M$>)DKNNXU>?^)(_1?XN?M?> M;_8G_8<_9>^%V@>*-.U_P#9Q\>?$7XQ?%C5
M/$UCIB>&_%7Q.\4^(5U3PY)X=CL]4O;O5-&T?3KK4=/O9=7M=+NGBDBA@A9&
M9T^J/VI?VHO^"8'[7WQ0N/VO/BQX6_;"M/CIXH\/>%8OB3^S?X2N_A]I_P )
MO%_B_P )Z#9:!:_8OC/J-U?^*?"G@75+/3;*#4VTGPW<>(%LHYI-,T[2M4N9
M9E_$#!]!^0_^*I1GCC]!_P#%5Q/@G*8U:&(PV)S; 8JA7SFL\7@OBJ=.5/V2HXC#1I48X?$4G2C([H<=YM[*O0Q6&RG'X6O
MA\FH+!XW TJV&P\\@P/]GY5BL/2]U0Q&'PU6NI\SJ4<1.M5EB*-71'[V?$S_
M (*:_LT_%#]O?]@W]J._^&WQ$\+_ ?_ &4OA/X-\.:Y\)M!T;PY/-HGC3P9
M%XLU'2]-^&L%QXGALM1\$Z=X@U/PTEA?ZY>Z/J[:9H0>?3UN2B'YK^'_ .WI
MX,\->!_^"I.L:YHWC6[^.O[>27.@^!M9LK?2I/#GA/P?XJ\?^(O$WCJV\5:E
M/JT.IP7MUHNM6VF6%OH^GZE;RRZ;"LLL$169?RQHHPW '#M#"T,+3IXWV.'P
MF78"FJF-JU)K#99G<^(MB:
MD\&J]?%9CC)RIX.C"/UC,\FAD.(FH*/+!4\O@UA(4U"&%K5*E6C&+DE&9/NC
M\>@QW/I7W_\ &+]JCX>^*_V!/V1_V/\ P!HWBW3=;^$?C[XL?%?XT:QK=KI=
MIH'B?QMXWO+J#PV?"K6.IWMY?VNBZ!J5_975UJ]GILRR+ EO$ZY9?@!/NC\?
MYFG5]#C,PF-E&C)_P *52K@J,)S
M2;E1=2GHIL_0'XL?M4?#[Q)_P3Z_97_8[\ Z/XML/$7PQ^)_Q4^+_P :=9UF
MVTJU\-^(?&'B^:]L_":^%Y;/5+S4=0M]'\.ZCYL]5O-1U :1H&HI!?2:KINFB">R@^S"Y#!T_/ZI &P/E7]/\:\V/"V
M4TWA(\M>:P>?8OB2//7F_:YKC98V=:K7:Y74IQEF%=T:5U&ERT/B5""/1K\7
MYQ4EBI2EAXO&\/8/ABI[.A"'L\JP"P*I4Z/Q>SJS^H4?;5H^_4Y\0KKV\S]G
M_B7^U1^P!^W+9_"SXD?MH6O[3_PE_:,^'7PY\+?#'X@:Y^SWHWP_\7^#/CSH
M/@J.6WT+71'XSU/3[_P#XQFM99[>]F:WO]*@^T[4:[BLK,0X_B#_ (*+?"?Q
MG^TW^Q3<'X1Z_P##+]A3]ASQ-HNI?##X'>&KBP\4^-[]=&N5UV\\9>+;W4=1
MTK2->\?^+O$FFZ//J[C4HK*PL1J++?ZGJFHW]_>_CQAO[J_I_C4E>=A^ $R:GFM"OAL9+*J3C>C6^KXBMA\-6Q$L5/!8>I
M.A@W0IM)>E5\1\^JMU/8952Q>(K9?7S7'4LNHPQF>3RRM0Q&%6;U=?;TO;X:
MC7Q%*C'#4\97IPKXN->HKGZI_"#_ (*11_#W]NK]I3]ICQ?X$OOB+\'?VL+C
MXO>#OC'\+YK^#3->UCX/?$[5WFL;#3=09Y+"T\4>%M,M=)2UB>X33KE8]1TM
M=0LUNH=3MO1?!7[1G_!-;]CI?%?Q,_8_\ ?'[X[_ +0FM:%KGA_X8ZC^UEX?
M^'=M\,/@';>(K62QN==;PQX>GU(_$WQ;I^FW$]A8S:I;+ILZ\R3V275X;G\:
M!U'UJ>,'G*KV[#W]Q6F,X%R/$.,8RS'"86I@\NR_'X#!8^MAL#FV#RF'L\%1
MS.E!^TK*%)NA.5"MAIU\/;#UYU*48Q6%#Q"S[#TY)PRW%8F..Q^98#,,=@*&
M*QV4XW,ZBJ8ZOEM6HN6E*K4_?4XUJ>(IX;$+V^&A2JMR?Z3_ +%_[7GPH^'/
MPT_:6_9?_:I\,>.O%/[._P"U9%X=UCQ3XF^%AT1/B;\-OB9X/U&35M!^(7AW
M3-;>ST+6X)KIK=M6T1Y;50VG68M8+BT>[LWZSXH?M4?LM_!_]F+X@?LG_L,>
M&_BEJT7QWU70[[]HC]HSX]:5X6TCQ[XL\.^&9VO- ^&W@'PCX;GU.S\(^%DN
MG>35;Z:]BU&\BN-4@,=PVJ?:K#\LQQVQ^&/Y9_G0?IG\,_S(K:OP9D];'U&PR5.-2&$J2PV'J5<+.
