Exhibit 99.1

For Further Information:
Simulations Plus, Inc.
42505 10th Street West
Lancaster, CA 93534-7059
| CONTACT: | |
| Simulations Plus Investor Relations | Hayden IR |
| Ms. Renee Bouche | Mr. Cameron Donahue |
| 661-723-7723 | 651-653-1854 |
| renee@simulations-plus.com | cameron@haydenir.com |
For Immediate Release:
April 13, 2016
Simulations Plus Reports Second Quarter FY2016 Financial Results
Record second quarter as revenues grow 12.9%, net income up 18%
LANCASTER, CA, April 13, 2016 – Simulations Plus, Inc. (NASDAQ: SLP), the premier provider of simulation and modeling software and consulting services for all phases of pharmaceutical discovery and development from the earliest discovery through all phases of clinical trials, today reported financial results for its second quarter of fiscal year 2016, the period ended February 29, 2016 (2QFY16).
2QFY16 highlights compared with 2QFY15:
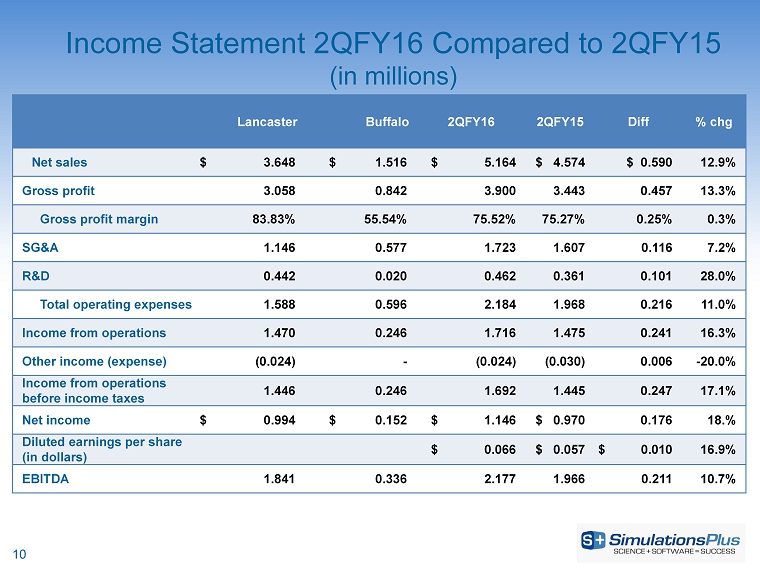
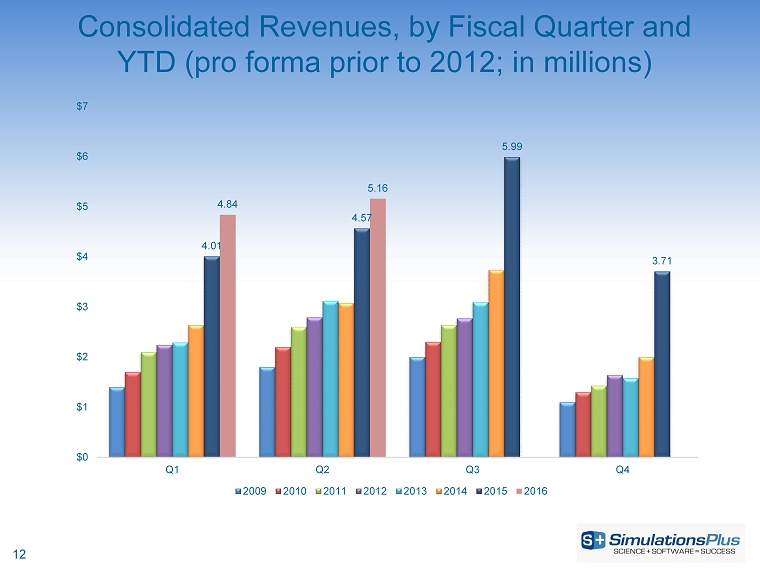
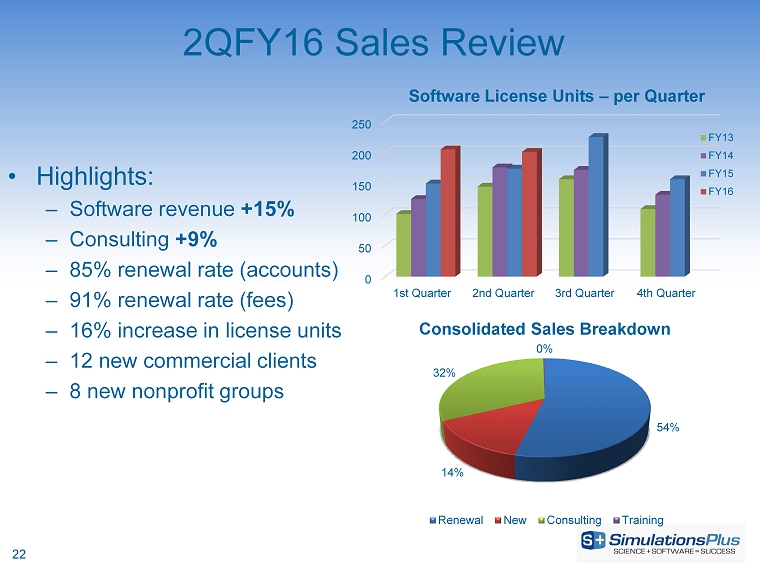
| · | Net revenues increased 12.9%, or $590,000, to $5.16 million from $4.57 million |
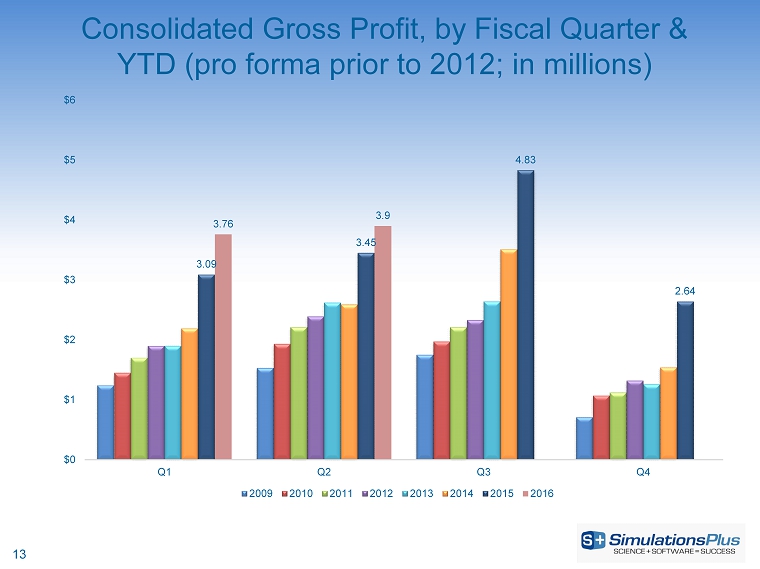
| · | Gross profit increased 13.3%, or $457,000, to $3.90 million from $3.44 million |
| · | SG&A increased 7.2%, or $115,000, to $1.72 million from $1.61 million |
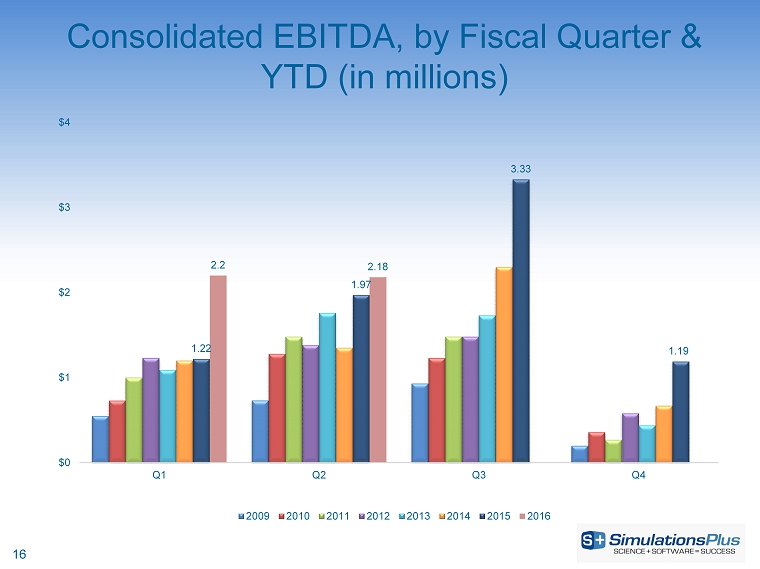
| · | Income before taxes increased 17.1%, or $247,000, to $1.69 million from $1.44 million |
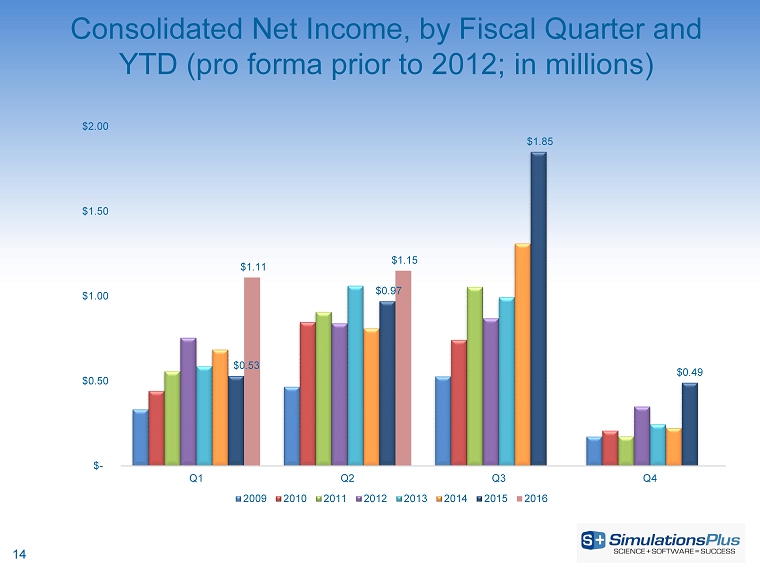
| · | Net income was up by 18%, or $175,000, to $1.15 million from $970,000 |
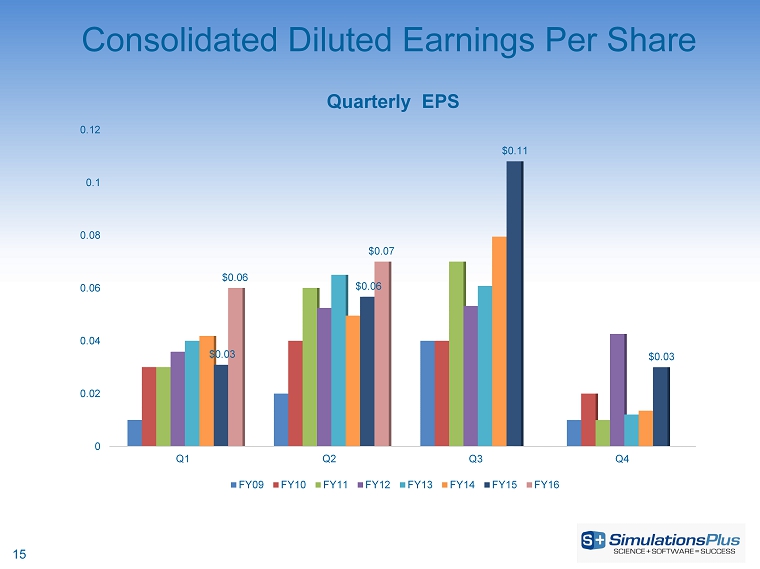
| · | Diluted earnings per share increased 16.9% to $0.066 from $0.057 per share |
6moFY16 highlights compared with 6moFY15:
