Table of Contents
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
Form 10-K
| x | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the Fiscal Year Ended December 31, 2011
OR
| ¨ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the Transition period from to .
Commission file number: 001-32836
MEDIVATION, INC.
(Exact name of Registrant as specified in its charter)
| Delaware | 13-3863260 | |
| (State or other jurisdiction of incorporation or organization) | (I.R.S. Employer Identification No.) |
201 Spear Street, 3rd Floor
San Francisco, California 94105
(Address of principal executive offices) (Zip Code)
Registrant’s telephone number, including area code: (415) 543-3470
Securities registered pursuant to Section 12(b) of the Act:
| Title of Each Class |
Name of Each Exchange on Which Registered | |
| Common Stock, par value $0.01 per share |
The NASDAQ Stock Market LLC |
Securities registered pursuant to Section 12(g) of the Act:
None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. YES x NO ¨
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. YES ¨ NO x
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports) and (2) has been subject to such filing requirements for the past 90 days. YES x NO ¨
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate Web site, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files). YES x NO ¨
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K is not contained herein, and will not be contained to the best of registrant’s knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K. x
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, or a smaller reporting company. See the definitions of “large accelerated filer,” “accelerated filer” and “smaller reporting company” in Rule 12b-2 of the Exchange Act.
Large accelerated filer x Accelerated filer ¨
Non-accelerated filer (Do not check if a smaller reporting company) ¨ Smaller reporting company ¨
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Act.). YES ¨ NO x
The aggregate market value of the voting stock held by non-affiliates of the registrant was approximately $712,838,841 as of June 30, 2011, based upon the closing sale price on The NASDAQ Global Market reported on June 30, 2011. Excludes an aggregate of 1,643,149 shares of the registrant’s common stock held by officers, directors and affiliated stockholders. For purposes of determining whether a stockholder was an affiliate of the registrant at June 30, 2011, the registrant assumed that a stockholder was an affiliate of the registrant at June 30, 2011 if such stockholder (i) beneficially owned 10% or more of the registrant’s common stock, as determined based on public filings, and/or (ii) was an executive officer or director or was affiliated with an executive officer or director of the registrant at June 30, 2011. Exclusion of such shares should not be construed to indicate that any such person possesses the power, direct or indirect, to direct or cause the direction of the management or policies of the registrant or that such person is controlled by or under common control with the registrant.
There were 36,014,735 shares of Registrant’s Common Stock, par value $0.01 per share, issued and outstanding as of February 22, 2012.
DOCUMENTS INCORPORATED BY REFERENCE
Portions of the registrant’s definitive Proxy Statement for the 2012 Annual Meeting of Stockholders to be filed with the Securities and Exchange Commission pursuant to Regulation 14A not later than 120 days after the end of the fiscal year covered by this Form 10-K are incorporated by reference in Part III, Items 10-14 of this Form 10-K.
Table of Contents
MEDIVATION, INC.
2011 ANNUAL REPORT ON FORM 10-K
| Page | ||||||
| PART I |
||||||
| Item 1. |
BUSINESS | 2 | ||||
| Item 1A. |
14 | |||||
| Item 1B. |
29 | |||||
| Item 2. |
29 | |||||
| Item 3. |
30 | |||||
| Item 4. |
30 | |||||
| PART II |
||||||
| Item 5. |
31 | |||||
| Item 6. |
33 | |||||
| Item 7. |
MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS |
33 | ||||
| Item 7A. |
53 | |||||
| Item 8. |
53 | |||||
| Item 9. |
CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE |
53 | ||||
| Item 9A. |
53 | |||||
| Item 9B. |
54 | |||||
| PART III |
||||||
| Item 10. |
55 | |||||
| Item 11. |
55 | |||||
| Item 12. |
SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS |
55 | ||||
| Item 13. |
CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS , AND DIRECTOR INDEPENDENCE |
55 | ||||
| Item 14. |
PRINCIPAL ACCOUNTING FEES AND SERVICES | 55 | ||||
| PART IV |
||||||
| Item 15. |
EXHIBITS, FINANCIAL STATEMENT SCHEDULES | 56 | ||||
| 60 | ||||||
| FINANCIAL STATEMENTS |
||||||
| REPORT OF PRICEWATERHOUSECOOPERS LLP, INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM |
61 | |||||
| 62 | ||||||
| 63 | ||||||
| 64 | ||||||
| 65 | ||||||
| 66 | ||||||
i
Table of Contents
FORWARD-LOOKING STATEMENTS
This Annual Report on Form 10-K, or Annual Report, includes forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, or the Exchange Act. All statements other than statements of historical facts contained in this Annual Report, including statements regarding our anticipated future clinical and regulatory events, future financial position, business strategy and plans and objectives of management for future operations, are forward-looking statements. Forward looking statements are generally written in the future tense and/or are preceded by words such as “may,” “will,” “should,” “forecast,” “could,” “expect,” “suggest,” “believe,” “estimate,” “continue,” “anticipate,” “intend,” “plan,” or similar words, or the negatives of such terms or other variations on such terms or comparable terminology. Such forward-looking statements include, without limitation, statements regarding the potential future commercialization of our product candidates, the anticipated start dates, durations and completion dates, as well as the potential future results, of our ongoing and future clinical trials, the anticipated designs of our future clinical trials, anticipated future regulatory submissions and events, our anticipated future cash position and future events under our current and potential future collaborations. These forward-looking statements are subject to a number of risks, uncertainties and assumptions, including without limitation the risks described in “Risk Factors” in Part I, Item 1A of this Annual Report. These risks are not exhaustive. Other sections of this Annual Report include additional factors that could adversely impact our business and financial performance. Moreover, we operate in a very competitive and rapidly changing environment. New risk factors emerge from time to time and it is not possible for our management to predict all risk factors, nor can we assess the impact of all factors on our business or the extent to which any factor, or combination of factors, may cause actual results to differ materially from those contained in any forward-looking statements. You should not rely upon forward-looking statements as predictions of future events. We cannot assure you that the events and circumstances reflected in the forward-looking statements will be achieved or occur and actual results could differ materially from those projected in the forward-looking statements. We assume no obligation to update or supplement forward-looking statements.
1
Table of Contents
PART I
| Item 1. | Business. |
Overview
We are a biopharmaceutical company focused on the rapid development of novel small molecule drugs to treat serious diseases for which there are limited treatment options. Together with our collaboration partner Astellas Pharma Inc., or Astellas, we are developing MDV3100 for multiple stages of advanced prostate cancer. We have successfully completed a Phase 3 trial in the latest stage patients—those who have failed docetaxel-based chemotherapy—and we are conducting an additional Phase 3 trial and two Phase 2 trials in men with earlier stages of advanced prostate cancer. Based on the positive results of the AFFIRM trial, we have exercised our right under our collaboration agreement with Astellas to co-promote MDV3100 in the U.S., should it receive marketing approval. We and Astellas expect to file applications for marketing approval for MDV3100 both in the U.S. and in Europe in 2012. MDV3100 has received “Fast Track” designation from the U.S. Food and Drug Administration, or FDA, for the post-docetaxel indication.
We have not generated any revenue from product sales to date, and we may never generate any revenue from product sales. We have funded our operations primarily through private and public offerings of our common stock and from the up-front, development milestone and cost-sharing payments from Astellas and from our former collaboration partner Pfizer, Inc., or Pfizer. We have incurred cumulative net losses of $250.3 million through December 31, 2011, and we expect to incur substantial additional losses for the foreseeable future as we pursue regulatory approval for, and, if approved, commercial launch of, MDV3100 and continue to finance clinical and preclinical studies of our existing and potential future product candidates and our corporate overhead costs.
We are a corporation formed in Delaware in October 1995, under our former name Orion Acquisition Corp. II, to identify and consummate a business combination. Our three subsidiaries are Medivation Prostate Therapeutics, Inc., or MPT, Medivation Neurology, Inc., or MNI, and Medivation Technologies, Inc., or MTI. MPT holds our MDV300 series technology, which is in development for the treatment of advanced prostate cancer under our collaboration agreement with Astellas, and MTI holds our earlier stage technologies. MNI holds our dimebon technology, which previously was in development for the treatment of Alzheimer’s disease and Huntington disease under our former collaboration agreement with Pfizer. As discussed elsewhere in this Annual Report, in January 2012, Pfizer exercised its right to terminate the collaboration agreement and we and Pfizer discontinued development of dimebon for all indications.
Our MDV300 Series Prostate Cancer Program
We have obtained an exclusive, worldwide commercial license to a series of novel small molecules, referred to as the MDV300 series compounds. Our lead development candidate from the MDV300 series is a molecule we refer to as MDV3100, which is in Phase 3 development for a type of advanced prostate cancer known as castration-resistant prostate cancer, or CRPC. We are conducting this program in collaboration with Astellas.
Prostate Cancer Statistics
According to the American Cancer Society, prostate cancer is the most commonly diagnosed cancer among men in the United States, other than skin cancer. The American Cancer Society estimates that approximately 241,000 new cases of prostate cancer were diagnosed, and approximately 34,000 men died of prostate cancer, in the United States alone during 2011. Prostate cancer is thus the second-leading cause of cancer death in men in the United States, after lung cancer. According to the American Cancer Society, about 1 in 6 men will be diagnosed with prostate cancer during his lifetime and about 1 in 36 men will die of prostate cancer.
Advanced Prostate Cancer
Prostate cancer is frequently diagnosed at a stage where it is believed to be confined to the prostate gland and its immediate surroundings—i.e., it has not yet metastasized to other areas of the body. Prostate cancer
2
Table of Contents
detected at this stage generally is treated either with prostatectomy (surgical removal of the prostate gland) or with radiation. For most men, these procedures are successful in curing the disease. However, for some men, these procedures are not curative and their prostate cancer continues to spread. This disease progression is typically detected by rising levels of serum prostate specific antigen, or PSA. Men whose disease continues to progress following surgery or radiation are considered to have advanced prostate cancer.
Treatment of Advanced Prostate Cancer
The Testosterone Signaling Pathway. Prostate cancer is fueled by the male sex hormone testosterone. Testosterone is produced primarily in the testes, although lesser amounts of testosterone are also produced in the adrenal glands and in prostate cancer tumors themselves. In order to fuel prostate cancer growth, testosterone must first bind to its receptor, known as the androgen receptor, which is located predominantly in the cytoplasm of prostate cancer cells (the area within the cell membrane but outside the nucleus). Once binding has occurred, the bound testosterone/androgen receptor complex must then pass from the cytoplasm into the nucleus of the cell, a process known as nuclear translocation. Finally, once inside the nucleus, the bound complex must then bind to and activate DNA, which triggers cell growth and thus tumor progression.
Established Hormonal Therapies. Because testosterone is the primary fuel of prostate cancer growth, first-line medical therapy for advanced prostate cancer typically entails treatment with a class of drug known as luteinizing hormone releasing hormone, or LHRH, analogs, which reduce testosterone to castrate levels—i.e., the levels that would be achieved following surgical castration. Patients treated with LHRH analogs typically remain on those drugs for the remainder of their lives, in order to keep testosterone levels suppressed to castrate levels. Estimated sales of LHRH analog drugs in the United States, United Kingdom, France, Germany, Italy, Spain and Japan, or the G7 countries, were approximately $2.6 billion in 2009 according to Decision Resources. Another class of marketed hormonal drugs, known as anti-androgens, block the ability of testosterone to bind its receptor, the androgen receptor. These drugs are often added on to LHRH analog treatment as second-line therapy for advanced prostate cancer. In some cases, advanced prostate cancer patients are started on both an LHRH analog and an anti-androgen simultaneously, a treatment regimen known as combined androgen blockade. Casodex® (bicalutamide), sold by AstraZeneca PLC, is the largest selling anti-androgen drug, with global annual sales of approximately $580 million in 2010 according to the public disclosures of AstraZeneca PLC. Generic versions of bicalutamide are now available.
Most advanced prostate cancer initially responds to these hormonal therapies. However, according to a study published in the October 7, 2004 issue of The New England Journal of Medicine, virtually all advanced prostate cancer undergoes changes in a median of 18-24 months after initiation of hormonal therapy that allows the cancer to continue to grow despite the reduction of testosterone to very low (i.e., castrate) levels. Prostate cancer that has reached this state is known as CRPC. The development of CRPC following initiation of hormonal therapy is generally determined based on either rising levels of PSA or documented disease progression as evidenced by imaging tests or clinical symptoms. Due to biological changes that have occurred in CRPC, drugs such as bicalutamide that initially decrease androgen receptor signaling and inhibit prostate cancer growth may have precisely the opposite effect and start to fuel the growth of CRPC.
Chemotherapies. It was previously believed that prostate cancers that had entered the CRPC state would no longer respond to hormonal therapies. Thus, the next line of treatment for these patients has typically been chemotherapy. The primary chemotherapy for CRPC patients is Taxotere® (docetaxel), which has been shown in clinical studies to prolong survival by approximately 10 weeks. However, docetaxel is an infused cytotoxic chemotherapy, and thus entails an increased risk of serious adverse effects, including fluid retention, liver toxicity, low white blood cell counts, and death. Nonetheless, according to Decision Resources, sales of Taxotere for the treatment of prostate cancer in the G7 countries were $629 million in 2009. In 2010, the FDA approved a new second-line chemotherapy, Jevtana® (cabazitaxel), for use in CRPC patients who had previously failed docetaxel treatment. Jevtana (cabazitaxel) was shown in clinical studies to prolong median survival by approximately 10 weeks, but like docetaxel is an infused cytotoxic chemotherapy that entails increased risk of death and other serious adverse events.
3
Table of Contents
Prostate Cancer Vaccines. In 2010, the FDA approved the first vaccine for CRPC. Prostate cancer vaccines operate by enhancing the ability of the body’s immune system to attack and destroy prostate cancer cells. This agent, Provenge® (sipuleucel-T), was approved based on data demonstrating a median overall survival advantage of approximately four months in CRPC patients, the large majority of whom had not previously undergone chemotherapy.
Novel Hormonal Therapies. In April 2011, Zytiga (abiraterone acetate) was approved by the FDA for use in combination with the steroid prednisone to treat CRPC patients who had previously failed docetaxel-based chemotherapy. Zytiga (abiraterone acetate) operates by reducing production of testosterone in the adrenal glands, a secondary source of testosterone production in the body. Zytiga (abiraterone acetate) demonstrated an overall survival advantage of approximately four months in post-docetaxel CRPC patients and that prostate cancers that continue to grow despite testosterone having been reduced to castrate levels remain responsive to hormonal therapies.
MDV3100
MDV3100 is an investigational agent that is the first in a new class of medicines called androgen receptor signaling inhibitors.
Mechanism of Action
While MDV3100, like all other hormonal therapies for prostate cancer, operates through the testosterone signaling pathway, it does so in a manner that is distinct from that of currently approved drugs. An article published in May 2009 in Science described the novel mechanism of action of MDV3100. In the Science article, researchers using various preclinical models of CRPC provided evidence that MDV3100 inhibits 1) testosterone binding to androgen receptors; 2) nuclear translocation of androgen receptors; and 3) DNA binding and activation by androgen receptors. This preclinical research also found that, by interfering with testosterone signaling in these three distinct ways, MDV3100 induces the death of CRPC cells.
Completed Clinical Trials
Phase 3 AFFIRM Trial
In November 2011, we reported positive results from a planned interim analysis of the AFFIRM trial, a randomized, double-blind Phase 3 trial evaluating MDV3100 (160 mg once daily) versus placebo in 1,199 patients with advanced prostate cancer who had previously failed docetaxel-based chemotherapy. The primary endpoint of the AFFIRM trial was overall survival. The Independent Data Monitoring Committee, or IDMC, overseeing the AFFIRM trial determined that MDV3100 demonstrated a clinically meaningful and statistically significant (p<0.0001) improvement in overall survival compared to placebo, and as a result recommended that the study be stopped early and men who received placebo be offered MDV3100. The IDMC informed us that 1) MDV3100 produced a 4.8-month advantage in median survival compared to placebo; 2) the estimated median survival for men treated with MDV3100 was 18.4 months compared to 13.6 months for men treated with placebo; and 3) MDV3100 provided a 37% reduction in risk of death compared to placebo (hazard ratio = 0.631). The IDMC further determined, considering the observed safety profile, that MDV3100 demonstrated a favorable risk-to-benefit ratio sufficient to stop the study.
In February 2012, we reported further results from the AFFIRM trial at the American Society of Clinical Oncology’s 2012 Genitourinary Cancers Symposium, or ASCO GU. In addition to confirming the results reported in November 2011, we reported that MDV3100 had achieved all of the trial’s secondary endpoints with strong statistical significance and was well tolerated. As compared to patients taking placebo, MDV3100 patients in the AFFIRM trial experienced longer median radiographic progression-free survival (8.3 versus 2.9 months; p<0.0001; hazard ratio = 0.404), a higher soft tissue response rate (28.9% versus 3.8%; p<0.0001), and longer
4
Table of Contents
median time to PSA progression (8.3 versus 3.0 months; p<0.0001; hazard ratio = 0.249). PSA declines of 50% or greater were more common in the MDV3100 group than in the placebo group (54.0% versus 1.5%; p<0.0001), as were PSA declines of 90% or greater (24.8% versus 0.9%; p<0.0001).
MDV3100 was well tolerated in the AFFIRM trial. Common side effects included fatigue, diarrhea and hot flush. Serious adverse events, adverse events causing patients to stop treatment, and adverse events causing death all were lower in the MDV3100 group than in the placebo group. Serious side effects of interest were fatigue (6.3% in the MDV3100 group versus 7.3% in the placebo group), cardiac disorders (0.9% versus 2.0%) including myocardial infarction (0.3% versus 0.5%), seizure (0.6% versus 0.0%) and liver function test abnormalities (0.4% versus 0.8%). The period during which AFFIRM patients were followed for safety issues was more than twice as long for MDV3100 patients (9.3 months) as compared to placebo patients (4.0 months).
The FDA has granted Fast Track designation for the development of MDV3100 in the post-chemotherapy indication, a designation that is reserved for development programs that the FDA determines to be for a life-threatening condition with unmet medical need. Receipt of Fast Track designation enables us to request that the FDA grant us priority review for our anticipated new drug application, or NDA, in post-chemotherapy patients. In considering requests for priority review, the FDA applies the same standard it uses to award Fast Track designation. We plan to hold a pre-NDA meeting with the FDA in early 2012. We and Astellas expect to file both a NDA with the FDA and a marketing authorization application, or MAA, with the European Medicines Agency in 2012 seeking approval to market MDV3100 for post-docetaxel advanced prostate cancer in the U.S. and Europe, respectively.