M=*#7-0XZSJAE=/+81R]U*&7XW*,+F\\)3J9U@\IS*I6K8[+L-CJCER4*\\5B
M_P!XZ3Q5.&*Q%.EB(PJRB_V3\6_M5?\ !/\ _:0_9T_91\-_M+^'OVI/#/Q9
M_9'^$O\ PI^P\&? ]/A[%\/OBUX>L)[*XTZ\?Q=XKFNK_P WFJO80MKFH6^
MA7M_8O<7GV"VU00:;+%B?M*_\% ?@C^T';_\$Z? UA\*M:^&'P5_9,>WNOB5
M\-?#-A9W.FHESXU\*:C?:%\.9-1\07-UXI6W\%>>F?Q/XON=#U3Q/K^IZC
MK>I6MK)?SA?R&Y]/T'_Q5)@^@_(?_%5P4. ,CH8FAB/;9M5C@L3F>*R[#5!?&6LZGX']0U/7?B1;>$&O)(O#UI+%<":&WMWGFMM46'7;/\8Z*ZJG >23IX>C0K9M
M@:%+*,!D.)H9?F-;!T\TRC+83A@\'F:HJ,ZL:<:V(A*K0EA<1.GB<11G5=&H
MZ9S4?$?/Z=7$UZ]+*-O$WB/QEXADM9]?\6Z]K'B76YK*PL]*LY=
M5US4+G4]0:STO3HH-/TVS^U7,BVFGV,$-I96JPVMM%'#$B#[5_8$_:Y\*_LK
M>.OB?IGQ5\#ZM\1_@'^T3\)_$'P0^.'A/PY?6FF>+1X2U[+VOB/P?>7\D6G_
M /"0>'[F2Y:&TOI[6"\M;Z[5;NWNHK:2O@M/NTZO>S')L!FN78C)L92;P%>C
M3I2IT)O#SIJE*G4H3P]6G:5"KAZM*E5H5(>]3J4H26J/E,JSO,G4J4J]*:Y:E.%?V%-'^.7Q&^-?Q\\):A\,]=^/7[1N@^"?#B_";X5:RR-KGAWX<^$?
M#-SJBZAXNUN-(HK[Q7?_ &*.&>&TO[1F73X--E\#_:$_:B^'_P 0OV-/V(?V
M5?AQH7BC2!^SQ9?$[Q+\4M0URTTNSTWQ+\3OB'K,%^+WPY]AU74+F\TK3+*?
M5[1;O4[?3+EEN8%2SQYC#X&HKQ(Q6
M)QD(<52H^PPF!Q->EAL/1I4:5*8<>9SBL+BLOITDZA
MX=\%Z;X?NK?5+G5+^33]'GL6OSJ.F:=%;7-F5M'N$*E?I[6OVK?V%OVPOAO\
M!;?]N2P_:2^'OQP_9[^'.B_"%OB%^SWIO@7Q-H_QJ^&_AAY#X9MO$&G^+[JT
MF\*^+]-BDDC;4X8+JR:YOM1N?/EM[BVL-+_&P=!]!_*EJJO!>4U(QG0K9E@,
M;#-,TSFCF> QGU?,*&-SIR_M%4ZWLITY86O1<*"PU:C5A3A2I3@U6ITZD)P_
M'V=4I2AB*.69C@:F3Y7DE;*LPP2KY=5P>3*']G2E14X5(8G#U(.K'$4:U*I*
M=6O&3=.O6IS^]_VJOVQ_#?Q1\6? _2?V;OA/I7P#^"7[+4&FP_ OPUZCJ%[-
M]H_$?]JS_@ES^T3\;+3]M'XX?#;]J*'XW7UOX7USXF_LS^#T\!S_ 3^)7Q(
M\*:98:;::DGQ%U/4H?$>C^"=<_LG3I->TIM,34+FWMVAD@8W-\EY^&]/3K^'
M]145N!9T,;C*GMJV)>.Q5
M-8FKB)26+IXA^VP]>C4NWI1\1,\I5LRKXG#Y/F5/,L7@,?/!9AE>&K9?A<=E
M=&IALMQ.!P=.-&CAOJ.%K3PM&A"/U6>':HXBA6A&*7Z_^ O^"FJ/\6_V^/VE
M/B?XW>H:3?VFE^'O
M#VCZ!!;:MIVGW^IZQJ::IJ-U;6LMT OAG[)'[5'PX^&'[.?[7_[*WQQT/Q?K
M_P +/VB/ ^AWW@6;P?8Z/J>I> ?CKX)G:?P;XR>UUG5='CBTIS'I@URXL+B6
M^,&AV=O':S1S.4_/C-(RAU9&SM=64[3M."I'##D'GJ.1VYJI<#\.K#8O#4L-
M6PU+%3R&:^K8B=*>#?#<<-#)U@:DE.5!83ZM"K%2]ISUYUIS;A4]G'E7B'Q+
M+%8/%5L31K5<+3SZE4=;#QJ0QT>)JF)GG,L?2YHPQ,\5'$RI;4U"E2H1BE.D