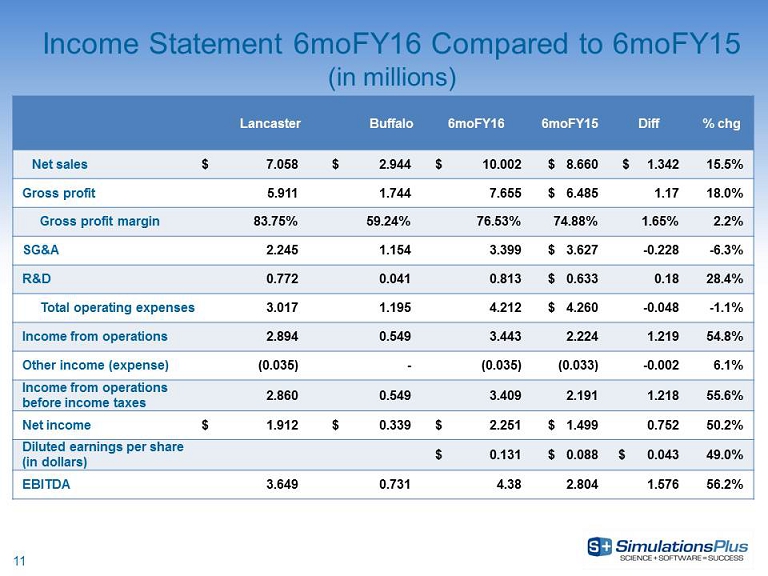
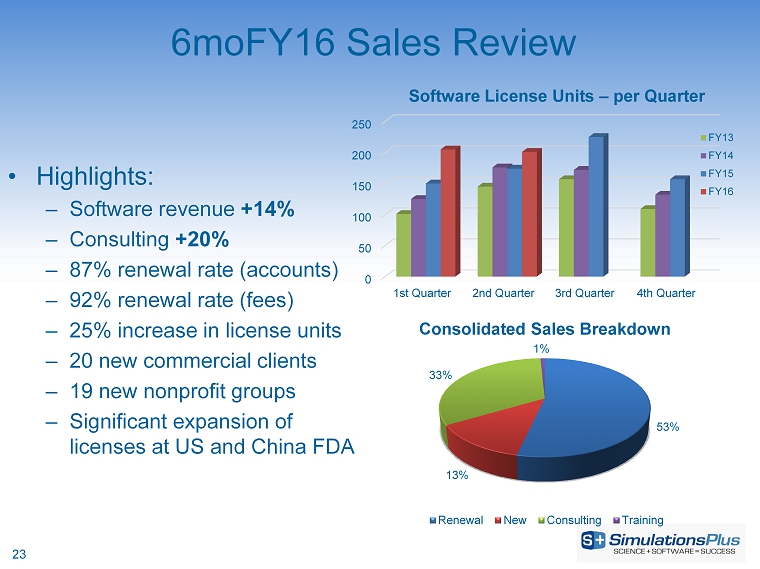
| · | Net revenues increased 15.5%, or $1.34 million, to $10 million from $8.66 million |
| · | Gross profit increased 18%, or $1.17 million, to $7.66 million from $6.48 million |
| · | SG&A decreased 6.3%, or $228,000, to $3.4 million from $3.63 million |
| · | R&D expenditures increased 8.8%, or $110,000, to $1.36 million from $1.25 million in 6moFY15 |
| o | In 6moFY16, $545,000 was capitalized and $813,000 was expensed |
| o | In 6moFY15, $631,000 was capitalized and $633,000 was expensed |
| · | Income before taxes increased 55.6%, or $1.22 million, to $3.41 million from $2.19 million |
| · | Net income increased 50.2%, or $753,000, to $2.25 million from $1.5 million |
| · | Diluted earnings per share increased 49.0% to $0.131 per share from $0.088 per share |