Based on the positive results from the AFFIRM trial, we elected to exercise our right under the Astellas Collaboration Agreement to co-promote MDV3100 in the U.S. market. Should MDV3100 receive marketing approval, we will provide 50% of the sales and medical affairs support for MDV3100 in the U.S. market.
Phase 1-2 Trial
In December 2008, we completed enrollment in an open-label Phase 1-2 clinical trial of MDV3100 in patients with CRPC. We enrolled 140 patients in seven dose groups, ranging from 30 mg/day to 600 mg/day. Of the 140 patients, 75 had previously failed docetaxel-based chemotherapy and 65 were chemotherapy-naïve. All 140 patients also had failed at least one line of prior hormonal therapy. The trial endpoints included safety, tolerability, pharmacokinetics, circulating tumor cell, or CTC, counts, PSA levels, radiographic change in soft tissue and bony metastases, and time to progression.
Data from this trial were published in The Lancet in 2010. These data showed that MDV3100 consistently demonstrated anti-tumor activity across endpoints, as evaluated by reductions in PSA levels, radiographic findings and CTC counts. This activity was consistently stronger in the earlier stage (i.e. chemotherapy naïve) as compared to the later stage (i.e., post-chemotherapy) patients. Key efficacy data as reported in The Lancet were as follows:
| Chemotherapy-Naïve Patients | Post-Chemotherapy Patients | |||
| PSA decline > 50% from baseline |
62% | 51% | ||
| Radiographic control: soft-tissue lesions (partial response or stable disease) |
80% | 65% | ||
| Radiographic control: bony lesions (stable disease) |
63% | 51% | ||
| CTC conversion from “poor prognosis” to “good prognosis” |
75% | 37% |
In February 2011, we presented new long-term follow-up data covering all 140 patients enrolled in the trial at 2011 ASCO GU. PSA progression data reported at 2011 ASCO GU were calculated using three distinct
5
Table of Contents
reporting criteria: the criteria specified in the Phase 1-2 trial protocol; the most recent published PSA reporting consensus criteria (the Prostate Cancer Clinical Trials Working Group 2, or PCWG2, criteria); and an older commonly used reporting method (the Prostate-Specific Antigen Working Group 1, or PSAWG1, criteria). Key efficacy data as presented at 2011 ASCO GU were as follows:
| Chemotherapy-Naïve Patients |
Post-Chemotherapy Patients | |||
| Median time to PSA progression (per protocol criteria) |
Not reached | 316 days (45 weeks) | ||
| Median time to PSA progression (per PCWG2 criteria) |
281 days (40 weeks) | 148 days (21 weeks) | ||
| Median time to PSA progression (per PSAWG1 criteria) |
420 days (60 weeks)* 812 days (116 weeks)** |
166 days (24 weeks) | ||
| Median time to radiographic progression |
394 days (56 weeks) | 173 days (25 weeks) |
| * | All chemotherapy-naïve patients |
| ** | Subpopulation of chemotherapy-naïve patients who were also ketoconazole-naïve |
MDV3100 was well tolerated in this trial at doses up to and including 240 mg/day. The most frequently reported adverse event was fatigue. Seizures were observed in two patients, one each at doses of 600 and 360 mg/day. Both patients were taking concomitant medications that can cause seizures. A possible but unwitnessed seizure was reported in a patient taking a dose of 480 mg/day.
Ongoing Clinical Trials
Phase 3 PREVAIL Trial
PREVAIL is a randomized, double-blind, placebo-controlled Phase 3 trial evaluating MDV3100 (160 mg once daily) versus placebo in approximately 1,700 patients with advanced prostate cancer who have not previously been treated with chemotherapy. The co-primary endpoints are progression-free survival and overall survival. We began enrollment in the PREVAIL trial in September 2010.
Phase 2 TERRAIN Trial
TERRAIN is a randomized, double-blind Phase 2 trial evaluating MDV3100 versus bicalutamide, the leading marketed anti-androgen drug, in approximately 370 advanced prostate cancer patients who have progressed following medical castration with an LHRH analog drug or surgical castration. The primary endpoint is progression-free survival. We began enrollment in the TERRAIN trial in March 2011.
Phase 2 Hormone-Naïve Trial
In May 2011, we initiated a Phase 2 trial evaluating MDV3100 in approximately 60 patients with advanced prostate cancer who have not had any previous hormonal treatment. This is the first trial to examine the effects of MDV3100 in the earlier-stage population of advanced prostate cancer patients who have not yet undergone medical or surgical castration. The primary endpoint in the trial is PSA response.
The Astellas Collaboration Agreement
Our global development and commercialization agreement with Astellas, or the Astellas Collaboration Agreement, became effective in October 2009. Under the Astellas Collaboration Agreement, we and Astellas agreed to collaborate on the development of MDV3100 for the United States market, including associated regulatory filings with the FDA. In addition, if approved by the FDA, following such approval and the launch of MDV3100 in the United States, we and Astellas will co-promote MDV3100 in the United States. Astellas is responsible for development of, seeking regulatory approval for, and commercialization of MDV3100 outside the
6
Table of Contents
United States. Astellas will be responsible for commercial manufacture of MDV3100 on a global basis. Both we and Astellas have agreed not to commercialize certain other products having a similar mechanism of action as MDV3100 for the treatment of specified indications for a specified time period, subject to certain exceptions.
We and Astellas share equally the costs of developing and commercializing MDV3100 for the United States market (subject to the exceptions noted below), and we and Astellas will share equally profits (or losses) resulting from the commercialization of MDV3100 in the United States. Costs of clinical trials supporting development in both the United States and in either Europe or Japan are borne two-thirds by Astellas and one-third by us. Both we and Astellas will be responsible for all costs incurred in fielding and supporting our respective MDV3100 sales forces, and each of us will be entitled to receive a fee for each detail made by our respective sales forces. Outside the United States, Astellas will bear all development and commercialization costs and will pay us tiered, double-digit royalties on the aggregate net sales of MDV3100.
The agreement establishes several joint committees consisting of an equal number of representatives from both parties that operate by consensus to oversee the collaboration. In the event that a joint committee is unable to reach consensus on a particular issue, then, depending on the issue, a dispute may be decided at the joint committee level by the party with the final decision on the issue or escalated to senior management of the parties. If a dispute is escalated to senior management and no consensus is reached, then the dispute may be decided by the party to whom the contract grants final decision on such issue. Other issues can only be decided by consensus of the parties, and unless and until the parties’ representatives reach agreement on such issue, no decision on such issue will be made, and the status quo will be maintained.
Under the Astellas Collaboration Agreement, Astellas paid us a non-refundable, up-front cash payment of $110.0 million in the fourth quarter of 2009. We are also eligible to receive up to $335.0 million in development milestone payments, plus up to an additional $320.0 million in commercial milestone payments. As of December 31, 2011, we had received an aggregate of $13.0 million in development milestone payments under the Astellas Collaboration Agreement. Should the FDA or European Medicines Agency accept for filing an NDA or MAA, respectively, seeking approval of MDV3100 in post-chemotherapy patients based on the results of our AFFIRM trial, we would be entitled to a $10.0 million and a $5.0 million milestone payment, respectively, under the Astellas Collaboration Agreement, respectively. In addition, should the NDA be approved by the FDA or the MAA be approved by the European regulators, we would be entitled to a $30.0 million and a $15.0 million milestone payment under the Astellas Collaboration Agreement. We are required to share 10% of the up-front and development milestone payments received under the Astellas Collaboration Agreement with The Regents of the University of California, or UCLA, pursuant to the terms of our MDV3100 license agreement, which is discussed in the section below titled “License Agreement with UCLA.”
Each of Medivation and Astellas is permitted to terminate the Astellas Collaboration Agreement for an uncured material breach by the other party or for the insolvency of the other party. Astellas has a right to terminate the Astellas Collaboration Agreement unilaterally by advance written notice to us, but, except in certain specific circumstances, generally cannot exercise that termination right until the first anniversary of MDV3100’s first commercial sale. Following any termination of the Astellas Collaboration Agreement in its entirety, all rights to develop and commercialize MDV3100 will revert to us, and Astellas will grant a license to us to enable us to continue such development and commercialization. In addition, except in the case of a termination by Astellas for our uncured material breach, Astellas will supply MDV3100 to us during a specified transition period.
License Agreement with UCLA
Under an August 2005 license agreement with UCLA, and subsequent amendments to this agreement, our subsidiary MPT holds an exclusive worldwide license under several UCLA patents and patent applications related to our MDV300 series compounds. Under our Astellas Collaboration Agreement, we granted Astellas a sublicense under the patent rights licensed to us by UCLA.
7
Table of Contents
We are required to pay UCLA an annual maintenance fee, up to $5.5 million in aggregate milestone payments upon the achievement of certain development and regulatory milestone events, and 10% of any up-front and development milestone payments we receive from sublicensees. We are also required to pay UCLA a single-digit royalty on sales of products falling within the scope of the patent rights licensed from UCLA. Should we receive marketing approval of MDV3100 either in the U.S. or Europe, we would be required to pay UCLA a single, one-time milestone payment of $2.0 million. UCLA may terminate the agreement if we do not meet a general obligation to diligently proceed with the development, manufacture, and sale of licensed products, or if we commit any other uncured material breach of the agreement. UCLA may also terminate the agreement if we fail to meet specific development, regulatory, and commercialization milestones by agreed-upon deadlines, which we may extend for a limited time period by paying an extension fee. We may terminate the agreement at any time upon advance written notice to UCLA. If neither party terminates the agreement early, the agreement will continue in force until the expiration of the last-to-expire licensed patent.
Termination of Pfizer Collaboration Agreement and Dimebon Program
In January 2012, we reported negative top line results from our Phase 3 CONCERT trial of our product candidate dimebon in patients with mild-to-moderate Alzheimer’s disease. We previously had reported negative top line results from our Phase 3 CONNECTION trial of dimebon in patients with mild-to-moderate Alzheimer’s disease and our Phase 3 HORIZON trial of dimebon in patients with Huntington disease. In January 2012, Pfizer exercised its right to terminate our collaboration agreement for the development and commercialization of dimebon and we and Pfizer discontinued development of dimebon for all indications. During the ensuing 180 days, we and Pfizer will work together to wind down our respective remaining collaboration activities.
Intellectual Property
We have an exclusive license to multiple issued patents and pending applications covering the MDV300 series compounds and uses thereof, including issued composition of matter patents covering MDV3100 in the U.S., Europe and Japan. The terms of these issued MDV3100 composition of matter patents expire in 2027 in the U.S. and in 2026 in Europe and Japan. We also own multiple pending patent applications covering our earlier stage technology programs. We intend to prosecute our owned intellectual property, and request that our licensors prosecute our licensed intellectual property, in the U.S., Europe, Japan and other jurisdictions that we deem appropriate.
Government Regulation and Product Approvals
FDA Approval Process
In the United States, pharmaceutical products are subject to extensive regulation by the FDA. The Federal Food, Drug, and Cosmetic Act, or the FDC Act, and other federal and state statutes and regulations, govern, among other things, the research, development, testing, manufacture, storage, recordkeeping, approval, labeling, promotion and marketing, distribution, post-approval monitoring and reporting, sampling, and import and export of pharmaceutical products. Failure to comply with applicable U.S. requirements may subject a company to a variety of administrative or judicial sanctions, such as FDA refusal to approve pending NDAs, warning letters, product recalls, product seizures, total or partial suspension of production or distribution, injunctions, fines, civil penalties, and criminal prosecution.
Pharmaceutical product development in the U.S. typically involves preclinical laboratory and animal tests, the submission to the FDA of either a notice of claimed investigational exemption or an investigational new drug application, or IND, which must become effective before clinical testing may commence, and adequate and well-controlled clinical trials to establish the safety and effectiveness of the drug for each indication for which FDA approval is sought. Satisfaction of FDA pre-market approval requirements typically takes many years and the actual time required may vary substantially based upon the type, complexity, and novelty of the product or disease.
8
Table of Contents
Preclinical tests include laboratory evaluation of product chemistry, formulation, and toxicity, as well as animal trials to assess the characteristics and potential safety and efficacy of the product. The conduct of the preclinical tests must comply with federal regulations and requirements, including good laboratory practices. The results of preclinical testing are submitted to the FDA as part of an IND along with other information, including information about product chemistry, manufacturing and controls, and a proposed clinical trial protocol. Long-term preclinical tests, such as animal tests of reproductive toxicity and carcinogenicity, may begin or continue after the IND is submitted.
A 30-day waiting period after the submission of each IND is required prior to the commencement of clinical testing in humans. If the FDA has neither commented on nor questioned the IND within this 30-day period, the clinical trial proposed in the IND may begin.
Clinical trials involve the administration of the IND to healthy volunteers or patients under the supervision of a qualified investigator. Clinical trials must be conducted: 1) in compliance with federal regulations; 2) in compliance with good clinical practice, or GCP, an international standard meant to protect the rights and health of patients and to define the roles of clinical trial sponsors, administrators, and monitors; as well as 3) under protocols detailing the objectives of the trial, the parameters to be used in monitoring safety, and the effectiveness criteria to be evaluated. Each protocol involving testing on U.S. patients and subsequent protocol amendments must be submitted to the FDA as part of the IND.
The FDA may order the temporary, or permanent, discontinuation of a clinical trial at any time, or impose other sanctions, if it believes that the clinical trial either is not being conducted in accordance with FDA requirements or presents an unacceptable risk to the clinical trial patients. The study protocol and informed consent information for patients in clinical trials must also be submitted to an institutional review board (IRB) or Ethics Committee (EC) for approval. An IRB/EC may also require the clinical trial at the site to be halted, either temporarily or permanently, for failure to comply with the IRB/EC’s requirements, or may impose other conditions.
Clinical trials to support NDAs for marketing approval are typically conducted in three sequential phases, but the phases may overlap. In Phase 1, the initial introduction of the drug into healthy human subjects or patients, the drug is tested to assess metabolism, pharmacokinetics, pharmacological actions, side effects associated with increasing doses, and, if possible, early evidence on effectiveness. Phase 2 usually involves trials in a limited patient population to determine the effectiveness of the drug for a particular indication, dosage tolerance, and optimum dosage, and to identify common adverse effects and safety risks. If a compound demonstrates evidence of effectiveness and an acceptable safety profile in Phase 2 evaluations, Phase 3 trials are undertaken to obtain the additional information about clinical efficacy and safety in a larger number of patients, typically at geographically dispersed clinical trial sites, to permit FDA to evaluate the overall benefit-risk relationship of the drug and to provide adequate information for the labeling of the drug.
After completion of the required clinical testing, a NDA is prepared and submitted to the FDA. FDA approval of the NDA is required before marketing of the product may begin in the U.S. The NDA must include the results of all preclinical, clinical, and other testing and a compilation of data relating to the product’s pharmacology, chemistry, manufacture, and controls. The cost of preparing and submitting a NDA is substantial. Under federal law, the submission of most NDAs is additionally subject to a substantial application user fee, currently exceeding $1.8 million and the manufacturer and/or sponsor under an approved NDA are also subject to annual product and establishment user fees, currently approximately $0.1 million per product and $0.5 million per establishment. These fees are typically increased annually.
The FDA has 60 days from its receipt of a NDA to determine whether the application will be accepted for filing based on the FDA’s threshold determination that it is sufficiently complete to permit substantive review. Once the submission is accepted for filing, the FDA begins an in-depth review. The FDA has agreed to certain performance goals in the review of NDAs. Most such applications for standard review drug products are reviewed within ten months; most applications for priority review drugs are reviewed in six months. We expect
9
Table of Contents
the FDA to amend each of these goals to extend them by two months for applications received after September 2012. Priority review can be applied to drugs that the FDA determines offer major advances in treatment, or provide a treatment where no adequate therapy exists. For biologics, priority review is further limited only for drugs intended to treat a serious or life-threatening disease relative to the currently approved products. The review process for both standard and priority review may be extended by FDA for three additional months to consider certain late-submitted information, or information intended to clarify information already provided in the submission. The FDA may also refer applications for novel drug products, or drug products which present difficult questions of safety or efficacy, to an advisory committee—typically a panel that includes clinicians and other experts—for review, evaluation, and a recommendation as to whether the application should be approved. The FDA is not bound by the recommendation of an advisory committee, but it generally follows such recommendations. Before approving a NDA, the FDA will typically inspect one or more clinical sites to assure compliance with GCP. Additionally, the FDA will inspect the facility or the facilities at which the drug is manufactured. The FDA will not approve the product unless compliance with current good manufacturing practices, or cGMP—a quality system regulating manufacturing—is satisfactory and the NDA contains data that provide substantial evidence that the drug is safe and effective in the indication studied.
After FDA evaluates the NDA and the manufacturing facilities, it issues either an approval letter or a complete response letter. A complete response letter generally outlines the deficiencies in the submission and may require substantial additional testing or information in order for the FDA to reconsider the application. If, or when, those deficiencies have been addressed to the FDA’s satisfaction in a resubmission of the NDA, the FDA will issue an approval letter. FDA has committed to reviewing such resubmissions in two or six months depending on the type of information included.
An approval letter authorizes commercial marketing of the drug with specific prescribing information for specific indications. As a condition of NDA approval, the FDA may require a risk evaluation and mitigation strategy, or REMS, to help ensure that the benefits of the drug outweigh the potential risks. REMS can include medication guides, communication plans for healthcare professionals, and elements to assure safe use, or ETASU. ETASU can include, but are not limited to, special training or certification for prescribing or dispensing, dispensing only under certain circumstances, special monitoring, and the use of patient registries. The requirement for a REMS can materially affect the potential market and profitability of the drug. Moreover, product approval may require substantial post-approval testing and surveillance to monitor the drug’s safety or efficacy. Once granted, product approvals may be withdrawn if compliance with regulatory standards is not maintained or problems are identified following initial marketing.
The Hatch-Waxman Act
In seeking approval for a drug through a NDA, applicants are required to list with the FDA each patent whose claims cover the applicant’s product. Upon approval of a drug, each of the patents listed in the application for the drug is then published in the FDA’s Approved Drug Products with Therapeutic Equivalence Evaluations, commonly known as the Orange Book. Drugs listed in the Orange Book can, in turn, be cited by potential competitors in support of approval of an abbreviated new drug application, or ANDA. An ANDA provides for marketing of a drug product that has the same active ingredients in the same strengths and dosage form as the listed drug and has been shown through bioequivalence testing to be therapeutically equivalent to the listed drug. Other than the requirement for bioequivalence testing, ANDA applicants are not required to conduct, or submit results of, pre-clinical or clinical tests to prove the safety or effectiveness of their drug product. Drugs approved in this way are commonly referred to as “generic equivalents” to the listed drug, and can often be substituted by pharmacists under prescriptions written for the original listed drug.