MJC_HQ\=^-?V;_@5_P3?_ &!OV/OVIO!WCR?P5^T-X"\9_M2^*/&GPCGTI?BC
M\*/&^L^,?MWPV\6Z+H7B-HM"\5Z-JF@>(->\-^(]"U66VN+G3+>&?3IQ'GT#PO+/I'A3X?>&I;6UN[;2A=2W5_=6T/F6MJ)-4N=:^._CK^T9\6
M?VD-7\"ZU\6==L-9NOAK\-O#7PC\$P:3H&E>&].T/P%X1%S_ &%I$.G:/!;V
MTDT!O+@W%_*CWEY^Z^T2OY4>WPZO%X1O.L1F6*QF&Q<<(H4Y5*F"HU:%*@G46%4Z,,1]5^L0A67O<2^)-7%U*V#R
M##83#9?#(\!PU@LRJX"E3SV&1X7+<)@<3@/K<6YT:./JT*]?$.,IXCV>+JX9
M8E4)U*4OU^O?VI?V,_VM?@W\ ?!W[;$?[07PY^,7[-7P_L_A#X>^+OP$T/P7
MXVTKXH?"O1Y/,\.Z/XN\,>+M2TB70?%6AHOE6NJV+WNGS3W%_>73-#?+I]AQ
MOQQ_;8^$'CU/V8_V=?A1\,O%7PM_85_9S^(^B^-KOPGJ%YI?B'XN_%K6)-=M
M;OQM\1?B!=V]Q9>'[GQCJVCOK=GHFCV]]_9]E+KEZ9]12V:QLM(_+J'.&XSR
M.P/\R*FQ_LC_ +Y'_P 57KT>!,DH5JVG6A@'7J?48X=.R\+$^)/$%>E4@Z654\7BL/@,)FF:T
M2YC]K]"_P""F/PO
M@_X*0_M,_MV:UX8^(,]OXP^%7C_P5^SSHEKI^AOK?AO7M5\$^'/ G@FZ\71R
MZ_'8:1IVG:5I&JRZBVB7NL3VLFJM';0W(WD_$'_!/?X^_#7]E_\ :U^%7[07
MQFT;Q9XJ\/?#-?%6O1:7X0M=-O=9U'QE?>%=6T;PY/LUC4M)LUL;?5=5;4K^
MYDN_M$:VZ/!#+(<+\8<^GZ#_ .*I,'T'_?(_^*K6EP3D=# 9CEU&&*IX?-,B
MP'#F(<<5/VL,IR[!5\#AZ&'J6O1G['$XB4ZJO*5:K*K92.6OX@\0XG,LKS2O
M/"5<3E/$69<4X:-3"PE1GG.:XS#8[%XC$4OAK1E7P>&C"E+W84:-.E'W8H_5
M/]B?]L7X%_#[4?VXM%_:@TWXMIHW[:G@C5?"VL?$+X-P>'-2\;>#6UWQ-XA\
M2^(H;?3/$]Y9:?>1Z]-KZJ;HO>+#)I:6]UI]Q;79F@_-/Q,OAH>)O$2^##K1
M\&C7M67PB?$ALSXC;PP+^== ?Q!_9RIIPUR32A:OJRV"BQ34&N$M,VZQL>;C
M^\. .O8#M[$U8KOPF18'+LTS7,\)+$TZN;PP$<5AG63F?$V8YOE64Y5C:>#G#)JF/GA,9##\F/G',<5
M4QV(HXFNI\M>E'%UJU:DI4E.#JRBYR@HJ/WAI_[3W@?P_P#\$WO$O[(7AG1_
M$T/Q6^)7[2FG?%SXA>);JVT^/P=/X&\+:!#IOA7P]8WD6I2:Q+M=BM-*U+5K5=/U:YBTS[+/+,JRIRBXR4,'1IX7
M"1LE0H&C@JV795E,X0P\(U/J&2XK$8[!T(U8N,
MUSXW$U,5C)-REBJRA*7)R(_1KXO?M6?"#XC_ H_X)T? G2]#\'4;>XTRPUBRTF?69M'FGDU%(
M[J&WCWR5Y?\ MZ?M':)^U_\ M:?%SX[Z19:[HO@CQKJ/AW3/"VEZS;6L>OZ/
MX&\->'-&\-6-KWMA#?B"PO;Q;2VU">V26Z5!=XWNOQS'G>N/?\ D?>YK3SG,ZTDTN:M7QU
M*G)5+Q<*4(TM8I-=&8<89UF^%Q.!QE3#O#8JOD6)JTZ6'A2][AO)I9%E%*+C
MMA\-@:DU[%IQ=:4JR<9MM_M)K7_!0[]G_P 0?\%+O@/^U=?^#OB?V=OA
MKX+\$> /!2Z3X=;QVEQX"\":[I&ARRZ:?$0T*UM!XPUU]4>2+6WE33[&VF6(
MW'[E?/OV3_\ @H%\//@+:_MG^*/B+\-=;^*OC[]KKQKI%AXDT22:TT_P[=?!