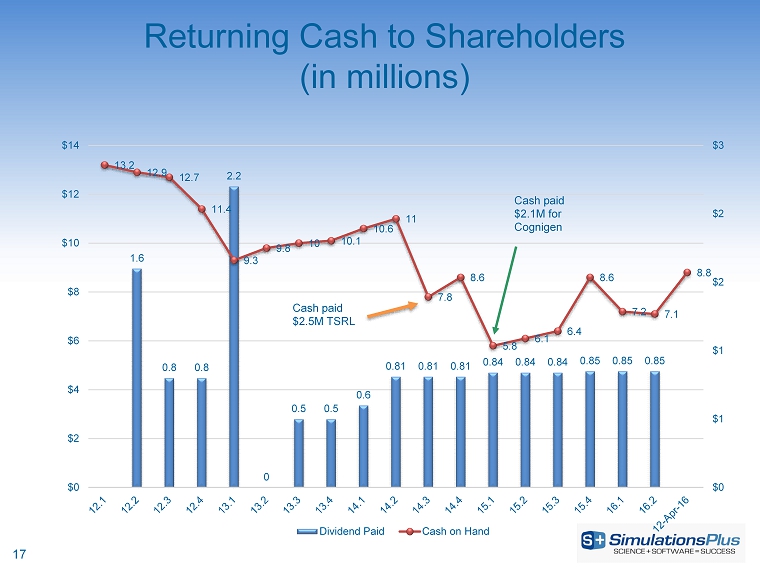
John Kneisel, chief financial officer of Simulations Plus, said: “We continue our nearly decade-long trend of consistent revenue and earnings growth. Cash is back up above $8 million after two dividend distributions totaling over $1.7 million. As announced earlier, we will use approximately $1.47 million in cash this fiscal year to make a payment to TSRL as part of the royalty agreement buyout announced in May 2014, and the final payment to the former shareholders of Cognigen Corporation to close out our acquisition of Cognigen that took place in September 2014. We expect our cash reserves to remain more than adequate after these disbursements, and we anticipate continuing our quarterly dividend distributions; although, such distributions are always at the discretion of the Board of Directors, which votes on them each quarter.”