The ANDA applicant is required to certify to the FDA concerning any patents listed for the approved product in the FDA’s Orange Book. Specifically, the applicant must certify that: 1) the required patent information has not been filed; 2) the listed patent has expired; 3) the listed patent has not expired, but will expire on a particular date and approval is sought after patent expiration; or 4) the listed patent is invalid or will not be
10
Table of Contents
infringed by the new product. A certification that the new product will not infringe the already approved product’s listed patents, or that such patents are invalid, is called a Paragraph IV certification. If the applicant does not challenge the listed patents, the ANDA application will not be approved until all the listed patents claiming the referenced product have expired.
If the ANDA applicant has provided a Paragraph IV certification to the FDA, the applicant must also send notice of the Paragraph IV certification to the NDA and patent holders once the ANDA has been accepted for filing by the FDA. The NDA and patent holders may then initiate a patent infringement lawsuit in response to the notice of the Paragraph IV certification. The filing of a patent infringement lawsuit within 45 days of the receipt of a Paragraph IV certification automatically prevents the FDA from approving the ANDA until the earlier of 30 months, expiration of the patent, settlement of the lawsuit, or a decision in the infringement case that is favorable to the ANDA applicant.
The ANDA application also will not be approved until any non-patent exclusivity listed in the Orange Book for the referenced product has expired. Federal law provides a period of five years following approval of a drug containing no previously approved active ingredients during which ANDAs for generic versions of those drugs cannot be submitted, unless the submission contains a Paragraph IV challenge to a listed patent—in which case the submission may be made four years following the original product approval. Federal law provides for a period of three years of exclusivity during which FDA cannot grant effective approval of an ANDA based on the approval of a listed drug that contains previously approved active ingredients but is approved in a new dosage form, route of administration or combination, or for a new use; the approval of which was required to be supported by new clinical trials conducted by, or for, the applicant.
Advertising and Promotion
Once a NDA is approved, a product will be subject to certain post-approval requirements. For instance, the FDA closely regulates the post-approval marketing and promotion of drugs, including standards and regulations for direct-to-consumer advertising, off-label promotion, industry-sponsored scientific and educational activities and promotional activities involving the internet.
Drugs may be marketed only for the approved indications and in accordance with the provisions of the approved labeling. Changes to some of the conditions established in an approved application, including changes in indications, labeling, or manufacturing processes or facilities, require submission and FDA approval of a new NDA or NDA supplement before the change can be implemented. A NDA supplement for a new indication typically requires clinical data similar to that in the original application, and the FDA uses the same procedures and actions in reviewing NDA supplements as it does in reviewing NDAs.
Adverse Event Reporting and GMP Compliance
Adverse event reporting and submission of periodic reports is required following FDA approval of a NDA. The FDA also may require post-marketing testing, known as Phase 4 testing, risk minimization action plans, and surveillance to monitor the effects of an approved product, or the FDA may place conditions on an approval that could restrict the distribution or use of the product. In addition, quality-control, drug manufacture, packaging, and labeling procedures must continue to conform to GMP after approval. Drug manufacturers and certain of their subcontractors are required to register their establishments with FDA and certain state agencies. Registration with the FDA subjects entities to periodic unannounced inspections by the FDA, during which the agency inspects manufacturing facilities to assess compliance with current GMP. Accordingly, manufacturers must continue to expend time, money, and effort in the areas of production and quality-control to maintain compliance with current GMP. Regulatory authorities may withdraw product approvals or request product recalls if a company fails to comply with regulatory standards, if it encounters problems following initial marketing, or if previously unrecognized problems are subsequently discovered.
11
Table of Contents
Fast Track Designation
The FDA is required to facilitate the development, and expedite the review, of drugs that are intended for the treatment of a serious or life-threatening condition for which there is no effective treatment and which demonstrate the potential to address unmet medical needs for the condition. Under the fast track program, the sponsor of a new drug candidate may request that the FDA designate the drug candidate for a specific indication as a fast track drug concurrent with, or after, the filing of the IND for the drug candidate. The FDA must determine if the drug candidate qualifies for fast track designation within 60 days of receipt of the sponsor’s request.
In addition to other benefits, such as the ability to use surrogate endpoints and have greater interactions with FDA, the FDA may initiate review of sections of a fast track drug’s NDA before the application is complete. This rolling review is available if the applicant provides, and FDA approves, a schedule for the submission of the remaining information and the applicant pays applicable user fees. However, the FDA’s time period goal for reviewing an application does not begin until the last section of the NDA is submitted. Additionally, the fast track designation may be withdrawn by the FDA if it believes that the designation is no longer supported by data emerging in the clinical trial process.
Accelerated Approval
Under the FDA’s accelerated approval regulations, the FDA may approve a drug for a serious or life-threatening illness that provides meaningful therapeutic benefit to patients over existing treatments based upon a surrogate endpoint that is reasonably likely to predict clinical benefit. In clinical trials, a surrogate endpoint is a measurement of laboratory or clinical signs of a disease or condition that substitutes for a direct measurement of how a patient feels, functions, or survives. Surrogate endpoints can often be measured more easily or more rapidly than clinical endpoints. A drug candidate approved on this basis is subject to rigorous post-marketing compliance requirements, including the completion of Phase 4 or post-approval clinical trials to confirm the effect on the clinical endpoint. Failure to conduct required post-approval studies, or confirm a clinical benefit during post-marketing studies, will allow the FDA to withdraw the drug from the market on an expedited basis. All promotional materials for drug candidates approved under accelerated regulations are subject to prior review by the FDA.
Anti-Kickback, False Claims Laws and the Prescription Drug Marketing Act
In addition to FDA restrictions on marketing of pharmaceutical products, several other types of state and federal laws have been applied to restrict certain marketing practices in the pharmaceutical industry in recent years. These laws include anti-kickback statutes and false claims statutes. The federal healthcare program anti-kickback statute prohibits, among other things, knowingly and willfully offering, paying, soliciting or receiving remuneration to induce; or in return for; purchasing, leasing, ordering or arranging for the purchase, lease or order of any healthcare item or service reimbursable under Medicare, Medicaid, or other federally financed healthcare programs. This statute has been interpreted to apply to arrangements between pharmaceutical manufacturers on the one hand and prescribers, purchasers, and formulary managers on the other. Violations of the anti-kickback statute are punishable by imprisonment, criminal fines, civil monetary penalties, and exclusion from participation in federal healthcare programs. Although there are a number of statutory exemptions and regulatory safe harbors protecting certain common activities from prosecution or other regulatory sanctions, the exemptions and safe harbors are drawn narrowly, and practices that involve remuneration intended to induce prescribing, purchases, or recommendations may be subject to scrutiny if they do not qualify for an exemption or safe harbor.
Federal false claims laws prohibit any person from knowingly presenting, or causing to be presented, a false claim for payment to the federal government, or knowingly making, or causing to be made, a false statement to have a false claim paid. Recently, several pharmaceutical and other healthcare companies have been prosecuted under these laws for allegedly inflating drug prices they report to pricing services, which in turn were used by the
12
Table of Contents
government to set Medicare and Medicaid reimbursement rates, and for allegedly providing free product to customers with the expectation that the customers would bill federal programs for the product. In addition, certain marketing practices, including off-label promotion, may also violate false claims laws. The majority of states also have statutes or regulations similar to the federal anti-kickback law and false claims laws, which apply to items and services reimbursed under Medicaid and other state programs, or, in several states, apply regardless of the payor.
Physician Drug Samples
As part of the sales and marketing process, pharmaceutical companies frequently provide samples of approved drugs to physicians. The Prescription Drug Marketing Act, or the PDMA, imposes requirements and limitations upon the provision of drug samples to physicians, as well as prohibits states from licensing distributors of prescription drugs unless the state licensing program meets certain federal guidelines that include minimum standards for storage, handling, and record keeping. In addition, the PDMA sets forth civil and criminal penalties for violations.
Competition
The biopharmaceutical industry is intensely competitive in general. Furthermore, our business strategy is to target large unmet medical needs, and those markets are even more highly competitive. For example, since 2010 a new second-line chemotherapy drug, Jevtana (cabazitaxel), and a new oral hormonal drug, Zytiga (abiraterone acetate), have received marketing approval in the post-chemotherapy CRPC patient population we studied in our Phase 3 AFFIRM trial of MDV3100, and a new prostate cancer vaccine, Provenge (sipuleucel-T), received marketing approval covering both the post-chemotherapy CRPC population we studied in our Phase 3 AFFIRM trial and the chemotherapy-naïve CRPC population we are studying in our Phase 3 PREVAIL trial. Jevtana (cabazitaxel) and Zytiga (abiraterone acetate) have since acquired substantial shares in the market for treatment of post-chemotherapy patients, which may make it more difficult for us to compete successfully in this market, notwithstanding the positive results from our AFFIRM trial. In addition, enrollment has already been completed in a Phase 3 trial of Zytiga (abiraterone acetate) in the chemotherapy-naïve CRPC population we are studying in our Phase 3 PREVAIL trial. Several other drugs are also in advanced clinical development in both populations. Companies currently marketing, or expected to be marketing in the near future, products that will compete directly with any of our investigational drugs that may receive marketing approval include some of the world’s largest and most experienced pharmaceutical companies, such as Johnson & Johnson and sanofi-aventis. There are also multiple additional small molecule and recombinant protein candidates in development targeting advanced prostate cancer, including compounds already in Phase 3 clinical trials. Most, if not all, of these competing drug development programs are being conducted by pharmaceutical and biotechnology companies with considerably greater financial resources, human resources and experience than ours. Any of our product candidates that receive regulatory approval will face significant competition from both approved drugs and from any of the drugs currently under development that may subsequently be approved. Bases upon which our product candidates would have to compete successfully include efficacy, safety, price and cost-effectiveness. In addition, our product candidates would have to compete against these other drugs with several different categories of decision makers, including physicians, patients, government and private third-party payors, technology assessment groups and patient advocacy organizations. For example, Medicare Part B, which covers infused medicines, generally provides lower patient out-of-pocket expenses and better physician economics than does Medicare Part D, which covers oral medicines. One of our primary competitors in post-docetaxel patients, the infused chemotherapy drug Jevtana (cabazitaxel), is reimbursed under Medicare Part B while MDV3100, should it receive marketing approval and be reimbursed by Medicare, would be covered under Medicare Part D. Even if one of our product candidates is approved, we cannot guarantee that we, Astellas or any of our potential future partners will be able to compete successfully on any of these bases. Any future product candidates that we may subsequently acquire will face similar competitive pressures. If we or our current or potential future partners cannot compete successfully on any of the bases described above, our business will not succeed.
13
Table of Contents
Research and Development Expense
A significant portion of our operating expense is related to research and development, and we intend to maintain our strong commitment to research and development. For the years ended December 31, 2011, 2010 and 2009, we recorded $73.4 million, $72.2 million and $87.7 million, respectively, of research and development expenses. Research and development expenses represented 71%, 76% and 75% of total operating expenses in the years ended December 31, 2011, 2010 and 2009, respectively. More information regarding our research and development activities can be found in the section entitled “Management’s Discussion and Analysis of Financial Condition and Results of Operations” under Item 7 of this Annual Report.
Manufacturing
Our business strategy is to use current GMP compliant contract manufacturers for manufacture of clinical supplies as well as for commercial supplies if required by our commercialization plans, and to transfer manufacturing responsibility to our collaboration partners when possible.
The MDV3100 being used in our completed and ongoing trials was manufactured by cGMP-compliant contract manufacturers. Pursuant to the Astellas Collaboration Agreement, Astellas has agreed to assume commercial manufacturing responsibility for MDV3100, after we complete transfer of those responsibilities to Astellas. Commercial manufacturing processes for MDV3100 have not yet been validated. Based on currently available information, we believe that MDV3100 drug product can be manufactured at commercial scale on a cost-effective basis. However, we caution you that this is a forward-looking statement and that we cannot guarantee that we will be able to complete this work on a timely basis or at all.
Employees
As of December 31, 2011, we had 121 full-time employees, none of which are represented by labor unions or covered by collective bargaining agreements. We consider our relationships with our employees to be good.
Available Information
Our website address is www.medivation.com; however, information found on, or that can be accessed through, our website is not incorporated by reference into this Annual Report on Form 10-K. We file or furnish electronically with the SEC our Annual Report on Form 10-K, quarterly reports on Form 10-Q, current reports on Form 8-K and amendments to those reports filed or furnished pursuant to Section 13(a) or 15(d) of the Exchange Act. We make available free of charge on or through our website copies of these reports as soon as reasonably practicable after we electronically file such material with, or furnish it to, the SEC.
The SEC maintains an internet site that contains reports, proxy and information statements and other information regarding our filings at www.sec.gov. You may also read and copy any of our materials filed with the SEC at the SEC’s Public Reference Room at 100 F Street, NE, Washington, DC 20549. Information regarding the operation of the Public Reference Room can be obtained by calling the SEC at 1-800-SEC-0330.
| Item 1A. | Risk Factors. |
Our business faces significant risks, some of which are set forth below to enable readers to assess, and be appropriately apprised of, many of the risks and uncertainties applicable to the forward-looking statements made in this Annual Report. You should carefully consider these risk factors as each of these risks could adversely affect our business, operating results, cash flows and financial condition. If any of the events or circumstances described in the following risks actually occurs, our business may suffer, the trading price of our common stock could decline and our financial condition or results of operations could be harmed. Given these risks and
14
Table of Contents
uncertainties, you are cautioned not to place undue reliance on forward-looking statements. These risks should be read in conjunction with the other information set forth in this Annual Report. The risks and uncertainties described below are not the only ones we face. Additional risks and uncertainties not presently known to us, or that we currently believe to be immaterial, may also adversely affect our business.
Risks Related to Our Business
We have incurred net losses since inception, expect to incur additional losses in the future as we continue our development activities and may never achieve sustained revenues or profitability. Our only revenue to date has been collaboration revenue under our collaboration agreement with Astellas Pharma Inc., or Astellas, and our former collaboration agreement with Pfizer Inc., or Pfizer. MDV3100 has not received marketing approval, and we do not know when or if it will receive marketing approval or become commercially available. We have incurred losses since inception and expect to continue to incur substantial additional losses for the foreseeable future as we continue to finance clinical and preclinical studies of MDV3100 and potential future product candidates, potential launch costs of MDV3100 should that agent receive marketing approval, and our corporate overhead costs. Our operating losses have had, and will continue to have, an adverse impact on our working capital, total assets and stockholders’ equity. We do not know when or if we will ever generate any additional revenue, including any milestone payments, profit sharing payments or royalty payments under our collaboration agreement with Astellas, or become cash-flow positive, because of the significant uncertainties with respect to our ability to generate product revenue from, and obtain approval from the FDA, or comparable foreign regulatory authorities for, MDV3100 or future product candidates.
Because we depend on financing from third parties for our operations, our business may fail if such financing becomes unavailable or is offered on commercially unreasonable terms. To date, we have financed our operations primarily through private and public offerings of our common stock and from up-front, development milestone and cost-sharing payments received pursuant to our collaboration agreement with Astellas and our former collaboration agreement with Pfizer. As of December 31, 2011 we had cash, cash equivalents and short-term investments totaling $145.1 million available to fund our operations. Based upon our current expectations, we believe our capital resources at December 31, 2011 will be sufficient to fund our currently planned operations for the next 12 months. This estimate is based on a number of assumptions that may prove to be wrong, and we could exhaust our available cash, cash equivalents and short-term investments earlier than presently anticipated. We may be required or choose to seek additional capital within the next 12 months to expand our clinical development activities for MDV3100 based on the positive results of our Phase 3 AFFIRM trial in post-chemotherapy advanced prostate cancer patients, to fund costs of planning for and executing a commercial launch of MDV3100, should it receive marketing approval, if we face challenges or delays in connection with our clinical trials or the potential approval and commercialization of MDV3100, or to maintain minimum cash balances that we deem reasonable and prudent. In addition, we intend to evaluate the capital markets from time to time to determine whether to raise additional capital in the form of equity, convertible debt or otherwise, depending on market conditions relative to our need for funds at such time, and we may seek to raise additional capital within the next 12 months should we conclude that such capital is available on terms that we consider to be in the best interests of our company and our stockholders.
We have incurred losses since inception and expect to continue to incur substantial additional losses for the foreseeable future as we pursue regulatory approval for, and, if approved, commercial launch of, MDV3100 and continue to finance clinical and preclinical studies of MDV3100 and potential future product candidates and our corporate overhead costs. Our future capital requirements will depend on many factors, including without limitation:
| • | potential launch costs of MDV3100, should that agent receive marketing approval for post-chemotherapy patients; |
| • | the timing and magnitude of any potential sales of MDV3100, should that agent receive marketing approval for post-chemotherapy patients; |
15
Table of Contents
| • | whether any changes are made to the scope of our ongoing clinical development activities; |
| • | the scope and results of our and our collaboration partner’s preclinical and clinical trials; |
| • | whether we experience delays in our preclinical and clinical development programs, including potential delays in recruiting, or inability to recruit, patients into our ongoing PREVAIL trial of MDV3100 in pre-chemotherapy advanced prostate cancer as a result of the availability of Zytiga (abiraterone acetate), which was approved by the FDA in April 2011 for post-chemotherapy patients, or other investigational and approved prostate cancer therapies, including MDV3100 itself should that agent be approved to treat post-chemotherapy patients, or slower than anticipated product development; |
| • | whether opportunities to acquire additional product candidates arise and the timing and costs of acquiring and developing those product candidates; |
| • | whether we are able to enter into additional third-party collaborative partnerships to develop and/or commercialize potential future product candidates on terms, including development cost share terms, that are acceptable to us; |
| • | the timing and requirements of, and the costs involved in, conducting studies required to obtain regulatory approvals for MDV3100 or potential future product candidates from the FDA and comparable foreign regulatory agencies; |
| • | the availability of third parties to perform the key development tasks for MDV3100 and potential future product candidates, including conducting preclinical and clinical studies and manufacturing our product candidates to be tested in those studies, and the associated costs of those services; |
| • | expenses associated with the pending purported securities class action lawsuits, as well as any other litigation; and |
| • | the costs involved in preparing, filing, prosecuting, maintaining, defending the validity of and enforcing patent claims and other costs related to patent rights and other intellectual property rights, including litigation costs and the results of such litigation. |
Our current view of the worldwide capital markets is that they are extremely volatile with limited accessibility, and many biotechnology companies have had limited or no success in obtaining funding in this environment. Continuation of this challenging market climate may significantly limit our ability to raise funds, and there can be no assurance we will be able to raise additional funds on acceptable terms or at all. If we are unable to raise additional funds when needed, we could be required to delay, scale back or eliminate some or all of our development programs and other operations. We may seek to raise additional funds through public or private financing or other arrangements. Any additional equity financing would be dilutive to stockholders, and debt financing, if available, may involve restrictive covenants. Our failure to raise capital when needed may harm our business and operating results.