M3Q)XG\6ZK\8=#N/$;7XU32?&&JZ5XNN(/"<]KI5_I]OJFG:?=:A*D*87\F\'
MT'Y#_P"*JW7DSX X=E@_[.J4\;/!_P!FY/E,H+&UZ=1X'),?/-,)35>DZ=2$
MJF-JSK8NM%JMB)6O4@U<]ZCXC\31QTLQA5P,,4LUSG.8OZC0G1CC\]RRCD^+
MFJ%53INC1P%%4<'0:<:$933<^>5_US\)?M3?L4?L;>'_ !YXI_83T+]H7QE^
MT5\1O".L>!?#OQ/_ &B;3P5H^G?L]>$?$J)#KG_"':5X0N+@^+?'"8S+;&_TS5/G/7/VF/!?_#NGPI^QSX6T[Q5;_$#4_P!H[7_CC\6_
M%&I6M@OAW5[--#O-"\&:?I.HQZI-9>T._\ U^S?$XK,,7C,%D>-AFE/*8YA@*^6IY=BL5DE
M&='+LQE0I5J=6ECZ-*M6I2J8?$4:56G5G2JT949.D?JUJ/\ P4%\ >.?VN?V
M2/'_ (A^&.K?#O\ 9"_8XU#2;;X1_ KP9]@\1^(-+T+P]$U_9ZIJDVHW^DZ5
MK7C'Q'XBT_PY=>(+MKR&WMK'3A#!(;_5;.UG*LZ^=:6-Q:VDFQV0- 0C,F#7EV/\
M9'_?(_\ BJGB^Z>,<^F.P]":]3*^&*MY^9<49SG5*KA\QQ,:T*^:3S>
MJXT*-&4L2\#ALMI4TJ,*=.G@\)@L)0P^"PM*G"EA:<7"FK2=YE4-E6 92.58
M @\CJ#D'UI?(A_YXQ?\ ?M/\*$Z_A_45+7K5F^?Y1Z=KVOWMS2M?:[[GDT8J
M4]5>T7;RNX7^_EC?T79$JQQH'M4LO#9M[;5-2TJR:"UUF]M;Z
MY>6]C:..UW0I)(%4?)]30?Q\9Y';/;W(Z_TKSRK>PQ-&="JJ53EG[.;IU))2Y6TW=*^IZ^68VME.88',\)&E]9R[&87'8
M=5J?M:/ML)7IUZ7M*?-%SASTHWASQ36CNKI^D?%CQ[??%7XJ_$WXH:CYJWWQ
M&^(7C+QO,DYWS0KXF\0:AJUO;3,K,ADM+6Z@M6V,R?N<1ED537)Z)_9(US0S
MX@%T_AY-3Q!'80QS7\FA1ZE:R:S'8Q330PRWLFFK=):1RS0Q/<-&LDL:%
MG7*Q_LC_ +Y'_P 53T!W#@?D/0_[5%.A3H86CA:*=.C2HPP]-1=G"E1IQHTU
M%])0IQ48NVEM%;0EXBK6Q-3%59>TK5:U3$U925^>M5JNM4;OKRRJ2D^6^B=K
M]3]T?VG/VJ?^"4_[5_Q3_P"%M?$[P9^W%#K=MX6\-^"M.T;PE?\ PPT'PY8^
M&O"<-S#I-C:6-QJ6J7-M)(+NXEO)QJ!\RXF:1!$H"#YN^)O[66GZC<^;9W%Y964&E1:CJ+7GYCU*G3\?Z"OC\#P'DV"IX##O$YSCL#E<
MZ%3 Y=F6;8G%9=1GA6I86I+!7IX>M4P\HQG2=>G5A"HO:QIJJH5(?;X[Q!SS
M'5,?B%A\ER_'9I#$4\PS/*\HPF%S/$PQ:<<5#Z].->O0AB(-PJK#2H2G3_=R
MDZ'/VA[FY^)'Q@B^)OB_5O@Q%X,T];
MJQ\.1Z##X+T1-7\1:KYTEM:_V(MYJ=HVCJAGD1+>ZVJ[-PGC/4O^"3)\'>*Q
M\/?"W[%E\5M93+H,GB,V$,^5/VU24VFSV/\ 9\\5^"_ /QN^#GCSXE6&KZOX#\#_
M !'\)>,/%FE^'K6UN]:U/3/#&KVVM&QTRUO[NRLY9[N\L;6'9'-/OE@
MGN($N[+0K'3[>YC@GF@CG218I9$ =O HON#@'D]@?YD5)C_9'_?(_P#BJ]1Y
M?AI9HLW?/];CE[RR*]HO8PPTL73QDW"D[J-:=6E1]K43]^.'I148QA%+QEFF
M)CE,LEC[-8*>8QS2:5.*K3Q<<++"0K*[S$TB!)