| 1 |
John DiBella, vice president for marketing and sales of Simulations Plus, said: “At the halfway point of FY2016, we have maintained solid renewal rates for software licenses and continued growth in new license sales and consulting revenues. The marketing and sales staff and our scientists have supported our aggressive marketing and sales program with attendance and presentations at key scientific meetings, conducting training workshops and webinars, and providing the strong customer support that is a hallmark of Simulations Plus in the industry.”
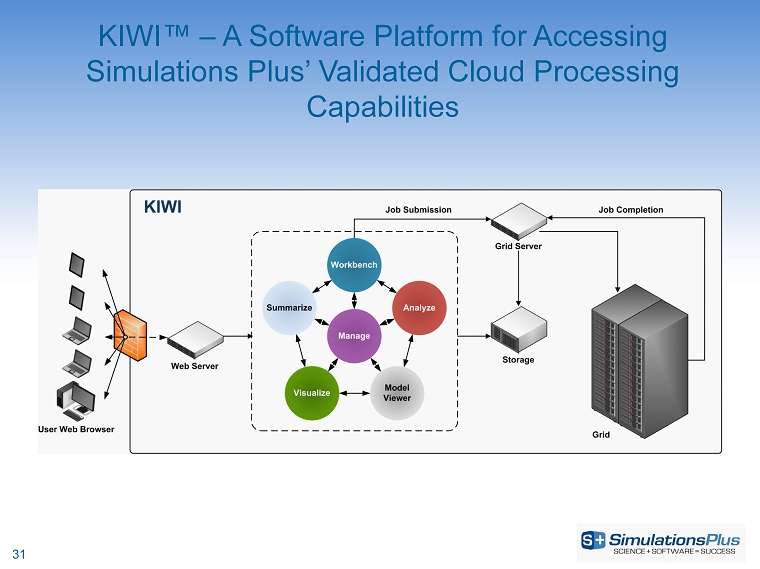
Ted Grasela, president of Simulations Plus, added: “The addition of the recently announced 5-year, $4.7 million contract for data management and consulting services with a major research foundation will add significantly to the revenue growth of our Buffalo division. We believe this effort, which is based on our secure in-house computing cloud and our unique KIWI™ software platform, has the potential to lead to additional opportunities for such services with a number of other research organizations around the world. And we are now routinely providing physiologically based pharmacokinetics (PBPK) analysis to clinical pharmacology groups as we realize the expected synergies between our Lancaster and Buffalo divisions.”
Walt Woltosz, chairman and chief executive officer of Simulations Plus, concluded: “As John DiBella noted, we’re halfway through FY2016, with six-month results showing excellent continued revenues and earnings growth. Now we are beginning to perform on our 5-year contract and expecting the release of our newest software product, PKPlus™, along with updates to two other programs, in the third fiscal quarter. We’re excited about these developments and expect them to add to a strong finish to FY2016.”
Investor Conference Call
The Company will host a conference call on April 13, 2016, at 4:15 p.m. Eastern Time. All interested parties are invited to join the call by registering at: https://attendee.gotowebinar.com/register/5014190111345805827. On registering, you will receive a confirmation e-mail with instructions for joining the call. Please dial in five to ten minutes prior to the scheduled start time. For listen-only mode, you may dial (646) 307-1720, and enter access code 439-293-425.
About Simulations Plus, Inc.
Simulations Plus, Inc. is a premier developer of drug discovery and development software as well as a leading provider of both preclinical and clinical pharmacometric consulting services for regulatory submissions. The company is a global leader focused on improving the ways scientists use knowledge and data to predict the properties and outcomes of pharmaceutical and biotechnology agents. Our software is licensed to and used in the conduct of drug research by major pharmaceutical and biotechnology companies and regulatory agencies worldwide. Our innovations in integrating new and existing science in medicinal chemistry, computational chemistry, pharmaceutical science, biology, and physiology into our software have made us the leading software provider for physiologically based pharmacokinetic modeling and simulation. For more information, visit our website at www.simulations-plus.com.