Even if we receive regulatory approval of MDV3100 or any potential future product candidates, which we may never do, we must successfully commercialize such products before we can become profitable. It is not unusual for new pharmaceutical products to be commercially unsuccessful or achieve a lower than expected level of commercial success. We expect to incur substantial expenses associated with our commercialization efforts even prior to obtaining regulatory approval of such product candidates, including MDV3100, as well as thereafter. We may never generate significant revenues and, even if we do generate revenues, the magnitude of those revenues may never be sufficient for us to achieve or sustain profitability.
As we evolve from a company primarily involved in research and development to a company also potentially involved in commercialization, we may encounter difficulties in managing our growth and expanding our operations successfully. Although certain of our employees have commercialization experience, as a company we currently have only limited commercial capabilities. To be prepared to perform our co-promotion activities for MDV3100, should it be approved in the U.S., we will need to expand our organization substantially, including
16
Table of Contents
marketing and sales capabilities or contract with third parties to provide these capabilities for us, which will be expensive and time consuming. Any failure or delay in the development of our internal commercial capabilities would adversely impact the commercialization of MDV3100 in the event it receives regulatory approval. If we are not successful in commercializing MDV3100 or potential future product candidates in the event they receive regulatory approval, our future product revenue will suffer and we may incur significant additional losses.
As our operations expand, we expect that we will need to manage additional relationships with various collaborative partners, suppliers and other third parties. Future growth will impose significant added responsibilities on members of management. Our future financial performance and our ability to commercialize MDV3100 and to compete effectively will depend, in part, on our ability to manage any future growth effectively. To that end, we must be able to manage our development efforts effectively, and hire, train and integrate additional management, administrative and sales and marketing personnel. We may not be able to accomplish these tasks, and our failure to accomplish any of them could prevent us from successfully growing our company.
Our business strategy depends on our ability to identify and acquire additional product candidates which we may never acquire or identify for reasons that may not be in our control, or are otherwise unforeseen or unforeseeable to us. A key component of our business strategy is to diversify our product development risk by identifying and acquiring new product opportunities for development. However, we may not be able to identify promising new technologies. In addition, the competition to acquire promising biomedical technologies is fierce, and many of our competitors are large, multinational pharmaceutical, biotechnology and medical device companies with considerably more financial, development and commercialization resources and experience than we have. Thus, even if we succeed in identifying promising technologies, we may not be able to acquire rights to them on acceptable terms or at all. If we are unable to identify and acquire new technologies, we will be unable to diversify our product risk. We believe that any such failure would have a significant negative impact on our prospects because the risk of failure of any particular development program in the pharmaceutical industry is high.
Because we depend on our management to oversee the execution of development plans for MDV3100 and to identify and acquire promising new product candidates, the loss of any of our executive officers would harm our business. Our future success depends upon the continued services of our executive officers. We are particularly dependent on the continued services of David Hung, M.D., our president and chief executive officer and a member of our board of directors. Dr. Hung identified MDV3100 for acquisition and has primary responsibility for identifying and evaluating other potential product candidates. We believe that Dr. Hung’s services in this capacity would be difficult to replace. None of our executive officers is bound by an employment agreement for any specific term, and they may terminate their employment at any time. In addition, we do not have “key person” life insurance policies covering any of our executive officers. The loss of the services of any of our executive officers could delay the development of MDV3100 and delay or preclude the identification and acquisition of new product candidates, either of which events could harm our business.
Our reliance on third parties for the operation of our business may result in material delays, cost overruns and/or quality deficiencies in our development programs. We rely on outside vendors to perform key product development tasks, such as conducting preclinical and clinical studies and manufacturing our product candidates at appropriate scale for preclinical and clinical trials and, in situations where we are unable to transfer those responsibilities to a corporate partner, for commercial use as well. In order to manage our business successfully, we will need to identify, engage and properly manage qualified external vendors that will perform these development activities. For example, we need to monitor the activities of our vendors closely to ensure that they are performing their tasks correctly, on time, on budget and in compliance with strictly enforced regulatory standards. Our ability to identify and retain key vendors with the requisite knowledge is critical to our business and the failure to do so could negatively impact our business. Because all of our key vendors perform services for other clients in addition to us, we also need to ensure that they are appropriately prioritizing our projects. If we fail to manage our key vendors well, we could incur material delays, cost overruns or quality deficiencies in our development programs, as well as other material disruptions to our business.
17
Table of Contents
Risks Related to Our Product Development Candidates
Pharmaceutical product candidates require extensive, time-consuming and expensive preclinical and clinical testing to establish safety and efficacy. We may never attract additional partners for our technologies or receive marketing approval in any jurisdiction. The research and development of pharmaceuticals is an extremely risky industry. Only a small percentage of product candidates that enter the development process ever receive marketing approval. MDV3100 is not currently approved for sale anywhere in the world, and it may never receive such approval. The process of conducting the preclinical and clinical testing required to establish safety and efficacy and obtain marketing approval is expensive and uncertain and takes many years. If we are unable to complete preclinical or clinical trials of MDV3100 or future product candidates, or if the results of these trials are not satisfactory to convince regulatory authorities or partners of their safety or efficacy, we will not be able to obtain marketing approval or attract additional partners for those product candidates. Furthermore, even if we or our partners are able to obtain marketing approvals for any of our product candidates, those approvals may be for indications that are not as broad as desired or may contain other limitations that would adversely affect our ability to generate revenue from sales of those products. If this occurs, our business will be materially harmed and our ability to generate revenue will be severely impaired.
Because our ongoing Phase 3 PREVAIL trial of MDV3100 in pre-chemotherapy patients has overall survival as a primary endpoint, the availability of approved and/or experimental agents that prolong survival, including the approved hormonal agent Zytiga (abiraterone acetate), the approved chemotherapy agents docetaxel and Jevtana (cabazitaxel), and the approved prostate cancer vaccine Provenge (sipuleucel-T), as well as the potential commercial availability of MDV3100 itself should it be approved for post-chemotherapy patients, may make it more difficult for our PREVAIL trial to succeed or may prevent it from succeeding, and could reduce the magnitude of any potential survival benefit that MDV3100 may demonstrate in PREVAIL even if that trial does succeed. Our ongoing Phase 3 PREVAIL trial in pre-chemotherapy advanced prostate cancer is attempting to demonstrate a statistically significant difference in survival between drug-treated and placebo-treated patients. Overall survival and progression-free survival are the co-primary endpoints in our ongoing PREVAIL trial. Patients participating in our PREVAIL trial may elect to leave the trial and switch to alternative treatments that are, or may in the future become, available to them commercially, such as Zytiga (abiraterone acetate), docetaxel, Jevtana (cabazitaxel), and Provenge (sipuleucel-T), or MDV3100 itself should it be approved for post-chemotherapy patients based on the positive results in the AFFIRM trial. Each of these alternative treatments has demonstrated statistically significant survival benefits of between two and one-half and five months in advanced prostate cancer patients and, except for MDV3100, is commercially available. Patients from our PREVAIL trial may be able to obtain commercial access to these alternative agents notwithstanding the fact that, other than Provenge (sipuleucel-T), none of them has been shown to prolong survival in pre-chemotherapy patients. However, Zytiga (abiraterone acetate) is being studied in an ongoing Phase 3 clinical trial evaluating its potential ability to prolong survival in pre-chemotherapy patients, and positive results from that trial could become available at any time. The survival of any patients who leave our PREVAIL trial to take an alternative treatment will continue to be included in the analysis of the trial. Any survival benefit conferred by these alternative treatments may have a negative impact on the results of our PREVAIL trial, particularly in the case of the one-half of all patients in our PREVAIL trial who were randomized to placebo. Patients in our PREVAIL trial are free to leave the trial at any time, and are free to take any alternative treatment once they have left the trial. We have no ability to control or influence either of these decisions. Use of other alternative life-prolonging treatments by patients leaving our PREVAIL trial could make it more difficult for the trial to succeed, could prevent it from succeeding, and could reduce any potential survival benefit that may be demonstrated even if does succeed. Failure of our PREVAIL trial could have significant negative effects on us, including preventing us from obtaining marketing approval in pre-chemotherapy advanced prostate cancer, which is a far larger commercial opportunity than post-chemotherapy advanced prostate cancer, being required to conduct additional trials, or causing our partner Astellas to elect to terminate our collaboration agreement. Even if our PREVAIL trial succeeds, any negative impact on the survival benefit shown in that trial could reduce or eliminate MDV3100’s ability to compete effectively with other treatments that have shown longer survival benefits.
18
Table of Contents
Enrollment and retention of patients in clinical trials is an expensive and time-consuming process, could be made more difficult or rendered impossible by multiple factors outside our control, including the availability of competing treatments or clinical trials of competing drugs for the same indication and the results of other studies of our product candidates in the same or other indications, and could result in significant delays, cost overruns, or both, in our product development activities, or in the failure of such activities. We may encounter delays in enrolling, or be unable to enroll, a sufficient number of patients to complete any of our clinical trials, and even once enrolled we may be unable to retain a sufficient number of patients to complete any of our trials. Patient enrollment and retention in clinical trials depends on many factors, including the size of the patient population, the nature of the trial protocol, the existing body of safety and efficacy data with respect to the study drug, the number and nature of competing treatments and ongoing clinical trials of competing drugs for the same indication, the proximity of patients to clinical sites and the eligibility criteria for the study. For example, there are multiple ongoing Phase 3 trials competing with our ongoing PREVAIL trial to recruit pre-chemotherapy advanced prostate cancer patients, including an ongoing Phase 3 trial of an investigational agent from Takeda Pharmaceuticals that operates by the same molecular mechanism of action as the approved drug Zytiga (abiraterone acetate). Furthermore, because patients in our PREVAIL trial have a 50% chance of being randomized to placebo, the availability of competing treatments may make it more difficult, or impossible, to complete enrollment in the PREVAIL trial. Such competing treatments include the approved hormonal agent Zytiga (abiraterone acetate), the approved chemotherapy agents Jevtana (cabazitaxel) and docetaxel, and the approved prostate cancer vaccine Provenge (sipuleucel-T), all of which have been shown to prolong overall survival in advanced prostate cancer patients. To date, Zytiga (abiraterone acetate) has been shown to prolong overall survival only in post-chemotherapy advanced prostate cancer patients. However, Zytiga (abiraterone acetate) is being studied in an ongoing Phase 3 trial evaluating that drug’s potential ability to prolong overall survival in pre-chemotherapy advanced prostate cancer patients, the same population being studied in our PREVAIL trial. Should data from the ongoing Zytiga (abiraterone acetate) Phase 3 trial become available that demonstrate an overall survival benefit in pre-chemotherapy patients, such data could make it more difficult or impossible for us to recruit and retain patients in our PREVAIL trial, as those patients may prefer to take Zytiga (abiraterone acetate) rather than participate in our PREVAIL trial. In addition, should MDV3100 receive marketing approval in post-chemotherapy patients based on the positive results in the AFFIRM trial, patients in our PREVAIL trial may elect to leave the study and attempt to receive MDV3100 commercially, despite the fact that MDV3100 has not been shown to prolong overall survival in pre-chemotherapy patients. Such development could make it more difficult or impossible for us to retain patients in our PREVAIL trial. Furthermore, any negative results we may report in clinical trials of MDV3100 or any potential future product candidates may make it difficult or impossible to recruit and retain patients in other clinical studies of that same product candidate. Delays or failures in planned patient enrollment and/or retention may result in increased costs, program delays or both, which could have a harmful effect on our ability to develop MDV3100 or any other product candidates, or could render further development impossible.
Positive results from our AFFIRM trial in post-chemotherapy advanced prostate cancer may not be predictive of results of our PREVAIL trial in pre-chemotherapy advanced prostate cancer or any of our other ongoing and potential future clinical trials of MDV3100. Product candidates in clinical trials, including Phase 3 clinical trials, often fail to show the desired safety and efficacy outcomes despite having progressed successfully through prior stages of preclinical and clinical testing. Even where we achieve positive results in clinical trials, subsequent clinical trials may fail, even if those subsequent trials are designed very similarly to their predecessors. Accordingly, despite the positive results from our Phase 3 AFFIRM trial of MDV3100 in post-chemotherapy advanced prostate cancer, our ongoing Phase 3 PREVAIL trial in pre-chemotherapy advanced prostate cancer, our ongoing Phase 2 TERRAIN trial comparing MDV3100 to bicalutamide, our ongoing Phase 2 study of MDV3100 in hormone-naïve patients, and any other of our ongoing or planned studies of MDV3100 may fail.
MDV3100 and any of our other potential future product candidates, should any of them, receive marketing approval, will face significant competition from other approved products and other products in development. The biopharmaceutical industry is intensely competitive in general. Furthermore, our business strategy is to target large unmet medical needs, and those markets are even more highly competitive. For example, since 2010 a new
19
Table of Contents
second-line chemotherapy drug, Jevtana (cabazitaxel), and a new oral hormonal drug, Zytiga (abiraterone acetate), have received marketing approval in the post-chemotherapy CRPC patient population we studied in our Phase 3 AFFIRM trial of MDV3100, and a new prostate cancer vaccine, Provenge (sipuleucel-T), received marketing approval covering both the post-chemotherapy CRPC population we studied in our Phase 3 AFFIRM trial and the chemotherapy-naïve CRPC population we are studying in our Phase 3 PREVAIL trial. Jevtana (cabazitaxel) and Zytiga (abiraterone acetate) have since acquired substantial shares of the market for treatment of post-chemotherapy patients, which may make it more difficult for us to compete successfully in this market, notwithstanding the positive results from our AFFIRM trial. In addition, enrollment has already been completed in a Phase 3 trial of Zytiga (abiraterone acetate) in the chemotherapy-naïve CRPC population we are studying in our Phase 3 PREVAIL trial. Several other drugs are also in advanced clinical development in both populations. Companies currently marketing, or expected to be marketing in the near future, products that will compete directly with any of our investigational drugs that may receive marketing approval include some of the world’s largest and most experienced pharmaceutical companies, such as Johnson & Johnson and sanofi-aventis. There are also multiple additional small molecule and recombinant protein candidates in development targeting advanced prostate cancer, including compounds already in Phase 3 clinical trials. Most, if not all, of these competing drug development programs are being conducted by pharmaceutical and biotechnology companies with considerably greater financial resources, human resources and experience than ours. Any of our product candidates that receive regulatory approval will face significant competition from both approved drugs and from any of the drugs currently under development that may subsequently be approved. Bases upon which our product candidates would have to compete successfully include efficacy, safety, price and cost-effectiveness. In addition, our product candidates would have to compete against these other drugs with several different categories of decision makers, including physicians, patients, government and private third-party payors, technology assessment groups and patient advocacy organizations. For example, Medicare Part B, which covers infused medicines, generally provides lower patient out-of-pocket expenses and better physician economics than does Medicare Part D, which covers oral medicines. One of our primary competitors in post-docetaxel patients, the infused chemotherapy drug Jevtana (cabazitaxel), is reimbursed under Medicare Part B while MDV3100, should it receive marketing approval and be reimbursed by Medicare, would be covered under Medicare Part D. Even if one of our product candidates is approved, we cannot guarantee that we, Astellas or any of our potential future partners will be able to compete successfully on any of these bases. Any future product candidates that we may subsequently acquire will face similar competitive pressures. If we or our partners cannot compete successfully on any of the bases described above, our business will not succeed.
MDV3100 and any other potential future product candidate that is eventually approved for sale may not be commercially successful if not widely-covered and appropriately reimbursed by third-party payors at levels that produce patient co-payments that are acceptable and affordable to patients. Third-party payors, including public insurers such as Medicare and Medicaid and private insurers, pay for a large share of health care products and services consumed in the United States. In Europe, Canada and other major international markets, third-party payors also pay for a significant portion of health care products and services and many of those countries have nationalized health care systems in which the government pays for all such products and services and must approve product pricing. Even if approved by the FDA and foreign regulatory agencies, MDV3100 and any potential future product candidates are unlikely to achieve commercial success unless they are covered widely by third-party payors and reimbursed at a rate that generates an acceptable commercial return for us and any collaborative partner. In addition, even if they ultimately elect to reimburse our product candidates, most third-party payors will not reimburse 100% of the cost, but rather require a portion of the cost to be borne by the patients themselves. Thus, even if reimbursement is available, the percentage of drug cost required to be borne by the patients may make use of our product candidates financially difficult or impossible for certain patients, which would have a negative impact on sales of our product candidates. It is increasingly difficult to obtain coverage and acceptable reimbursement levels from third-party payors, and we may be unable to achieve these objectives. Achieving coverage and acceptable reimbursement levels typically involves negotiating with individual payors and is a time-consuming and costly process. Moreover, comprehensive health care reform legislation was recently enacted in the United States that substantially changes the way health care is financed by both governmental and private insurers, and significantly impacts the pharmaceutical industry. The new legislation
20
Table of Contents
contains a number of provisions that are expected to impact our business and operations, including those relating to the increased use of comparative effectiveness research on health care products, changes to enrollment in federal healthcare programs, reimbursement changes and fraud and abuse provisions, all of which will impact existing government health care programs and will result in the development of new programs. Many of the details regarding the implementation of this legislation have yet to be determined and implementation may ultimately adversely affect our business. Further, we expect that there will continue to be a number of federal and state proposals to implement government controls over drug product pricing. We are currently unable to predict what additional legislation or regulations, if any, relating to the pharmaceutical industry or third-party payor coverage and reimbursement may be enacted in the future, or what effect the recently enacted federal health care reform legislation or any such additional legislation or regulation will or would have on our business. In addition, we would face competition in such negotiations from other approved drugs against which we compete, which may include other approved drugs marketed by Astellas, and the marketers of such other drugs are likely to be significantly larger than us and therefore enjoy significantly more negotiating leverage with respect to the individual payors than we may have. Our commercial prospects would be further weakened if payors approved coverage for MDV3100 or any potential future product candidates only as second- or later-line treatments, or if they placed any of those product candidates in tiers requiring unacceptably high patient co-payments. Failure to achieve acceptable coverage and reimbursement levels could materially harm our or our partner’s ability to successfully market our product candidates.