M(F"RQRK+$S)O42Q,DD9>-N)$WJN^,\.N5/#5^MG[0?[1O[ /[67B;0_VAOC-
MHG[4N@_&M? OA/PKX_\ @_\ #J/P)_PKWQOJ7@S3_P"SK&;1?B3K5Q)JW@O0
MM2BW1WKVVB3ZI:VS&2UT\:F9KVX_)*/[G3'S'MCL/<_Y_1]&E)1G!TJWU:C+F]G[:G.C3G0JTI)M^
MCDN>8K*\)C\'##X#&X/,*F%J5\)F6$CB\.L3@'5EA,5"#E3E"M16)Q$-)^SJ
MTZLH5Z=2%DOUD^-G[.?A=H]EI=
M]:Z=+X7\1Z?XA32?AO-=ZVIUS2&&D:/ID>H^(7T74;F"Q%W=PF>9UKXT^,WQ
MDT#XY?M:>.OCKXKL]:C\%>/OC8/&NIZ3'#:S>((?A_'XALC%HR6[70LGU>+P
M?80:8MO]M%G]L79]J$'[ROFJ/O\ A_6ID^\/Q_D:X\MX5RG*?9?4EB:;H8#$
M9;1E/%U*TZ>%Q>85,SQ'+.JYREB*F)K3;Q%7VLVJ=%R4IT^:7J9AQ5FV;SJO
M&RPTU6S'"YK6A3PM*A3JXO!9;3RK#J5.BJ<8X>GA:45&A25*$7.LHVC5<8_J
M'XE_X*&^1_P4DNOVW_!OA74K[P;!?V'A[3_ >O/9Z9K-[\*(O!%KX&U'P],]
MK/J6GZ9JTUG]NU+3RL]Y90:A]E^T220O.1Z5\(OVAO\ @FE^S?\ M'>&?C[\
M,? 7[2GQ D_X235O$$'AWQ]IW@NRT/X)QZI::A+<'P58V.L+>^/O%,%[=KI'
MAW4-?UBSTGPOH\UUJ:R:OK,%@$_'BK,6=G3N>P_^*'\JX<1P+D=?#0PD*F98
M3#_V+0X?Q%/!YA.@L=E>%A.&%HXWEBG5G0]OB)1K4W2J3=:K"JZE"K.A/TZ/
M'.=TL5+&SIY9BL5_;=?B'#U<9@*>(^I9KBITI8JOA(U)5'\2Z]?ZP
ML$C!BI:WBNX[?Y25'E85B.3Q3IYB/&V"LB,A&.H=2I'4]CZ'Z'I2_BW_ 'S_
M /8T'IU;_OG_ .QKZZC3IX:C1HT4H4\/3A2I1O%\M.E&,:<=OLQBDVK(J3JU9-QO*I5?-4DVV_BFY/6_*GRZI)GZ]_&K_@I#X9\
M=?L8>"?@5X%\)^)-%^-NL?"WX;?!7X[?%#4H--@M=9^&7PVBU":#PKX6U"TU
M.ZU2YL_$.JW>=3^VV.F[=-N]7MG\\W<9B^*OV,_BQ\//@1^TY\)/C)\4=*\1
MZUX/^&^MWWB:?2_"EE87^M7>L6^BZG:^'Q;VVIZAIED8[?6+NUO;B26\B:..
MV+Q+)( *^8$^XOT],?I4\??\/ZU\G@^$\ER_*5.%&@Z='"PA:%"G32C'WI,^BQW%FL_!WPC#
M\/M7\-0W]]-)=WVF>'/%NHW5Q=Z3H\US(Z6P$T9L(9,6=O:0QQ6\'Y>5*G3\
M?Z"C%<,8+$5Z&*P^*S/*\31P5#+95\JQL\+/$8'"J7U;#XF#C5HU?8.(5.FJU*52IAZO)"52C.<*!? ,'PG^!'P*\)
M-X%^#?PY%^=6U/3=(F>V;4]<\3:M]R^\0ZTUA8_:$@:6WM(K;!NM0O+F\OKC
M9^!_[2'@KX1?LC?M:?!U-)\2S_%C]HUO!7AK2]?L[;3_ /A&=&\!:!=1W.MV
MFI7TFHQZG'?Z@E]KD4%M:Z9<6\IFMC-<1AF"?$=68L[.G<]A_P#%#^5;/AW*
MXY7A(I.4^>*ACB,SQ.+P.7Y?4]FL%ET<0L-0A3C&//BZCJ5
M\15=N:MBJGN4G7F[JC1H4X1CR2<_MC]K#]H[P=^T/X)_90M]+TOQ)8^/?@M\
M#++X1_$6_P!9M;"'2M;N-$EL&T2]\.W%KJ-Y=7=HD::EYS7]KI\J&>)5A?+-
M7QFGWA^/\C3:@ZGL/_
M (H?RKDK?'\D!-SZC\O_ *](N?GY'WU[?[ ]Z3'^R/\ OD?_ !5 '#\#[X[#
M^X/]K^IK+7^E_P 'U_I:ZPV^?Z(LI]T?C_.G4U/NC_/>G4&BW7JOS)8^C_0?