Follow Us on Twitter
Safe Harbor Statement Under the Private Securities Litigation Reform Act of 1995 – With the exception of historical information, the matters discussed in this press release are forward-looking statements that involve a number of risks and uncertainties. Words like “believe,” “expect” and “anticipate” mean that these are our best estimates as of this writing, but that there can be no assurances that expected or anticipated results or events will actually take place, so our actual future results could differ significantly from those statements. Factors that could cause or contribute to such differences include, but are not limited to: our ability to maintain our competitive advantages, acceptance of new software and improved versions of our existing software by our customers, the general economics of the pharmaceutical industry, our ability to finance growth, our ability to continue to attract and retain highly qualified technical staff, our ability to identify and close acquisitions on terms favorable to the Company, and a sustainable market. Further information on our risk factors is contained in our quarterly and annual reports as filed with the U.S. Securities and Exchange Commission.
--Tables follow --
| 2 |
SIMULATIONS PLUS, INC.
CONDENSED CONSOLIDATED BALANCE SHEETS
As of
| (Unaudited) | (Audited) | |||||||
| February 29, | August 31, | |||||||
| ASSETS | ||||||||
| 2016 | 2015 | |||||||
| Current assets | ||||||||
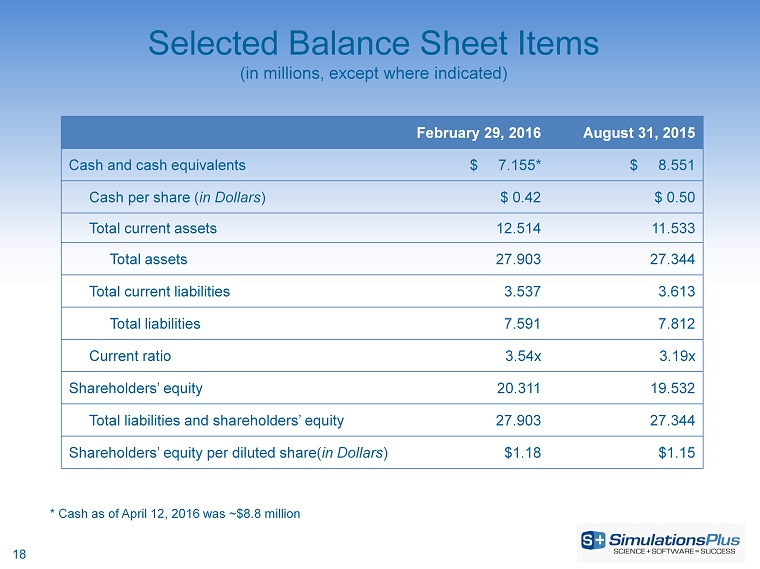
| Cash and cash equivalents | $ | 7,062,993 | $ | 8,551,275 | ||||
| Accounts receivable, net of allowance for doubtful accounts of $0 | 3,808,081 | 1,593,707 | ||||||
| Revenues in excess of billings | 856,558 | 795,125 | ||||||
| Prepaid income taxes | 240,116 | – | ||||||
| Prepaid expenses and other current assets | 309,152 | 381,718 | ||||||
| Deferred income taxes | 237,557 | 210,972 | ||||||
| Total current assets | 12,514,457 | 11,532,797 | ||||||
| Long-term assets | ||||||||
| Capitalized computer software development costs, | ||||||||
| net of accumulated amortization of $8,126,958 and $7,632,421 | 3,849,025 | 3,798,339 | ||||||
| Property and equipment, net | 317,019 | 413,510 | ||||||
| Intellectual property, net of accumulated amortization of $1,105,000 and $801,250 | 4,970,000 | 5,273,750 | ||||||
| Other intangible assets net of accumulated amortization of $221,250 and $147,500 | 1,428,750 | 1,502,500 | ||||||
| Goodwill | 4,789,248 | 4,789,248 | ||||||
| Other assets | 34,082 | 34,082 | ||||||
| Total assets | $ | 27,902,581 | $ | 27,344,226 | ||||
| LIABILITIES AND SHAREHOLDERS' EQUITY | ||||||||
| Current liabilities | ||||||||
| Accounts payable | $ | 126,078 | $ | 209,407 | ||||
| Accrued payroll and other expenses | 441,088 | 429,580 | ||||||
| Accrued bonuses to officer | 60,500 | 121,000 | ||||||
| Income taxes payable | – | 43,602 | ||||||
| Other current liabilities | 18,204 | 19,859 | ||||||
| Current portion - Contracts payable | 2,604,404 | 2,604,404 | ||||||