We are dependent upon our collaborative relationship with Astellas to further develop, manufacture and commercialize MDV3100. There may be circumstances that delay or prevent Astellas’ ability to develop, manufacture and commercialize MDV3100 or that result in Astellas terminating our agreement with them. In October 2009, we announced that we had entered into a collaboration agreement with Astellas for the development, manufacture and commercialization of MDV3100 to treat prostate cancer. Under the agreement, Astellas is responsible for developing, seeking regulatory approval for, and commercializing MDV3100 outside the United States and, following a transition period, is responsible globally for all manufacture of product for both clinical and commercial purposes. We and Astellas are jointly responsible for developing, seeking regulatory approval for, and commercializing MDV3100 in the United States. We and Astellas share equally the costs, profits and losses arising from development and commercialization of MDV3100 in the United States. For clinical trials useful both in the United States and in Europe or Japan, we will be responsible for one-third of the total costs and Astellas will be responsible for the remaining two-thirds.
We are subject to a number of risks associated with our dependence on our collaborative relationship with Astellas, including:
| • | Astellas’ right to terminate the collaboration agreement with us on limited notice for convenience (subject to certain limitations), or for other reasons specified in the respective collaboration agreement; |
| • | the need for us to identify and secure on commercially reasonable terms the services of third parties to perform key activities currently performed by Astellas in the event that Astellas were to terminate its collaboration with us, including clinical and, if approved, commercial manufacturing, development activities outside of the United States and commercialization activities globally; |
| • | adverse decisions by Astellas regarding the amount and timing of resource expenditures for the development and commercialization of MDV3100; |
| • | decisions by Astellas to prioritize other of its present or future products more highly than MDV3100 for either development and/or commercial purposes; |
| • | possible disagreements as to the timing, nature and extent of our development plans, including clinical trials or regulatory approval strategy; |
| • | changes in key management personnel that are members of the collaboration’s various committees; and |
| • | possible disagreements with Astellas, including those regarding the development and/or commercialization of products, interpretation of the collaboration agreement and ownership of proprietary rights. |
21
Table of Contents
Due to these factors and other possible disagreements with Astellas, we may be delayed or prevented from further developing, manufacturing or potentially commercializing MDV3100 or we may become involved in litigation or arbitration, which would be time consuming and expensive.
If Astellas were to unilaterally terminate our collaborative relationship, we would need to undertake development, manufacturing and marketing activities for MDV3100 solely at our own expense and/or seek one or more other partners for some or all of these activities, worldwide. If we pursued these activities on our own, it would significantly increase our capital and infrastructure requirements, might limit the indications we are able to pursue for MDV3100, and could prevent us from effectively developing and potentially commercializing MDV3100. If we sought to find one or more other pharmaceutical company partners for some or all of these activities, we may not be successful in such efforts, or they may result in collaborations that have us expending greater funds and efforts than our current relationship with Astellas.
We are dependent on the efforts of, and funding by, Astellas for the development of MDV3100. Under the terms of the Astellas Collaboration Agreement, we and Astellas must agree on any changes to the development plan for MDV3100 that is set forth in the agreement. If we and Astellas cannot agree on any such changes, clinical trial progress could be significantly delayed or halted. Subject to certain limitations set forth in the Astellas Collaboration Agreement, Astellas is generally free to terminate the agreement at its discretion on limited notice to us. Similarly, in the event of an uncured material breach of the agreement by us, Astellas may elect to terminate the agreement, in which case all rights to develop and commercialize MDV3100 will revert to us. If Astellas terminates its co-funding of our MDV3100 program, we may be unable to fund the development and potential commercialization costs on our own and may be unable to find another partner, which could force us to raise additional capital or could cause our MDV3100 program to fail. In addition, Astellas is solely responsible for the development and regulatory approval of MDV3100 outside the United States, so we are entirely dependent on Astellas for the successful completion of those activities.
The financial returns to us, if any, under our collaboration agreement with Astellas depend in large part on the achievement of development and commercialization milestones, plus a share of any profits from any product sales in the United States and royalties on any product sales outside of the United States. Therefore, our success, and any associated financial returns to us and our investors, will depend in large part on the performance of Astellas under the Astellas Collaboration Agreement. If Astellas fails to perform or satisfy its obligations to us, the development, potential regulatory approval or commercialization of MDV3100 would be delayed or may not occur and our business and prospects could be materially and adversely affected for that reason.
We are dependent on the efforts of Astellas to market and promote MDV3100 if approved for commercial sale. Under our collaboration with Astellas, we and Astellas have the right to co-promote MDV3100 to all customers in the United States, and Astellas has the sole right to promote MDV3100 to all customers outside of the United States. We are thus partially dependent on Astellas to successfully promote MDV3100 in the United States and solely dependent on Astellas to successfully promote MDV3100 outside of the United States. We have limited ability to direct Astellas in its potential commercialization of MDV3100 in any country, including the United States. If Astellas fails to adequately market and promote MDV3100, whether inside or outside of the United States, we may be unable to obtain any remedy against Astellas. If this were to happen, any sales of MDV3100 may be harmed, which would negatively impact our business, results of operations, cash flows and liquidity.
We are dependent on Astellas to manufacture clinical and commercial requirements of MDV3100, which could result in the delay of clinical trials or regulatory approval or lost sales. Under our agreement with Astellas, after a transition period, Astellas has the primary right and responsibility to manufacture and/or manage the supply of MDV3100 for clinical trials and all commercial requirements. We are in the process of transitioning the manufacturing obligations for MDV3100 to Astellas. Consequently, we are, and expect to remain, dependent on Astellas to supply MDV3100. Astellas may encounter difficulties in production scale-up, including problems involving production yields, quality control and quality assurance, and shortage of qualified personnel. Astellas may not perform as agreed or may default in their obligations to supply clinical trial supplies
22
Table of Contents
and/or commercial product. Astellas may fail to deliver the required quantities of MDV3100 on a timely basis. Any such failure by Astellas could delay our future clinical trials and our applications for regulatory approval, or, if approved for commercial sale, could impair our ability to meet the market demand for MDV3100 and therefore result in decreased sales. If Astellas does not adequately perform, we may be forced to incur additional expenses, delays, or both, to arrange or take responsibility for other third parties to manufacture MDV3100 on our behalf, as we do not have any internal manufacturing capabilities.
If Astellas’ business strategies change, any such changes may adversely affect our collaborative relationship with Astellas. Astellas may change its business strategy. Decisions by Astellas to either reduce or eliminate its participation in the prostate cancer field, to emphasize other competitive agents currently in its portfolio at the expense of MDV3100, or to add additional competitive agents to its portfolio, could reduce its financial incentives to continue to develop, seek regulatory approval for, or potentially commercialize MDV3100. For example, Astellas has partnered with us based in part on Astellas’ desire to use MDV3100 as a component of building a global oncology franchise, which Astellas presently does not have. If Astellas’ strategic objective of building a global oncology franchise were to change, such change could negatively impact any commercial prospects of MDV3100.
Our industry is highly regulated by the FDA and comparable foreign regulatory agencies. We must comply with extensive, strictly enforced regulatory requirements in order to develop and obtain marketing approval for any of our product candidates. Before we, Astellas or any potential future partners can obtain regulatory approval for the sale of our product candidates, our product candidates must be subjected to extensive preclinical and clinical testing to demonstrate their safety and efficacy for humans.
The preclinical and clinical trials of any product candidates that we develop must comply with regulation by numerous federal, state and local government authorities in the United States, principally the FDA, and by similar agencies in other countries. We are required to obtain and maintain an effective IND application to commence human clinical trials in the United States and must obtain and maintain additional regulatory approvals before proceeding to successive phases of our clinical trials. Securing FDA approval requires the submission of extensive preclinical and clinical data and supporting information for each therapeutic indication to establish the product candidate’s safety and efficacy for its intended use. It takes years to complete the testing of a new drug or medical device and development delays and/or failure can occur at any stage of testing. Any of our present and future clinical trials may be delayed or halted due to any of the following:
| • | any preclinical test or clinical trial may fail to produce safety and efficacy results satisfactory to the FDA or foreign regulatory authorities; |
| • | preclinical and clinical data can be interpreted in different ways, which could delay, limit or prevent regulatory approval; |
| • | as we experienced with dimebon, negative or inconclusive results from a preclinical test or clinical trial or adverse medical events during a clinical trial could cause a preclinical study or clinical trial to be repeated or a program to be terminated, even if other studies or trials relating to the program are ongoing or have been completed and were successful; |
| • | the FDA or foreign regulatory authorities can place a clinical hold on a trial if, among other reasons, it finds that patients enrolled in the trial are or would be exposed to an unreasonable and significant risk of illness or injury; |
| • | the FDA might not approve the clinical processes or facilities that we utilize, or the processes or facilities of our consultants, including without limitation the vendors who will be manufacturing drug substance and drug product for us or any potential collaborators; |
| • | any regulatory approval we, Astellas or any potential future collaborators ultimately obtain may be limited or subject to restrictions or post-approval commitments that render the product not commercially viable; and |
23
Table of Contents
| • | we may encounter delays or rejections based on changes in FDA policies or the policies of foreign regulatory authorities during the period in which we develop a product candidate or the period required for review of any final regulatory approval before we are able to market any product candidate. |
In addition, information generated during the clinical trial process is susceptible to varying interpretations that could delay, limit, or prevent regulatory approval at any stage of the approval process. Failure to demonstrate adequately the quality, safety and efficacy of any of our product candidates would delay or prevent regulatory approval of the applicable product candidate. There can be no assurance that if clinical trials are completed, either we or our collaborative partners will submit applications for required authorizations to manufacture or market potential products or that any such application will be reviewed and approved by appropriate regulatory authorities in a timely manner, if at all.
If MDV3100 or any potential future product candidates cannot be manufactured in a cost-effective manner and in compliance with current GMP and other applicable regulatory standards, they will not be commercially successful. All pharmaceutical and medical device products in the United States, Europe and other countries must be manufactured in strict compliance with cGMP and other applicable regulatory standards. Establishing a cGMP-compliant process to manufacture pharmaceutical products involves significant time, cost and uncertainty. Furthermore, in order to be commercially viable, any such process would have to yield product on a cost-effective basis, using raw materials that are commercially available on acceptable terms. We face the risk that our contract manufacturers may have interruptions in raw material supplies, be unable to comply with strictly enforced regulatory requirements, or, for other reasons beyond their or our control, be unable to complete their manufacturing responsibilities on time, on budget, or at all. Under our collaboration agreement with Astellas, Astellas is responsible for all manufacture of MDV3100 for commercial purposes, but we cannot guarantee that Astellas will be able to supply MDV3100 in a timely manner or at all. Furthermore, commercial manufacturing processes have not yet been validated for MDV3100. We thus cannot guarantee that commercial-scale cGMP manufacture of MDV3100 will be possible, on a cost-effective basis or at all, which would materially and adversely affect the value of these programs.
We may be subject to product liability or other litigation, which could result in an inefficient allocation of our critical resources, delay the implementation of our business strategy and, if successful, materially and adversely harm our business and financial condition as a result of the costs of liabilities that may be imposed thereby. Our business exposes us to the risk of product liability claims that is inherent in the development of pharmaceutical products. If MDV3100 or any potential future product candidate harms people, or is alleged to be harmful, we may be subject to costly and damaging product liability claims brought against us by clinical trial participants, consumers, health care providers, corporate partners or others. We have product liability insurance covering our ongoing clinical trials, but do not have insurance for any of our other development activities. If we are unable to obtain insurance at an acceptable cost or otherwise protect against potential product liability claims, we may be exposed to significant litigation costs and liabilities, which may materially and adversely affect our business and financial position. If we are sued for injuries allegedly caused by any of our product candidates, our litigation costs and liability could exceed our total assets and our ability to pay. In addition, we may from time to time become involved in various lawsuits and legal proceedings which arise in the ordinary course of our business. Any litigation to which we are subject, including the purported securities class action lawsuits described in the section entitled “Legal Proceedings” under Part I, Item 3 of this Annual Report, could require significant involvement of our senior management and may divert management’s attention from our business and operations. Litigation costs or an adverse result in any litigation that may arise from time to time may adversely impact our operating results or financial condition.
24
Table of Contents
Risks Related to Intellectual Property
Intellectual property protection for our product candidates is crucial to our business, and is subject to a significant degree of legal risk, particularly in the life sciences industry. The success of our business will depend in part on our ability to obtain and maintain intellectual property protection—primarily patent protection—of our technologies and MDV3100 and any potential future product candidates, as well as successfully defending these patents against third-party challenges. We and our collaborators will only be able to protect our technologies and product candidates from unauthorized use by third parties to the extent that valid and enforceable patents or trade secrets cover them. Furthermore, the degree of future protection of our proprietary rights is uncertain because legal means afford only limited protection and may not adequately protect our rights or permit us or our potential future collaborators to gain or keep our competitive advantage.
The patent positions of life sciences companies can be highly uncertain and involve complex legal and factual questions for which important legal principles remain unresolved. Further, changes in either the patent laws or in interpretations of patent laws in the United States or other countries may diminish the value of our intellectual property rights. Accordingly, we cannot predict the breadth of claims that may be granted or enforced for our patents or for third-party patents that we have licensed. For example:
| • | we or our licensors might not have been the first to make the inventions covered by each of our pending patent applications and issued patents; |
| • | we or our licensors might not have been the first to file patent applications for these inventions; |
| • | others may independently develop similar or alternative technologies or duplicate any of our technologies; |
| • | it is possible that none of our pending patent applications or the pending patent applications of our licensors will result in issued patents; |
| • | our issued patents and future issued patents, or those of our licensors, may not provide a basis for protecting commercially viable products, may not provide us with any competitive advantages, or may be challenged by third parties and invalidated or rendered unenforceable; and |
| • | we may not develop additional proprietary technologies or product candidates that are patentable. |
Our existing and any future patent rights may not adequately protect MDV3100 or any potential future product candidates, which could prevent us from ever generating any revenues or profits. We cannot guarantee that any of our pending or future patent applications will mature into issued patents, or that any of our current or future issued patents will adequately protect MDV3100 or any potential future product candidates from competitors. For example, there is a large body of prior art, including multiple issued patents and published patent applications, disclosing molecules in the same chemical class as our licensed MDV300 series compounds. Since our licensed MDV300 series compounds include approximately 170 specific molecules, we expect that some members of this series may not be patentable in light of this prior art, or may infringe the claims of patents presently issued or issued in the future. Furthermore, we cannot guarantee that any of our present or future issued patents will not be challenged by third parties, or that they will withstand any such challenge. If we are not able to obtain adequate protection for, or defend, the intellectual property position of our technologies and product candidates, then we may not be able to attract collaborators to acquire or partner our development programs. Further, even if we can obtain protection for and defend the intellectual property position of our technologies and product candidates, we or any of our potential future collaborators still may not be able to exclude competitors from developing or marketing competing drugs. Should this occur, we and our potential future collaborators may not generate any revenues or profits from our product candidates or our revenue or profits would be significantly decreased.
We could become subject to litigation or other challenges regarding intellectual property rights, which could divert management attention, cause us to incur significant costs, prevent us from selling or using the challenged technology and/or subject us to competition by lower priced generic products. In recent years, there
25
Table of Contents
has been significant litigation in the United States and elsewhere involving pharmaceutical patents and other intellectual property rights. In particular, generic pharmaceutical manufacturers have been very aggressive in challenging the validity of patents held by proprietary pharmaceutical companies, especially if these patents are commercially significant. If MDV3100 or any future product candidates succeed, we may face similar challenges to our existing or future patents. If a generic pharmaceutical company or other third party were able to successfully invalidate any of our present or future patents, MDV3100 and any potential future product candidates that may ultimately receive marketing approval could face additional competition from lower priced generic products that would result in significant price and revenue erosion and have a significantly negative impact on the commercial viability of the affected product candidate(s).
In the future, we may be a party to litigation to protect our intellectual property or to defend our activities in response to alleged infringement of a third party’s intellectual property. These claims and any resulting lawsuit, if successful, could subject us to significant liability for damages and invalidation, or a narrowing of the scope, of our proprietary rights. These lawsuits, regardless of their success, would likely be time-consuming and expensive to litigate and resolve and would divert management time and attention. Any potential intellectual property litigation also could force us to do one or more of the following:
| • | discontinue our products that use or are covered by the challenged intellectual property; or |
| • | obtain from the owner of the allegedly infringed intellectual property right a license to sell or use the relevant technology, which license may not be available on reasonable terms, or at all. |
If we are forced to take any of these actions, our business may be seriously harmed. Although we carry general liability insurance, our insurance does not cover potential claims of this type.
In addition, our patents and patent applications, or those of our licensors, could face other challenges, such as interference proceedings, opposition proceedings and re-examination proceedings. Any such challenge, if successful, could result in the invalidation of, or in a narrowing of the scope of, any of our patents and patent applications subject to the challenge. Any such challenges, regardless of their success, would likely be time-consuming and expensive to defend and resolve and would divert our management’s time and attention.
We may in the future initiate claims or litigation against third parties for infringement in order to protect our proprietary rights or to determine the scope and validity of our proprietary rights or the proprietary rights of competitors. These claims could result in costly litigation and the diversion of our technical and management personnel and we may not prevail in making these claims.
We rely on license agreements for certain aspects of our product candidates and our technology. We may in the future need to obtain additional licenses of third-party technology that may not be available to us or are available only on commercially unreasonable terms, and which may cause us to operate our business in a more costly or otherwise adverse manner that was not anticipated. We have entered into agreements with third-party commercial and academic institutions to license intellectual property rights and technology for use in our product candidates. For example, we have a license agreement with UCLA pursuant to which we were granted exclusive worldwide rights to certain UCLA patents related to our MDV300 series compounds. Some of these license agreements, including our license agreement with UCLA, contain diligence and milestone-based termination provisions, in which case our failure to meet any agreed upon diligence requirements or milestones may allow the licensor to terminate the agreement. If our licensors terminate our license agreements or if we are unable to maintain the exclusivity of our exclusive license agreements, we may be unable to continue to develop and commercialize MDV3100 or any potential future product candidates based on licensed intellectual property rights and technology.