MSI],CZ/]!_.GT&X].OX?U%2U$G7\/ZBI: +46=G&.I[?_7%2\^H_+_Z]0Q?<
M' /)[ _S(J3'^R/^^1_\51K_ $O^#Z_UO=/XX^H[GU'Y?_7HY]1^7_UZ;C_9
M'_?(_P#BJ,?[(_[Y'_Q593O=>G;S]?7^EKVCN?4?E_\ 7IR AATQST&.WUJ/
M'^R/^^1_\53XQ\XX'?L/0_[1_E50O;Y]O)>?K_2U"Q4J=/Q_H*BJ5.GX_P!!
M61T#Z***#2FDV[KIUMW79^%'AO_D6O#G_8O:/_ .D-O6O7<:'\#/CS;:#H-M<_L_\ [0,%
MQ;Z'I,,T,OP*^+2R12I90!DD3_A#B588(P?KTP:U1\$?CEW^ _Q\'_="OBWT
M_P#"-%?8X'BOA>.!P,9<2\/1DL'A4T\\RI--4:<6FOK>C33371I]F?D>.X4X
MHGCL=*'#>?SB\9B;2ADV92BTZTFFG'#-.Z::[K4\RJ2/O^']:]*_X4C\N/\ H3/\_7BN
MK_6WA7_HIN'?_#YE7E_U%^?X/L<+&K+A?B/6W_,CS3NO^H7S/-Z0]/Q'
M\Q7I?_"E/CC_ -$'^/?_ (8OXN>F?^A,_P _3F@_!3XX9P?@1\>QSS_Q8OXM
M]CG_ *$S/-./%?"\K\O$O#SLKNV>95HN[_VO97U(7!_%MU_QB_$6Z_YD>9^7
M_4+Y_P!:'GM%>C_\*8^-_P#T0CX]_P#AB_BY_P#,91_PICXW_P#1"/CW_P"&
M+^+G_P QE'^MG"W_ $4O#W_A\RG_ .;/-?>+_4_BW_HE^(__ QYIY?]0OFC
MSBBO21\%?C>1G_A1'Q[_ /#%_%O_ .8T4[_A2OQO_P"B$_'S_P ,7\6__F-_
M'_Z_%+_6SA;_ **7A[_P^95Y?]1?FB?]4N*_^B8XB_\ #'FGE_U"?U]U_-1U
M'U'\ZNX']W_T'_&N^7X*?&_/_)"?CV,8//P+^+8_#_D335C_ (4S\;_^B%?'
MK_PQOQ:_^8JG'BSA5RBO]9N'5KUSW*DNC_Z"_P"OO%_JGQ5_T3'$7_ACS7R[
M83^K/R/."!@_+V/]W_&G$#^[GWX_J:]$/P8^-^#_ ,6*^/7_ (8WXM?_ #%#
M^=._X4S\;LG/P)^/8_[H9\6O&.(MNN1YJNJ?_0(OZ^1YQ@?W?\ T'_&C _N_P#H/^->C_\
M"F/C=_T0KX]_^&,^+7_S%TH^#'QM/7X&?'L?]T+^+1_]TNHCQ5PO)77$W#O_
M (?_\ PQ7Q;_\ F+I1\%_C9GGX&_'O'_9"_BW_
M /,71_K5PM_T4W#O_A\RO_YJ%_JAQ9_T2_$7_ACS7^[_ -0GI]_EIY_17HG_
M IOXUC_ )H5\>O_ QGQ<_^8RC_ (4W\:_^B%?'K_PQ?Q<_^8RM*?%?"SC?
M_6;AS5O3^WE_X4[\:O^B%_'G_ ,,9\7/?_J3/
M:L_]:^%E))\3<.WNO^9[E/6W_481_JAQ;_T2W$G_ (8
MI_+_ .O7H?\ PIWXTY&?@9\>0#U/_"B_BX<<9_Z$SUXI_P#PIWXS_P#1#OCS
M_P"&)^+7_P QM$^+.%>9_P#&3<.]/^9[E3[=5BVNOX,B?!_%KM;A;B3_ ,,.