| Billings in excess of revenues | 172,297 | 106,534 | ||||||
| Deferred revenue | 114,631 | 78,945 | ||||||
| Total current liabilities | 3,537,202 | 3,613,331 | ||||||
| Long-term liabilities | ||||||||
| Deferred income taxes | 3,054,076 | 3,190,419 | ||||||
| Payments due under Contracts payable | 1,000,000 | 1,000,000 | ||||||
| Other long-term liabilities | – | 8,274 | ||||||
| Total liabilities | $ | 7,591,278 | $ | 7,812,024 | ||||
| Commitments and contingencies | – | – | ||||||
| Shareholders' equity | ||||||||
| Preferred stock, $0.001 par value | ||||||||
| 10,000,000 shares authorized | ||||||||
| no shares issued and outstanding | $ | – | $ | – | ||||
| Common stock, $0.001 par value | ||||||||
| 50,000,000 shares authorized | ||||||||
| 17,018,001 and 16,943,001 shares issued and outstanding | 5,489 | 5,414 | ||||||
| Additional paid-in capital | 9,942,194 | 9,714,290 | ||||||
| Retained earnings | 10,363,620 | 9,812,498 | ||||||
| Total shareholders' equity | $ | 20,311,303 | $ | 19,532,202 | ||||
| Total liabilities and shareholders' equity | $ | 27,902,581 | $ | 27,344,226 | ||||
| 3 |
SIMULATIONS PLUS, INC.
CONDENSED CONSOLIDATED STATEMENTS OF OPERATIONS
For the three and six months ended February 29 and 28,
| Three months ended | Six months ended | |||||||||||||||
| (Unaudited) | (Unaudited) | |||||||||||||||
| 2016 | 2015 | 2016 | 2015 | |||||||||||||
| Net Revenues | $ | 5,163,726 | $ | 4,574,191 | $ | 10,002,346 | $ | 8,660,382 | ||||||||
| Cost of revenues | 1,263,741 | 1,130,907 | 2,347,088 | 2,175,582 | ||||||||||||
| Gross profit | 3,899,985 | 3,443,284 | 7,655,258 | 6,484,800 | ||||||||||||
| Operating expenses | ||||||||||||||||
| Selling, general, and administrative | 1,722,844 | 1,607,495 | 3,399,278 | 3,626,994 | ||||||||||||
| Research and development | 461,389 | 360,708 | 812,696 | 633,348 | ||||||||||||
| Total operating expenses | 2,184,233 | 1,968,203 | 4,211,974 | 4,260,342 | ||||||||||||
| Income from operations | 1,715,752 | 1,475,081 | 3,443,284 | 2,224,458 | ||||||||||||
| Other income (expense) | ||||||||||||||||
| Interest income | 4,486 | 4,412 | 8,953 | 9,004 | ||||||||||||
| Gain(loss) on currency exchange | (28,330 | ) | (34,684 | ) | (43,224 | ) | (42,475 | ) | ||||||||
| Total other income (expense) | (23,844 | ) | (30,272 | ) | (34,271 | ) | (33,471 | ) | ||||||||
| Income from operations before prook, vision for income taxes | 1,691,908 | 1,444,809 | 3,409,013 | 2,190,987 | ||||||||||||
| Provision for income taxes | (546,559 | ) | (474,576 | ) | (1,157,191 | ) | (691,851 | ) | ||||||||
| Net Income | $ | 1,145,349 | $ | 970,233 | $ | 2,251,822 | $ | 1,499,136 | ||||||||
| Earnings per share | ||||||||||||||||
| Basic | $ | 0.07 | $ | 0.06 | $ | 0.13 | $ | 0.09 | ||||||||
| Diluted | $ | 0.07 | $ | 0.06 | $ | 0.13 | $ | 0.09 | ||||||||
| Weighted-average common shares outstanding | ||||||||||||||||
| Basic | 17,005,649 | 16,848,983 | 16,985,869 | 16,839,599 | ||||||||||||
| Diluted | 17,268,144 | 17,105,412 | 17,230,099 | 17,096,357 | ||||||||||||
| 4 |