From time to time we may be required to license technology from additional third parties to develop MDV3100 and any future product candidates. For example, the commercial scale manufacturing processes that we are developing for MDV3100 may require licenses to third-party technology. Should we be required to obtain
26
Table of Contents
licenses to any third-party technology, including any such patents based on biological activities or required to manufacture our product candidates, such licenses may not be available to us on commercially reasonable terms, or at all. The inability to obtain any third-party license required to develop any of our product candidates could cause us to abandon any related development efforts, which could seriously harm our business and operations.
We may become involved in disputes with Astellas or any potential future collaborators over intellectual property ownership, and publications by our research collaborators and scientific advisors could impair our ability to obtain patent protection or protect our proprietary information, which, in either case, could have a significant impact on our business. Inventions discovered under research, material transfer or other such collaboration agreements, including our collaboration agreement with Astellas, may become jointly owned by us and the other party to such agreements in some cases and the exclusive property of either party in other cases. Under some circumstances, it may be difficult to determine who owns a particular invention, or whether it is jointly owned, and disputes could arise regarding ownership of those inventions. These disputes could be costly and time consuming and an unfavorable outcome could have a significant adverse effect on our business if we were not able to protect or license rights to these inventions. In addition, our research collaborators and scientific advisors generally have contractual rights to publish our data and other proprietary information, subject to our prior review. Publications by our research collaborators and scientific advisors containing such information, either with our permission or in contravention of the terms of their agreements with us, may impair our ability to obtain patent protection or protect our proprietary information, which could significantly harm our business.
Trade secrets may not provide adequate protection for our business and technology. We also rely on trade secrets to protect our technology, especially where we believe patent protection is not appropriate or obtainable. However, trade secrets are difficult to protect. While we use reasonable efforts to protect our trade secrets, our or any potential collaborators’ employees, consultants, contractors or scientific and other advisors may unintentionally or willfully disclose our information to competitors. If we were to enforce a claim that a third party had illegally obtained and was using our trade secrets, our enforcement efforts would be expensive and time consuming, and the outcome would be unpredictable. In addition, courts outside the United States are sometimes less willing to protect trade secrets. Moreover, if our competitors independently develop equivalent knowledge, methods or know-how, it will be more difficult or impossible for us to enforce our rights and our business could be harmed.
Risks Related to Ownership of Our Common Stock
We have been named as a defendant in a purported securities class action lawsuit. This lawsuit could result in substantial damages and may divert management’s time and attention from our business and operations. In March 2010, the first of several putative securities class action lawsuits was commenced in the U.S. District Court for the Northern District of California, naming as defendants us and certain of our officers. The lawsuits are largely identical and allege violations of the Securities Exchange Act of 1934, as amended. The plaintiffs allege, among other things, that we disseminated false and misleading statements about the effectiveness of dimebon for the treatment of Alzheimer’s disease. The plaintiffs purport to seek damages, an award of their costs and injunctive relief on behalf of a class of stockholders who purchased or otherwise acquired our common stock between September 21, 2006 and March 2, 2010. The actions were consolidated in September 2010 and, in April 2011, the court entered an order appointing Catoosa Fund, L.P. and its attorneys as lead plaintiff and lead counsel. Thereafter, the lead plaintiff filed a consolidated amended complaint, which was dismissed without prejudice as to all defendants in August 2011. The lead plaintiff filed a second amended complaint in November 2011. In January 2012, we filed a motion to dismiss the second amended complaint, which is scheduled to be heard by the Court on March 16, 2012.
Our management believes that we have meritorious defenses and intends to defend this lawsuit vigorously. However, this lawsuit is subject to inherent uncertainties, and the actual cost will depend upon many unknown factors. The outcome of the litigation is necessarily uncertain, we could be forced to expend significant resources in the defense of the suit and we may not prevail. Monitoring and defending against legal actions is time
27
Table of Contents
consuming for our management and detracts from our ability to fully focus our internal resources on our business activities. In addition, we may incur substantial legal fees and costs in connection with the litigation and, although we believe the Company is entitled to coverage under the relevant insurance policies, subject to a $350,000 retention, coverage could be denied or prove to be insufficient. We are not currently able to estimate the possible cost to us from this matter, as this lawsuit is currently at an early stage and we cannot be certain how long it may take to resolve this matter or the possible amount of any damages that we may be required to pay. We have not established any reserves for any potential liability relating to this lawsuit. It is possible that we could, in the future, incur judgments or enter into settlements of claims for monetary damages. A decision adverse to our interests on these actions could result in the payment of substantial damages, or possibly fines, and could have a material adverse effect on our cash flow, results of operations and financial position. In addition, the uncertainty of the currently pending litigation could lead to more volatility in our stock price.
Our stock price may be volatile, and our stockholders’ investment in our stock could decline in value. The market prices for our securities and those of other life sciences companies have been highly volatile and may continue to be highly volatile in the future. The following factors, in addition to other risk factors described in this Annual Report, may have a significant impact on the market price of our common stock:
| • | the receipt or failure to receive the additional funding necessary to conduct our business; |
| • | the progress and results of preclinical studies and clinical trials of our product candidates conducted by us, Astellas or any future collaborative partners or licensees, if any, and any delays in enrolling a sufficient number of patients to complete clinical trials of our product candidates; |
| • | the announcement by our competitors of results from clinical trials of their products or product candidates; |
| • | selling by existing stockholders and short-sellers; |
| • | announcements of technological innovations or new commercial products by our competitors or us; |
| • | developments concerning proprietary rights, including patents; |
| • | developments concerning our collaboration with Astellas or any future collaborations; |
| • | publicity regarding us, our product candidates or those of our competitors, including research reports published by securities analysts; |
| • | regulatory developments in the United States and foreign countries; |
| • | litigation, including the purported securities class action lawsuits pending against us and certain of our officers; |
| • | economic and other external factors or other disaster or crisis; and |
| • | period-to-period fluctuations in financial results. |
We do not intend to pay dividends on our common stock for the foreseeable future. We do not expect for the foreseeable future to pay dividends on our common stock. Any future determination to pay dividends on or repurchase shares of our common stock will be at the discretion of our board of directors and will depend upon, among other factors, our success in completing sales or partnerships of our programs, our results of operations, financial condition, capital requirements, contractual restrictions and applicable law.
Our principal stockholders exert substantial influence over us and may exercise their control in a manner adverse to your interests. Certain stockholders and their affiliates own a substantial amount of our outstanding common stock. These stockholders may have the power to direct our affairs and be able to determine the outcome of certain matters submitted to stockholders for approval. Because a limited number of persons controls us, transactions could be difficult or impossible to complete without the support of those persons. Subject to applicable law, it is possible that these persons will exercise control over us in a manner adverse to your interests.
28
Table of Contents
Provisions of our charter documents, our stockholder rights plan and Delaware law could make it more difficult for a third party to acquire us, even if the offer may be considered beneficial by our stockholders. Provisions of the Delaware General Corporation Law could discourage potential acquisition proposals and could delay, deter or prevent a change in control. The anti-takeover provisions of the Delaware General Corporation Law impose various impediments to the ability of a third party to acquire control of us, even if a change in control would be beneficial to our existing stockholders. Specifically, Section 203 of the Delaware General Corporation Law, unless its application has been waived, provides certain default anti-takeover protections in connection with transactions between us and an “interested stockholder.” Generally, Section 203 prohibits stockholders who, alone or together with their affiliates and associates, own more than 15% of the subject company from engaging in certain business combinations for a period of three years following the date that the stockholder became an interested stockholder of such subject company without approval of the board or the vote of two-thirds of the shares held by the independent stockholders. Our board of directors has also adopted a stockholder rights plan, or “poison pill,” which would significantly dilute the ownership of a hostile acquirer. Additionally, provisions of our amended and restated certificate of incorporation and bylaws could deter, delay or prevent a third party from acquiring us, even if doing so would benefit our stockholders, including without limitation, the authority of the board of directors to issue, without stockholder approval, preferred stock with such terms as the board of directors may determine.
Future sales of our common stock in the public market could cause our stock price to fall. Sales of a substantial number of shares of our common stock in the public market that were previously restricted from sale, or the perception that these sales might occur, could depress the market price of our common stock and could impair our ability to raise capital through the sale of additional equity securities. In the event that we do raise capital through the sale of additional equity securities or debt convertible into equity securities, the dilution represented by the additional shares of our equity securities in the public market could cause our stock price to fall, in which case investors may not be able to sell their shares of our equity securities at a price equal to or above the price they paid to acquire them.
| Item 1B. | Unresolved Staff Comments. |
None.
| Item 2. | Properties. |
Our corporate headquarters are currently located at 201 Spear Street, San Francisco, California, where we lease approximately 34,000 square feet of office space pursuant to lease agreements that expire in July 2012 and May 2013.
In December 2011, we entered into a lease agreement for approximately 57,000 square feet of office space located at 525 Market Street, San Francisco, California, which is intended to serve as our future corporate headquarters following construction and build-out. The term of the lease is for seven years, with an anticipated commencement date of June 2012. We have an option to extend the term of the lease for an additional five years.
In February 2012, we entered into a lease agreement for 15,336 square feet of office space located in Oakbrook Terrace, Illinois, which is intended to serve as our commercial headquarters following construction and build-out. We selected this location for our commercial headquarters based on its proximity to our collaboration partner, Astellas. The term of the lease is seven years and seven months, with an anticipated commencement date of May 2012.
We believe our properties are adequately maintained and suitable for their intended use. We continually evaluate our properties and believe that our current facilities plus any planned expansion are generally sufficient to meet our expected needs and near-term growth.
29
Table of Contents
| Item 3. | Legal Proceedings. |
In March 2010, the first of several putative securities class action lawsuits was commenced in the U.S. District Court for the Northern District of California, naming as defendants us and certain of our officers. The lawsuits are largely identical and allege violations of the Securities Exchange Act of 1934, as amended. The plaintiffs allege among other things that we disseminated false and misleading statements about the effectiveness of dimebon for the treatment of Alzheimer’s disease. The plaintiffs purport to seek damages, an award of their costs and injunctive relief on behalf of a class of stockholders who purchased or otherwise acquired our common stock between September 21, 2006 and March 2, 2010. The actions were consolidated in September 2010, and, in April 2011 the court entered an order appointing Catoosa Fund, L.P. and its attorneys as lead plaintiff and lead counsel. Thereafter, the lead plaintiff filed a consolidated, amended complaint, which was dismissed without prejudice as to all defendants in August 2011. The lead plaintiff filed a second amended complaint in November 2011. In January 2012, we filed a motion to dismiss the second amended complaint, which is scheduled to be heard by the Court on March 16, 2012.
Our management believes that we have meritorious defenses and intends to defend this lawsuit vigorously. However, this lawsuit is subject to inherent uncertainties, the actual cost may be significant, and we may not prevail. We believe we are entitled to coverage under our relevant insurance policies, subject to a $350,000 retention, but coverage could be denied or prove to be insufficient.
| Item 4. | Mine Safety Disclosures. |
None.
30
Table of Contents
PART II
| Item 5. | Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities. |
Market Information
Our common stock, par value $0.01, is listed on the NASDAQ Global Market under the symbol “MDVN.” The following table sets forth on a per share basis the high and low intraday sales prices of our common stock as reported by the NASDAQ Global Market:
| High | Low | |||||||
| 2011: |
||||||||
| Quarter ended March 31, 2011 |
$ | 19.84 | $ | 13.27 | ||||
| Quarter ended June 30, 2011 |
$ | 25.50 | $ | 18.71 | ||||
| Quarter ended September 30, 2011 |
$ | 22.97 | $ | 14.32 | ||||
| Quarter ended December 31, 2011 |
$ | 49.66 | $ | 15.51 | ||||
| 2010: |
||||||||
| Quarter ended March 31, 2010 |
$ | 40.49 | $ | 10.47 | ||||
| Quarter ended June 30, 2010 |
$ | 12.25 | $ | 8.79 | ||||
| Quarter ended September 30, 2010 |
$ | 13.13 | $ | 8.43 | ||||
| Quarter ended December 31, 2010 |
$ | 16.68 | $ | 10.96 | ||||
Stockholders
As of the close of business on February 22, 2012, there were 23 stockholders of record of our common stock and the last reported sales price per share of our common stock was $65.08. The number of stockholders of record is based upon the actual number of stockholders registered at such date and does not include holders of shares in “street names” or persons, partnerships, associates, or corporations, or other entities identified in security listings maintained by depositories.
Dividends
We have never paid our stockholders cash dividends and we do not anticipate paying any cash dividends in the foreseeable future as we intend to retain all of our cash for use in our business. Any future determination to pay dividends will be at the discretion of our Board of Directors, and will depend on a number of factors, including but not limited to any outstanding indebtedness, earnings, capital requirements, financial condition and future prospects, and applicable Delaware law.
31
Table of Contents
Performance Graph
We have presented below the cumulative total return to our stockholders during the period from December 31, 2006 through December 31, 2011 in comparison to the cumulative total return of the NASDAQ Composite Index and the NASDAQ Biotechnology Index. All values assume a $100 initial investment and the reinvestment of the full amount of all dividends and are calculated as of the last stock trading day of each year. The comparisons are based on historical data and are not indicative of, nor intended to forecast, the future performance of our common stock.
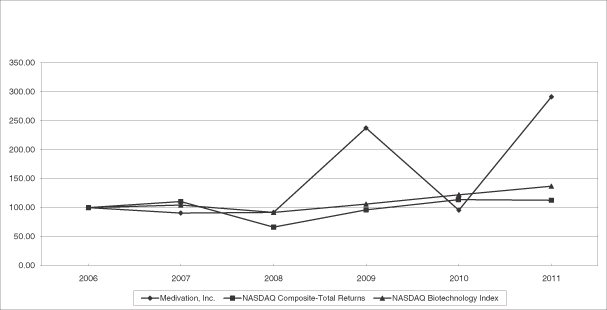
| 2006 | 2007 | 2008 | 2009 | 2010 | 2011 | |||||||||||||||||||
| Medivation, Inc. |
$ | 100 | $ | 91.02 | $ | 92.10 | $ | 237.99 | $ | 95.89 | $ | 291.46 | ||||||||||||
| NASDAQ Composite Index |
$ | 100 | $ | 110.65 | $ | 66.42 | $ | 96.54 | $ | 114.07 | $ | 113.17 | ||||||||||||
| NASDAQ Biotechnology Index |
$ | 100 | $ | 104.64 | $ | 91.77 | $ | 106.42 | $ | 122.62 | $ | 137.42 | ||||||||||||
The information under “Performance Graph” is not deemed to be “soliciting material” or “filed” with the Securities and Exchange Commission or subject to Regulation 14A or 14C, or to the liabilities of Section 18 of the Securities Exchange Act of 1934, as amended, and is not to be incorporated by reference in any filing of Medivation under the Securities Act of 1933, as amended, or the Securities Exchange Act of 1934, as amended, whether made before or after the date of this Annual Report on Form 10-K and irrespective of any general incorporation language in those filings.
Recent Sales of Unregistered Securities
None.
32
Table of Contents
| Item 6. | Selected Financial Data. |
The following is a summary of our historical consolidated financial data for the years ended and on the dates indicated below. The historical consolidated financial data for the years ended December 31, 2011, 2010 and 2009 and as of December 31, 2011 and 2010 have been derived from our audited consolidated financial statements included in Item 15 of this Annual Report. The historical financial data for the years ended December 31, 2008 and 2007 and as of December 31, 2009, 2008 and 2007 have been derived from our audited consolidated financial statements not included in this Annual Report. The information below is not necessarily indicative of results of future operations, and should be read in conjunction with Item 7, “Management’s Discussion and Analysis of Financial Condition and Results of Operations,” and the consolidated financial statements and related notes thereto included in Item 15 of this Annual Report to fully understand factors that may affect the comparability of the information presented below.
| Years Ended December 31, | ||||||||||||||||||||
| 2011 | 2010 | 2009 | 2008 | 2007 | ||||||||||||||||
| (in thousands, except per share data) | ||||||||||||||||||||
| Consolidated Statements of Operations Data: |
||||||||||||||||||||
| Collaboration revenue |
$ | 60,389 | $ | 62,508 | $ | 69,254 | $ | 12,578 | $ | — | ||||||||||
| Operating expenses: |
||||||||||||||||||||
| Research and development |
73,432 | 72,228 | 87,728 | 54,895 | 23,399 | |||||||||||||||
| Selling, general and administrative |
29,887 | 23,005 | 28,983 | 21,865 | 10,364 | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Total operating expenses |
103,319 | 95,233 | 116,711 | 76,760 | 33,763 | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Loss from operations |
(42,930 | ) | (32,725 | ) | (47,457 | ) | (64,182 | ) | (33,763 | ) | ||||||||||
| Other (expense) income, net |
(242 | ) | 260 | 976 | 1,712 | 2,022 | ||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Net loss before income tax (benefit) expense |
(43,172 | ) | (32,465 | ) | (46,481 | ) | (62,470 | ) | (31,741 | ) | ||||||||||
| Income tax (benefit) expense |
(4,331 | ) | 1,572 | 8,272 | (10 | ) | 2 | |||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Net loss |
$ | (38,841 | ) | $ | (34,037 | ) | $ | (54,753 | ) | $ | (62,460 | ) | $ | (31,743 | ) | |||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Basic and diluted net loss per common share |
$ | (1.11 | ) | $ | (0.99 | ) | $ | (1.71 | ) | $ | (2.12 | ) | $ | (1.14 | ) | |||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Weighted-average common shares used in the calculation of basic and diluted net loss per common share |
34,960 | 34,290 | 32,094 | 29,478 | 27,932 | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| December 31, | ||||||||||||||||||||
| 2011 | 2010 | 2009 | 2008 | 2007 | ||||||||||||||||
| (in thousands) | ||||||||||||||||||||
| Consolidated Balance Sheet Data: |
||||||||||||||||||||
| Cash and cash equivalents |
$ | 70,136 | $ | 107,717 | $ | 57,463 | $ | 71,454 | $ | 43,258 | ||||||||||
| Short-term investments |
74,996 | 100,039 | 220,781 | 149,968 | — | |||||||||||||||
| Working capital |
78,555 | 148,037 | 189,813 | 149,584 | 40,214 | |||||||||||||||
| Total assets |
175,117 | 239,603 | 296,690 | 229,272 | 45,596 | |||||||||||||||
| Deferred revenue |
143,271 | 200,660 | 253,168 | 212,423 | — | |||||||||||||||
| Accumulated deficit |
(250,291 | ) | (211,450 | ) | (177,413 | ) | (122,660 | ) | (60,200 | ) | ||||||||||
| Total stockholders’ equity |
1,321 | 7,684 | 25,274 | 3,408 | 41,058 | |||||||||||||||
| Item 7. | Management’s Discussion and Analysis of Financial Condition and Results of Operations. |
The following discussion and analysis should be read in conjunction with our audited consolidated financial statements and notes thereto for the fiscal year ended December 31, 2011 included elsewhere in this Annual Report. The following discussion and analysis contains “forward-looking statements” within the meaning of Section 27A of the Securities Act and Section 21E of the Exchange Act. We intend that these forward-looking
33
Table of Contents
statements be subject to the safe harbors created by those provisions. Forward-looking statements are generally written in the future tense and/or are preceded by words such as “may,” “should,” “forecast,” “could,” “expect,” “suggest,” “believe,” “anticipate,” “intend,” “plan,” or other similar words, or the negatives of such terms or other variations on such terms or comparable terminology. The forward-looking statements contained in this Annual Report involve a number of risks and uncertainties, many of which are outside of our control. Factors that could cause actual results to differ materially from projected results include, but are not limited to, those discussed in “Risk Factors” included elsewhere in this Annual Report. Readers are expressly advised to review and consider those Risk Factors, which include risks associated with (1) the effects of competing and alternative treatments on patient enrollment and retention in and the results of our ongoing and planned clinical trials, including our ongoing PREVAIL trial, (2) our ability to successfully launch and obtain reimbursement on acceptable terms for MDV3100, and to compete successfully against approved agents proven to extend life, should MDV3100 receive marketing approval for post-chemotherapy advanced prostate cancer patients; (3) our ability to successfully conduct clinical and preclinical trials for our product candidates, (4) our ability to obtain and maintain required regulatory approvals to develop and market our product candidates, (5) our ability to raise additional capital on favorable terms, (6) our ability to execute our development plan on time and on budget, (7) our ability to obtain commercial partners and maintain our relationships with our current and/or potential partners, (8) our ability, whether alone or with commercial partners, to successfully commercialize any of our product candidates that may be approved for sale, and (9) our ability to identify and obtain additional product candidates. Although we believe that the assumptions underlying the forward-looking statements contained in this Annual Report are reasonable, any of the assumptions could be inaccurate, and therefore there can be no assurance that such statements will be accurate. In light of the significant uncertainties inherent in the forward-looking statements included herein, the inclusion of such information should not be regarded as a representation by us or any other person that the results or conditions described in such statements or our objectives and plans will be achieved. Furthermore, past performance in operations and share price is not necessarily indicative of future performance. We disclaim any intention or obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise.