M;=;?]09YSQZG\O\ Z]3UW_\ PIWXS_\ 1#OCS_X8CXM?_,;3_P#A3_QG_P"B
M&_'K_P ,5\7/_F,_'_Z]72XKX6=[<3<.]-\]RI;^N+\_T[7S_P!3N+O^B5XE
M_P###FO_ ,R'GM3Q 9;J>G8?U)KN_P#A4'QG'/\ PH[X\_\ ABOBY_7P9C_/
MI4T?PA^,XR?^%'_'89['X&_%E2/P_P"$,)]^G]*=3BKA>R?^LW#MEN_[=RE[
MV[8SS#_4[B_IPKQ+_P"&+->Z_P"H-^?_ YPN!_=_1?\:,?[)'X+_7-=]_PJ
M/XS?]$0^.W_AC_BQZ$_]"9WX_$]!TI1\(OC.3Q\#_COCU_X4?\6/4_\ 4E^U
M5_K3PNX)+B;AR3:C9+/,/^B5XEZ?\ ,BS;^[_U!??Z
MONCS_'L?R2C'L?R2O0O^%0_&;_HB'QX_\,=\6?\ YB_\].M)_P *B^,P(!^"
M/QV .;>7_4%Y?GW1P-%>@_\*A^,?\ T1/XZ_\ ACOBU_\ ,;1_PJ+XQ_\ 1$OC
MK_X8[XM?_,;6G^M7"VW^L_#?_A^RG_YL_JWI?C_U-XP_Z)7B7_PPYQY?]0GE
M_5E;A$^Z/QIU=ZOPD^,0&/\ A2?QU[_\T.^+7]/!IIW_ J3XQ?]$3^.O_AC
MOBW_ /,9Z?X>])<4\+\S?^L_#EFO^A]E/E_U&"_U,XP_Z)/B;_PP9MY?]0GF
MC@**[\?"3XQ?]$3^.O\ X8[XMX_]0RG?\*C^,/\ T17XZ?\ ACOBWZX_Z$S_
M #UZ5$N*>&'+_DI>'7>VV>Y4^B[8OS_!F<^"^,7;_C$^)O\ PP9MUM_U!G"C
MH/H/Y4M=X/A-\80/^2*?'3C_ *H=\6_3_L3/\_7B@_"?XP]O@G\=#_W0[XMC
MOC_H3/\ /7I6BXJX6LO^,FX<3T5GGV4WZ=/KGG\R/]2^,?\ HD^)O_#!FOE_
MU"?UKV.#IZ=?P_J*[G_A4_QA_P"B)?'7_P ,?\6O_F,_STZT^/X4?& 'YO@G
M\=0.?^:'_%K_ .8S'^?\ 6EU+@KC)JRX2
MXFN[?\R#-O+_ *A/ZU.(HKO?^%4_%W_HBGQU_P##'_%O_P"8KU_Q]J0_"GXO
M=O@I\=/Q^!_Q;_\ F+I2XIX7<7;B;AQ[;9]E+_E[8SS_ #,O]2.-/^B1XG_\
M,&:^7_4)YHX.BN[_ .%4_%_K_P *5^.N/^R%_%W^?_"&8_SCK1_PJKXO?]$5
M^.O_ (8SXN?_ #&5,.*N%TK/B;AQ:]<^RE=O^HS^K$_ZE\9?]$EQ/_X8,U\O
M^H3^M>QQD/1OESR/3^IJ; _N_P#H/^-=G%\*_BZH.?@M\=!S_P!$-^+?IZ'P
M63^52_\ "K?BY_T1?XZ?^&-^+G_S$U?^M7"W_13\.?\ A]RKR[8OS1E+@GC-
MNZX1XGU_ZI_-O)?] ?FGP6^.A_P"Z
M'?%O^O@H4[_A5?Q>_P"B+?'7_P ,=\6O_F*_'_Z_%'^M7"W_ $4_#G_A]RK_
M .:_-?>+_4CC/_HD>*/_ !'\W\O^H/S1Q"?>'RXZ^GI[&IZ[)?A5\7N"/@M\
M=">?E/P.^+?YY'@O_.:G7X5_%X]?@M\).
M'I;?#GN4OM_U&>:%_J3QG_T27$__ (C^;^7_ %!^?X,XV/HOU_K5FNM3X6_%
MQ=H/P8^.'!Y_XL?\7/K_ -"9_GZ\58_X5A\6O^B,_''_ ,,?\7/_ )B_3_'V
MI?ZS<-_]%#D/_AZRO_YK_KY,VI\(<6QC9\*\2IW_ .B?S?K_ -R7S..C^^O&
M>O'X'UJU@?W?_0?\:ZF+X9?%<-G_ (4W\;\J<8_X4C\6QU!'?P9_^KIUJP?A
MK\5\_P1^+0]/^I+J?\ X5O\5/\ HC?QP/T^"'Q:/Z#P942XIX809PD]?\ L"_K\3E4SM'([]O<^]/&
M?%%1@_!SXX CL?@A\6_P#YBZ^-_4?\ -$/B
MW^?_ ")1X'>C_6?AK_HHL@^6=Y6W]RQ;?X7-O]5.*?\ HF>).G_,ASC^[_U
M_P!:^=L& 'YL\].P]_4_RJQ@?W?_ $'_ !KH$\ _$Y5WZ?]