Overview
We are a biopharmaceutical company focused on the rapid development of novel small molecule drugs to treat serious diseases for which there are limited treatment options. Together with our collaboration partner, Astellas Pharma, Inc., or Astellas, we are developing MDV3100 for multiple stages of advanced prostate cancer. We have successfully completed the Phase 3 AFFIRM trial in the latest stage patients—those who have already failed docetaxel-based chemotherapy—and we are conducting an additional Phase 3 trial and two Phase 2 trials in men with earlier stages of advanced prostate cancer. Based on the positive results of the AFFIRM trial, we have exercised our right under our collaboration agreement with Astellas, or the Astellas Collaboration Agreement, to co-promote MDV3100 in the U.S., should it receive marketing approval. We and Astellas expect to file applications for marketing approval for MDV3100 both in the U.S. and in Europe in 2012. MDV3100 has received “Fast Track” designation from the FDA for the post-docetaxel indication.
In October 2009, we entered into the Astellas Collaboration Agreement. Under the terms of the agreement, we and Astellas share equally the costs of developing and commercializing MDV3100 for the United States market (subject to the exceptions noted below), and we and Astellas will share equally profits (or losses) resulting from the commercialization of MDV3100 in the United States. Costs of clinical trials supporting development in both the United States and in either Europe or Japan are borne two-thirds by Astellas and one-third by us. Both we and Astellas will be responsible for all costs incurred in fielding and supporting our respective MDV3100 sales forces, and each of us will be entitled to receive a fee for each detail made by our respective sales forces. Outside the United States, Astellas will bear all development and commercialization costs and will pay us tiered, double-digit royalties on the aggregate net sales of MDV3100.
In January 2012, we reported negative top line results from our Phase 3 CONCERT trial of our product candidate dimebon in patients with mild-to-moderate Alzheimer’s disease. We previously had reported negative
34
Table of Contents
top line results from our Phase 3 CONNECTION trial of dimebon in patients with mild-to-moderate Alzheimer’s disease and our Phase 3 HORIZON trial of dimebon in patients with Huntington disease. In January 2012, Pfizer, Inc., or Pfizer, exercised its right to terminate our collaboration agreement for the development and commercialization of dimebon and we and Pfizer discontinued development of dimebon for all indications. During the ensuing 180 days, we and Pfizer will work together to wind down our respective remaining collaboration activities.
Our MDV300 Series Prostate Cancer Program
We have obtained an exclusive, worldwide commercial license to a series of novel small molecules, referred to as the MDV300 series compounds. Our lead development candidate from the MDV300 series is a molecule we refer to as MDV3100. A description of our completed and ongoing clinical trials is included below.
Completed Clinical Trials
Phase 3 AFFIRM Trial
In November 2011, we reported positive results from a planned interim analysis of the AFFIRM trial, a randomized, double-blind Phase 3 trial evaluating MDV3100 (160 mg once daily) versus placebo in 1,199 patients with advanced prostate cancer who had previously failed docetaxel-based chemotherapy. The primary endpoint of the AFFIRM trial was overall survival. The Independent Data Monitoring Committee, or IDMC, overseeing the AFFIRM trial determined that MDV3100 demonstrated a clinically meaningful and statistically significant (p<0.0001) improvement in overall survival compared to placebo, and as a result recommended that the study be stopped early and men who received placebo be offered MDV3100. The IDMC informed us that 1) MDV3100 produced a 4.8-month advantage in median survival compared to placebo; 2) the estimated median survival for men treated with MDV3100 was 18.4 months compared to 13.6 months for men treated with placebo; and 3) MDV3100 provided a 37% reduction in risk of death compared to placebo (hazard ratio = 0.631). The IDMC further determined, considering the observed safety profile, that MDV3100 demonstrated a favorable risk-to-benefit ratio sufficient to stop the study.
In February 2012, we reported further results from the AFFIRM trial at the American Society of Clinical Oncology’s 2012 Genitourinary Cancers Symposium, or ASCO GU. In addition to confirming the results reported in November 2011, we reported that MDV3100 had achieved all of the trial’s secondary endpoints with strong statistical significance and was well tolerated. As compared to patients taking placebo, MDV3100 patients in the AFFIRM trial experienced longer median radiographic progression-free survival (8.3 versus 2.9 months; p<0.0001; hazard ratio = 0.404), a higher soft tissue response rate (28.9% versus 3.8%; p<0.0001), and longer median time to prostate-specific antigen, or PSA, progression (8.3 versus 3.0 months; p<0.0001; hazard ratio = 0.249). PSA declines of 50% or greater were more common in the MDV3100 group than in the placebo group (54.0% versus 1.5%; p<0.0001), as were PSA declines of 90% or greater (24.8% versus 0.9%; p<0.0001).
MDV3100 was well tolerated in the AFFIRM trial. Common side effects included fatigue, diarrhea and hot flush. Serious adverse events, adverse events causing patients to stop treatment, and adverse events causing death all were lower in the MDV3100 group than in the placebo group. Serious side effects of interest were fatigue (6.3% in the MDV3100 group versus 7.3% in the placebo group), cardiac disorders (0.9% versus 2.0%) including myocardial infarction (0.3% versus 0.5%), seizure (0.6% versus 0.0%) and liver function test abnormalities (0.4% versus 0.8%). The period during which AFFIRM patients were followed for safety issues was more than twice as long for MDV3100 patients (9.3 months) as compared to placebo patients (4.0 months).
The FDA has granted Fast Track designation for the development of MDV3100 in the post-chemotherapy indication, a designation that is reserved for development programs that the FDA determines to be for a life-threatening condition with unmet medical need. Receipt of Fast Track designation enables us to request that the FDA grant us priority review for our anticipated new drug application, or NDA, in post-chemotherapy patients. In considering requests for priority review, the FDA applies the same standard it uses to award Fast Track designation. We plan to hold a pre-NDA meeting with the FDA in early 2012. We and Astellas expect to file
35
Table of Contents
both an NDA with the FDA and a marketing authorization application, or MAA, with the European Medicines Agency in 2012 seeking approval to market MDV3100 for post-docetaxel advanced prostate cancer in the U.S. and Europe, respectively.
Based on the positive results from the AFFIRM trial, we elected to exercise our right under the Astellas Collaboration Agreement to co-promote MDV3100 in the U.S. market. Should MDV3100 receive marketing approval, we will provide 50% of the sales and medical affairs support for MDV3100 in the U.S. market.
Phase 1-2 Trial
In December 2008, we completed enrollment in an open-label Phase 1-2 clinical trial of MDV3100 in patients with castration resistant prostate cancer, or CRPC. We enrolled 140 patients in seven dose groups, ranging from 30 mg/day to 600 mg/day. Of the 140 patients, 75 had previously failed docetaxel-based chemotherapy and 65 were chemotherapy-naïve. All 140 patients also had failed at least one line of prior hormonal therapy. The study endpoints include safety, tolerability, pharmacokinetics, circulating tumor cell, or CTC, counts, PSA levels, radiographic change in soft tissue and bony metastases, and time to progression.
Data from this trial were published in The Lancet in 2010. These data showed that MDV3100 consistently demonstrated anti-tumor activity across endpoints, as evaluated by reductions in PSA levels, radiographic findings and CTC counts. This activity was consistently stronger in the earlier stage (i.e. chemotherapy naïve) as compared to the later stage (i.e., post-chemotherapy) patients. Key efficacy data as reported in The Lancet were as follows:
| Chemotherapy-Naïve Patients | Post-Chemotherapy Patients | |||||||
| PSA decline ³ 50% from baseline |
62 | % | 51 | % | ||||
| Radiographic control: soft-tissue lesions (partial response or stable disease) |
80 | % | 65 | % | ||||
| Radiographic control: bony lesions (stable disease) |
63 | % | 51 | % | ||||
| CTC conversion from “poor prognosis” to “good prognosis” |
75 | % | 37 | % | ||||
In February 2011, we presented new long-term follow-up data covering all 140 patients enrolled in the trial at 2011ASCO GU. PSA progression data reported at 2011 ASCO GU were calculated using three distinct reporting criteria: the criteria specified in the Phase 1-2 trial protocol; the most recent published PSA reporting consensus criteria (the Prostate Cancer Clinical Trials Working Group 2, or PCWG2, criteria); and an older commonly used reporting method (the Prostate-Specific Antigen Working Group 1, or PSAWG1, criteria). Key efficacy data as presented at 2011 ASCO GU were as follows:
| Chemotherapy Naïve Patients | Post-Chemotherapy Patients | |||
| Median time to PSA progression (per protocol criteria) |
Not reached | 316 days (45 weeks) | ||
| Median time to PSA progression (per PCWG2 criteria) |
281 days (40 weeks) |
148 days (21 weeks) | ||
| Median time to PSA progression (per PSAWG1 criteria) |
420 days (60 weeks)* 812 days (116 weeks)** |
166 days (24 weeks) | ||
| Median time to radiographic progression |
394 days (56 weeks) |
173 days (25 weeks) |
| * | All chemotherapy-naïve patients |
| ** | Subpopulation of chemotherapy-naïve patients who were also ketoconazole-naïve |
36
Table of Contents
MDV3100 was well tolerated in this trial at doses up to and including 240 mg/day. The most frequently reported adverse event was fatigue. Seizures were observed in two patients, one each at doses of 600 and 360 mg/day. Both patients were taking concomitant medications that can cause seizures. A possible but unwitnessed seizure was reported in a patient taking a dose of 480 mg/day.
Ongoing Clinical Trials
Phase 3 PREVAIL Trial
PREVAIL is a randomized, double-blind, placebo-controlled Phase 3 trial evaluating MDV3100 (160 mg once daily) versus placebo in approximately 1,700 patients with advanced prostate cancer who have not previously been treated with chemotherapy. The co-primary endpoints are progression-free survival and overall survival. We began enrollment in the PREVAIL trial in September 2010.
Phase 2 TERRAIN Trial
TERRAIN is a randomized, double-blind Phase 2 trial evaluating MDV3100 versus bicalutamide, the leading marketed anti-androgen drug, in approximately 370 advanced prostate cancer patients who have progressed following medical castration with a luteinizing hormone releasing hormone analog drug or surgical castration. The primary endpoint is progression-free survival. We began enrollment in the TERRAIN trial in March 2011.
Phase 2 Hormone-Naïve Trial
In May 2011, we initiated a Phase 2 trial evaluating MDV3100 in approximately 60 patients with advanced prostate cancer who have not had any previous hormonal treatment. This is the first trial to examine the effects of MDV3100 in the earlier-stage population of advanced prostate cancer patients who have not yet undergone medical or surgical castration. The primary endpoint in the trial is PSA response.
Financial History
We have not generated any revenue from product sales to date, and we may never generate any revenue from product sales. We have funded our operations primarily through private and public offerings of our common stock and from the up-front, development milestone and cost-sharing payments under our collaboration agreements.
We entered into a collaboration agreement with Astellas in October 2009, pursuant to which we received a non-refundable up-front cash payment of $110.0 million in the fourth quarter of 2009. We subsequently received development milestone payments of $10.0 million and $3.0 million in the fourth quarter of 2010 and the second quarter of 2011, respectively. We recorded development and commercialization cost-sharing payments from Astellas totaling $43.8 million, $34.6 million and $2.9 million during the years ended December 31, 2011, 2010 and 2009, respectively. Additional information regarding the Astellas Collaboration Agreement is included in the section titled, “Management’s Discussion and Analysis of Financial Condition and Results of Operations—Liquidity and Capital Resources—Astellas Collaboration Agreement.”
We entered into a collaboration agreement with Pfizer in October 2008, pursuant to which we received a non-refundable up-front cash payment of $225.0 million in the fourth quarter of 2008. We recorded development and commercialization cost-sharing payments from Pfizer totaling $12.4 million, $28.1 million and $19.7 million during the years ended December 31, 2011, 2010 and 2009, respectively. In January 2012, we reported negative top line results from our Phase 3 CONCERT trial of dimebon in patients with mild-to-moderate Alzheimer’s disease. We previously had reported negative top line results from our Phase 3 CONNECTION trial of dimebon in patients with mild-to-moderate Alzheimer’s disease and our Phase 3 HORIZON trial of dimebon in patients with Huntington disease. In January 2012, Pfizer exercised its right to terminate the collaboration agreement and
37
Table of Contents
we and Pfizer discontinued development of dimebon for all indications in 2012 as discussed in the section titled, “Management’s Discussion and Analysis of Financial Condition and Results of Operations—Subsequent Events—Termination of Pfizer Collaboration Agreement and Dimebon Program.”
We have incurred cumulative net losses of $250.3 million through December 31, 2011, and we expect to incur substantial additional losses for the foreseeable future as we pursue regulatory approval for, and, if approved, commercial launch of, MDV3100 and continue to finance clinical and preclinical studies of our existing and potential future product candidates and our corporate overhead costs.
As of December 31, 2011, we had cash, cash equivalents and short-term investments totaling $145.1 million available to fund our operations. Based upon our current expectations, we believe our capital resources at December 31, 2011 will be sufficient to fund our currently planned operations for the next 12 months. This estimate is based on a number of assumptions that may prove to be wrong, and we could exhaust our available cash, cash equivalents, and short-term investments earlier than presently anticipated. We may be required or choose to seek additional capital within the next 12 months to expand our clinical development activities for MDV3100 based on the positive results of our Phase 3 AFFIRM trial in post-chemotherapy advanced prostate cancer patients, to fund costs of planning for and executing a commercial launch of MDV3100, should it receive marketing approval, if we face challenges or delays in connection with our clinical trials or the potential approval and commercialization of MDV3100, or to maintain minimum cash balances that we deem reasonable and prudent. In addition, we intend to evaluate the capital markets from time to time to determine whether to raise additional capital in the form of equity, convertible debt or otherwise, depending on market conditions relative to our need for funds at such time, and we may seek to raise additional capital within the next 12 months should we conclude that such capital is available on terms that we consider to be in the best interests of our company and our stockholders.
Subsequent Events
Termination of Pfizer Collaboration Agreement and Dimebon Program
In January 2012, we reported negative top line results from our Phase 3 CONCERT trial of dimebon in patients with mild-to-moderate Alzheimer’s disease. We previously had reported negative top line results from our Phase 3 CONNECTION trial of dimebon in patients with mild-to-moderate Alzheimer’s disease and our Phase 3 HORIZON trial of dimebon in patients with Huntington disease. In January 2012, Pfizer exercised its right to terminate the collaboration agreement and we and Pfizer discontinued development of dimebon for all indications. During the ensuing 180 days, we and Pfizer will work together to wind down our respective remaining collaboration activities.
We estimate that we and Pfizer will complete our respective collaboration wind down activities in the first half of 2012. Thus, during the first quarter of 2012 we revised our estimate of the remaining performance period under our former collaboration agreement with Pfizer. We expect the performance period to conclude in the first half of 2012. The remaining deferred revenue balance relating to the former collaboration agreement with Pfizer, which totaled $72.0 million at December 31, 2011, will be recognized as revenue over the remaining performance period.
Oakbrook Terrace Lease
In February 2012, we entered into a lease agreement for 15,336 square feet of office space located in Oakbrook Terrace, Illinois, which is intended to serve as our commercial headquarters following construction and build-out. The term of the lease is seven years and seven months, with an anticipated commencement date of May 2012. We selected this location for our commercial headquarters based on its proximity to our collaboration partner, Astellas. The annual non-cancelable rent payments are approximately $0.3 million per year. In connection with the execution of the lease, we delivered to the lessor a letter of credit collateralized by restricted cash totaling $0.8 million.
38
Table of Contents
Critical Accounting Policies and the Use of Estimates
The preparation of consolidated financial statements in conformity with accounting principles generally accepted in the United States, or U.S. GAAP, requires that management make estimates and assumptions in certain circumstances that affect the reported amounts of assets and liabilities and disclosures of contingent assets and liabilities at the date of the financial statements and the reported amounts of revenues and expenses during the reporting period. Actual results could differ materially from those estimates. A detailed description of our significant accounting policies is included in the footnotes to our audited consolidated financial statements included elsewhere in this Annual Report.