M1?GJ;TN$^*G%_P#&,<2/7_H09P_LQ_Z@E_PYR^!_=_\ 0?\ &IH_NGC'/MZ#
MTKH_^$ ^)W_1(?C?_P"&0^+7_P Q53)X!^)2C!^$7QN/.<_\*0^+@[#CCP1V
MH?$_#:WXAR%+N\YRQ+6UM7BDNO?N;0X3XJ4M>&.)%YO(,W2Z=7@]-_Z1SJ=?
MP_J*EKHE\!_$H,?\ ,DG\NOTYQ)_P@OQ([_"/XVX_
M[(A\7?7'7_A"?\]>E<]3B/AV4KQS_(Y*RUCF^7M?>L3YG32X6XGC)N7#?$*7
M+;_D19MW7_4&8-30?\M.,\KZ>GN:W_\ A!OB-_T27XU?^&3^+?\ \Q7^?SJ2
M/P3\14)_XM+\:QN(R?\ A27Q;XXQ_P!"3^/05B^).'4[//LE3[?VME]]D^F(
MWU.E<,<3/;AOB+O_ ,B+-O+_ *@_/^M#$P/[O_H/^-.0#XYQX)Q_+U.*R
M?$?#W+'_ (7LE3UT_M7 +K_V$6TZZEPX7XFN[\-\0K3KD>:]U_U"?U9F+4J=
M/Q_H*VO^$-^(/_1*?C1_X9+XN>F?^A*]*D3P?\0 "&^%7QG!R?\ FB?Q*E_P"$1\>_]$M^,W_AD_BZ/_=(
M_P#UU,N(^'7%I<09&WI9?VOE_=?]1']?(UAPOQ,KO_5SB"SM9_V'FO\ \Q^:
M,N(90<'J>R_UYJ3'L?R2M:/PIX] V_\ "K/C-W/'P4^+G3\?!!_*IO\ A$_'
MG_1+_C.?^Z)_%S^O@@5C_;^0_P#0^R3_ ,/.7Z_#T]O=_=U?OR,N/[G3^(^GH/3_/XYI]:R>%O'04 _"_XR]3U^"GQ;&#T
MY_XHKCIW_2G_ /"+>.!U^&'QC_\ #+?%O^G@DT+/\AZ9]DCM:_\ PL9>^VC_
M '^[[>;-Z/#?$48M/A_/5[V_]C9GRZI+67U7E3OT;3?2YE1]_P /ZU,GWA^/
M\C6G'X6\3_S1;XM__,.?YU/^LO#O_0_R7_P[X#R_Z??UJ:KASB&Z_P"$
M#.^^F4YATM_U#>91S[']/\:/P/Z?XUI#P[XV/7X9?&0?]T5^+7_S$5(OAKQH
M>OPU^,2_7X*_%L_EM\$'IWS^'-)\2\.V_P"1_DNJ_P"AO@/+_I]]_P SK60\
M0.ULCSQO3191F+?V==,+_6FVEJ"?='^>_MQ4T??\/ZU?7PYXS _X5K\8#UY
M_P"%+?%P>O.#X)'\JE3PYXS'_--?C!R <#X+?%PXZ]?^*)P/SKF_U@X?_P"A
M]DG_ (=LO_\ F@M<.<1?]"#.^F^4Y@NW1X5/OT_X&?4J=/Q_H*O_ /".^,_^
MB:_&'_PROQ<_^8G_ #TZU(GA_P 8 $'X;_&!3GC/P5^+N".!U_X0G%'^L'#_
M /T/LD_\.V7_ /S0:QX>X@44GD.=WZ_\).8?_,QGU:B V?=SR>>/ZFK']@>+
M_P#HG'Q>_P##+?%W_P"8BIX]#\6JN#\.OB^.>WP5^+A'./\ J1S^/IS4RX@R
M!JRS[)-?^IME_=?]1!HN'\_Y9?\ "#G>MO\ F4YAT?\ V#^94P/[O_H/^-*H
M&X?+W']WU^M7/[&\6#_FG?Q?_P##*_%P?S\#THT?Q7D?\6[^+_4=?@M\6P.O
M<_\ "#UG_;V0_P#0]R3_ ,.V7_\ S3Y_U9FT>'N(&E_P@YWLO^93F'E_U#^:
M(Z__%E_BYQ]
M3_PA&/UI//\ ($KO/X@4TWD6=)*][Y5CT]8]GA[]?
MS(:G'0?0?RJ3^R?%'_1/?BW_ .&8^+9_EX))J<:5XFP,_#_XM_\ AE_BY_\
M,34?ZQ\/_P#0]R7_ ,.N ZVZ_6+=?ZTOT_V#GO\ T),Y_P##7C__ )G_ *^1
M5JU$!L^[GD\\?U-+_97B7_HG_P 6_P#PR_Q<_P#F)J:/3?$H7!\ ?%L).W@+XMCZ_!?XMG^7@CK0--\2