We believe that certain of our accounting policies are critical because they are the most important to the preparation of our consolidated financial statements. These policies require our most subjective and complex judgments, often requiring the use of estimates about the effects of matters that are inherently uncertain. We apply estimation methodologies consistent from year to year. Other than changes required due to the issuance of new accounting guidance, there have been no significant changes in our application of our critical accounting policies during the periods presented. The following is a summary of accounting policies that we consider critical to our consolidated financial statements.
Estimated Performance Periods under our Collaboration Agreements
Both the Astellas Collaboration Agreement and the former collaboration agreement with Pfizer contain multiple elements and deliverables, and required evaluation pursuant to the Financial Accounting Standards Board, or FASB, Accounting Standards Codification, or ASC, 605-25 “Revenue Recognition—Multiple-Element Arrangements.” We evaluated the facts and circumstances of the collaboration agreements to determine whether we had obligations constituting deliverables under ASC 605-25. We concluded that we had multiple deliverables under both collaboration agreements, including deliverables relating to grants of technology licenses, and performance of manufacturing, regulatory and clinical development activities in the U.S. In the case of the Astellas Collaboration Agreement, the period in which we perform our deliverables began in the fourth quarter of 2009 and at December 31, 2011, management estimated that it would be completed in the fourth quarter of 2014. In the case of the former collaboration agreement with Pfizer, the period in which we perform our deliverables began in the fourth quarter of 2008 and at December 31, 2011, management estimated that it would be completed in the fourth quarter of 2013. We also concluded that our deliverables under each collaboration agreement should be accounted for as a single unit of accounting under ASC 605-25.
Estimation of the performance periods of our deliverables requires the use of our management’s judgment. Significant factors considered in management’s evaluation of the estimated performance period include, but are not limited to, our experience, along with Astellas’ and Pfizer’s experience, in conducting clinical development and regulatory activities. We review the estimated duration of our performance periods under both collaborations on a quarterly basis and make any appropriate adjustments on a prospective basis.
During the year ended December 31, 2010, we extended the estimated completion date of our performance period under the former collaboration agreement with Pfizer from the second quarter of 2012 to the fourth quarter of 2013, based on the failure of the CONNECTION trial and the resulting longer period required to complete the clinical trials evaluating dimebon’s potential safety and efficacy as a treatment for mild-to-moderate Alzheimer’s disease.
In January 2012, Pfizer exercised its right to terminate the collaboration agreement. As a result, during the first quarter of 2012, we revised our estimate of the remaining performance period under the collaboration agreement. We expect to complete our remaining performance obligations pursuant to the terms of the former collaboration agreement with Pfizer in the first half of 2012.
Future changes in estimates of either of the performance periods under our collaboration agreements could significantly impact the timing of future revenue recognized under the applicable collaboration agreement.
39
Table of Contents
Collaboration Agreement Payments
We account for the various payment flows under our collaboration agreements in a consistent manner, as follows:
Up-Front Payments. We received non-refundable up-front cash payments of $110.0 million and $225.0 million under our collaboration agreement with Astellas and our former collaboration agreement with Pfizer, respectively. We recognize these payments as revenue on a straight-line basis over the applicable estimated performance period.
Milestone Payments. We are eligible to receive milestone payments under the Astellas Collaboration Agreement based on achievement of specified development, regulatory and commercial events. Management evaluated the nature of the events triggering these contingent payments, and concluded that these events—except for (a) those relating to regulatory activities in Europe, development and regulatory activities in Japan, and commercial activities, all of which are areas in which we have no pertinent contractual responsibilities, and (b) the initiation of our Phase 3 PREVAIL trial, an event which management deemed to be reasonably assured at the inception of the Astellas collaboration—constituted substantive milestones. This conclusion was based primarily on the facts that (i) each triggering event represents a specific outcome that can be achieved only through successful performance by us of one or more of our deliverables, (ii) achievement of each triggering event was subject to inherent risk and uncertainty and would result in additional payments becoming due to us, (iii) each of these milestones was substantive, based primarily on the facts that the payments they trigger are non-refundable, (iv) achievement of the milestone entails risk and was not reasonably assured at inception of the collaboration agreement, (v) substantial effort is required to complete each milestone, (vi) the amount of each milestone payment is reasonable in relation to the value created in achieving the milestone, (vii) a substantial amount of time is expected to pass between the up-front payment and the potential milestone payments, and (viii) the milestone payments relate solely to past performance. Based on the foregoing, we will recognize any revenue from these milestone payments under the substantive milestone method in the period in which the underlying triggering event occurs.
For the contingent payments triggered by events that do not constitute substantive milestones, management concluded that the appropriate revenue recognition treatment depends on whether the triggering event occurs during or after the performance period. Where the triggering event occurs during the applicable performance period, we will amortize any revenue from this event on a straight-line basis over the applicable performance period. Where the triggering event occurs after the applicable performance period, we will recognize the associated revenue in the period in which the event occurs.
Royalties and Profit Sharing Payments. Under the Astellas Collaboration Agreement, we are eligible to receive profit sharing payments on sales of products in the U.S. and royalties on sales of products outside the U.S. We will recognize any revenue from these events based on the revenue recognition criteria set forth in ASC 605-10-25-1, “Revenue Recognition.” Based on those criteria, we consider these potential payments to be contingent revenues, and will recognize them as revenue in the period in which the applicable contingency is resolved.
Cost-Sharing Payments. Under both the Astellas Collaboration Agreement and the former collaboration agreement with Pfizer, we and our collaboration partners share certain development and commercialization costs in the U.S. The parties make quarterly cost-sharing payments to one another in amounts necessary to ensure that each party bears its contractual share of the overall development and commercialization costs as incurred. Our policy is to account for cost-sharing payments to our collaboration partners as increases in expense in our consolidated statements of operations, while cost-sharing payments from our collaborative partners are presented as reductions in expense. Cost-sharing payments related to development activities and commercialization activities are recorded in research and development and selling, general and administrative expenses, respectively.
40
Table of Contents
Stock-Based Compensation
We have granted stock options, restricted stock units, performance share awards and stock appreciation rights pursuant to the terms of our Amended and Restated 2004 Equity Incentive Award Plan. We account for stock-based compensation awards granted to employees and directors in accordance with ASC 718, “Stock Compensation,” and have applied the provisions of the Securities and Exchange Commission, or the SEC, Staff Accounting Bulletin, or SAB, No. 107 and No. 110, in our application of ASC 718. Stock compensation arrangements with consultants are accounted for in accordance with ASC 718 and ASC 505-50, “Equity-Based Payments to Non-Employees,” using a fair value approach. The compensation costs of these arrangements are subject to re-measurement over the vesting terms as earned.
We determine stock-based compensation expense associated with stock options based on the estimated grant date fair value using the Black-Scholes valuation model, which requires the use of subjective assumptions related to expected stock price volatility, expected option term, expected dividend yield and risk-free interest rate. For employee and director stock options, we recognize compensation expense over the vesting period of the awards. Stock options granted to consultants are valued at their respective measurement dates and recognized as expense based on the portion of the total consulting services provided during the applicable period. As further consulting services are provided in each period, we revalue the associated awards and recognize additional expense based on their then-current fair values.
Stock-based compensation expense associated with restricted stock units is based on the fair value of our common stock on the grant date, which equals the closing market price of our common stock on the grant date of the award. For restricted stock units, we recognize compensation expense over the vesting period of the awards.
Performance share awards allow the recipients of such awards to earn fully vested shares of our common stock based on the achievement of pre-established performance objectives specified in the awards. Stock-based compensation expense associated with performance share awards is based on the estimated grant date fair value of our common stock using the Black-Scholes valuation model and is recognized based upon our best estimates of the achievement of the performance objectives specified in such awards and the resulting number of shares that are expected to be earned. We evaluate on a quarterly basis the probability of achieving the performance criteria. The cumulative effect on current and prior periods of a change in the estimated number of performance share awards expected to be earned is recognized as compensation expense or as reduction of previously recognized compensation expense in the period of the revised estimate.
The fair value of stock-settled and cash-settled stock appreciation rights is initially measured on the grant date using the Black-Scholes valuation model, which requires the use of subjective assumptions related to the expected stock price volatility, expected term, expected dividend yield and risk-free interest rate. Similar to stock options, compensation expense for stock-settled stock appreciation rights is recognized on a straight-line basis over the vesting period based on the grant-date fair value. Cash-settled stock appreciation rights are liability-classified awards for which compensation expense and the liability are remeasured at each reporting date through the date of settlement based on the portion of the requisite service period rendered.
We apply a forfeiture rate when determining stock-based compensation expense to account for an estimate of the granted awards not expected to vest. If actual forfeitures differ from the expected rate, we may be required to make additional adjustments to compensation expense in future periods.
The Black-Scholes valuation model was developed for use in estimating the fair value of traded options that have no vesting restrictions and are fully transferrable; characteristics not present in our option grants. If the model permitted consideration of the unique characteristics of employee stock options and stock appreciation rights, the resulting estimate of the fair value of the stock options and stock appreciation rights could be different. In addition, if we had made different assumptions and estimates for use in the Black-Scholes valuation model, the amount of recognized and to be recognized stock-based compensation expense could have been different.
41
Table of Contents
Research and Development Expense and Accruals
Research and development expenses include personnel and facility-related expenses, outside contracted services including clinical trial costs, manufacturing and process development costs, research costs and other consulting services. Research and development costs are expensed as incurred. In instances where we enter into agreements with third parties to provide research and development services to us, costs are expensed as services are performed. Amounts due under such arrangements may be either fixed fee or fee for service, and may include upfront payments, monthly payments, and payments upon the completion of milestones or receipt of deliverables.
Our cost accruals for clinical trials and other research and development activities are based on estimates of the services received and efforts expended pursuant to contracts with numerous clinical trial centers and contract research organizations. In the normal course of business we contract with third parties to perform various research and development activities in the on-going development of our product candidates, including without limitation, third party clinical trial centers and contract research organizations that perform and administer our clinical trials on our behalf. The financial terms of these agreements are subject to negotiation and vary from contract to contract and may result in uneven payment flows. Payments under these agreements depend on factors such as the achievement of certain events, the successful enrollment of patients, and the completion of portions of the clinical trial or similar conditions. The objective of our accrual policy is to match the recording of expenses in our financial statements to the actual services received and efforts expended. As such, expense accruals related to clinical trials and other research and development activities are recognized based on our estimate of the degree of completion of the event or events specified in the specific agreement.
Our estimates are dependent upon the time lines and accuracy of data provided by third parties regarding the status and cost of studies, and may not match the actual services performed by the organizations. This could result in adjustment to our research and development expense in future periods. To date, we have had no significant adjustments.
Leases
At the inception of a lease, we evaluate the lease agreement to determine whether the lease is an operating or a capital lease using the criteria in ASC 840, “Leases.” For operating leases, we recognize rent expense on a straight-line basis over the non-cancellable lease term and record the difference between cash rent payments and the recognition of rent expense as a deferred liability. Where lease agreements contain rent escalation clauses, rent abatements and/or concessions, such as rent holidays and tenant improvement allowances, we apply them in the determination of straight-line rent expense over the lease term. We currently have no capital leases. Certain lease agreements also require us to make additional payments under the lease term for taxes, insurance, and other operating expenses incurred during the lease period, which are expensed as incurred.
Income Taxes
We establish reserves for uncertain income tax positions in accordance with ASC 740-10-25, “Accounting for Uncertainty in Income Taxes,” based on the technical support for the position, our past audit experiences with similar situations, and potential interest and penalties related to the matters. Our recorded reserves represent our best estimate of the amount, if any, that we may ultimately be required to pay to settle such matters. The resolution of our uncertain income tax positions is dependent on uncontrollable factors such as law changes, new case law, and the willingness of the income tax authorities to settle, including the timing thereof and other factors. Although we do not anticipate significant changes to our uncertain income tax positions in the next 12 months, items outside of our control could cause our uncertain income tax positions to change in the future, which would be recorded in our consolidated statements of operations. Interest and/or penalties related to income tax matters are recognized as a component of income tax expense as incurred.
42
Table of Contents
We maintained a full valuation allowance on our net deferred tax assets as of December 31, 2011. The valuation allowance was determined in accordance with the ASC 740-10, which requires an assessment of both positive and negative evidence when determining whether it is more likely than not that deferred tax assets are recoverable; such assessment is required on a jurisdiction by jurisdiction basis. Cumulative historic losses represented sufficient negative evidence under ASC 740-10 and accordingly, a full valuation allowance was recorded against U.S. deferred tax assets. We intend to maintain a full valuation allowance on the U.S. deferred tax assets until sufficient positive evidence exists to support reversal of the valuation allowance.
Basis of Presentation
The consolidated financial statements incorporate the accounts of Medivation, Inc. and its wholly-owned subsidiaries. All significant inter-company transactions and balances have been eliminated in consolidation. We operate in one business segment.
All tabular disclosures of dollar amounts are presented in thousands. All share and per share amounts are presented at their actual amounts. Percentages and amounts presented herein may not calculate or sum precisely due to rounding.
Recent Accounting Pronouncements
Information regarding recent accounting pronouncements applicable to us is included in the notes to our audited consolidated financial statement included elsewhere in this Annual Report.
Results of Operations
Collaboration Revenue
| Years Ended December 31, | ||||||||||||
| 2011 | 2010 | 2009 | ||||||||||
| Collaboration revenue from Astellas |
$ | 24,374 | $ | 23,492 | $ | 3,893 | ||||||
| Collaboration revenue from Pfizer |
36,015 | 39,016 | 65,361 | |||||||||
|
|
|
|
|
|
|
|||||||
| Total |
$ | 60,389 | $ | 62,508 | $ | 69,254 | ||||||
|
|
|
|
|
|
|
|||||||
| Percentage change |
(3 | %) | (10 | %) | ||||||||
The decrease in collaboration revenue for the year ended December 31, 2011 as compared to the prior year was due to a change in estimate of the remaining performance period under our former collaboration agreement with Pfizer, which is discussed below. Collaboration revenue for the year ended December 31, 2011 includes the incremental amortization resulting from $3.0 million and $10.0 million of development milestone payments received from our collaboration partner, Astellas, during the second quarter of 2011 and the fourth quarter of 2010, respectively, which have been deferred on our consolidated balance sheet and are being amortized on a straight-line basis over the estimated performance period with Astellas.
The $6.7 million decrease in collaboration revenue for the year ended December 31, 2010 as compared to the prior year was driven by lower collaboration revenues of $26.3 million from our former collaboration agreement with Pfizer due to the extension of our estimated performance period from the second quarter of 2012 to the fourth quarter of 2013, which is discussed below. This was partially offset by an increase of $19.6 million in collaboration revenue from the Astellas Collaboration Agreement, which was in effect for only one quarter in 2009.
43
Table of Contents
To date, we have received $123.0 million and $225.0 million of up-front and development milestone payments under the Astellas Collaboration Agreement and the former collaboration agreement with Pfizer, respectively. At December 31, 2011, deferred revenue balances with respect to these collaboration agreements were $71.2 million and $72.0 million, respectively. We amortize deferred revenue on a straight-line basis over the estimated performance period of our deliverables under the applicable collaboration agreement.
At December 31, 2011, management estimated that our performance periods under the Astellas Collaboration Agreement and the former collaboration agreement with Pfizer will be completed in the fourth quarter of 2014 and 2013, respectively. During the year ended December 31, 2010, we extended the estimated completion date of our performance period under the former collaboration agreement with Pfizer from the second quarter of 2012 to the fourth quarter of 2013, based on the failure of the CONNECTION trial and the resulting longer period required to complete the clinical trials evaluating dimebon’s potential safety and efficacy as a treatment for mild-to-moderate Alzheimer’s disease. The cumulative effect of these changes, which we applied on a prospective basis, was to extend the performance period by seven quarters and correspondingly reduce collaboration revenue related to the former collaboration agreement with Pfizer by $7.3 million per quarter over the remaining estimated performance period.
In January 2012, Pfizer exercised its right to terminate the collaboration agreement as discussed in the section titled “Management’s Discussion and Analysis of Financial Condition and Results of Operations—Subsequent Events—Termination of Pfizer Collaboration Agreement and Dimebon Program.” During the ensuing 180 days, we and Pfizer will work together to wind down our respective remaining collaboration activities. We estimate that we and Pfizer will complete our respective collaboration wind down activities in the first half of 2012. Thus, during the first quarter of 2012, we revised our estimate of the remaining performance period under our former collaboration agreement with Pfizer. We expect the performance period to conclude in the first half of 2012. The remaining deferred revenue balance relating to the former collaboration agreement with Pfizer, which totaled $72.0 million at December 31, 2011, will be recognized as revenue over the remaining performance period.
Future changes in estimates of the performance periods under our collaboration agreements could significantly impact the timing of future revenue recognized under the applicable collaboration agreement.
Research and Development Expense
| Years Ended December 31, | ||||||||||||
| 2011 | 2010 | 2009 | ||||||||||
| Research and development expense |
$ | 73,432 | $ | 72,228 | $ | 87,728 | ||||||
| Percentage change |
2 | % | (18 | %) | ||||||||
Research and development, or R&D, expenses increased by $1.2 million, or 2%, to $73.4 million in the year ended December 31, 2011 from $72.2 million in the year ended December 31, 2010. The increase was due primarily to a $4.5 million increase in payroll-related expenses associated with increased headcount and higher bonus expense, a $3.9 million increase in consulting and professional service expenses associated with increased workload on our AFFIRM and PREVAIL trials and $2.0 million in preclinical expenses associated with new programs. These items were partially offset by lower clinical expenses of $7.6 million resulting primarily from reduced dimebon development activities.
R&D expenses decreased by $15.5 million, or 18%, in the year ended December 31, 2010 as compared to the prior year. This decrease was due primarily to a $10.0 million decrease in up-front and development milestone sharing expense to The Regents of the University of California, or UCLA, pursuant to the terms of our MDV3100 license agreement and a $5.2 million decrease in payroll costs resulting from favorable changes in employee-related cost-sharing payments with our collaboration partner Astellas and former collaboration partner Pfizer.
44
Table of Contents
R&D expenses represented 71%, 76% and 75% of total operating expenses for the years ended December 31, 2011, 2010 and 2009, respectively.
We recorded development cost-sharing payments from Astellas and Pfizer, and corresponding reductions in research and development expense, as follows:
| Years Ended December 31, | ||||||||||||
| 2011 | 2010 | 2009 | ||||||||||
| Development cost-sharing payments from Astellas |
$ | 44,285 | $ | 34,125 | $ | 2,784 | ||||||
| Development cost-sharing payments from Pfizer |
12,365 | 29,139 | 20,435 | |||||||||
|
|
|
|
|
|
|
|||||||
| Total |
$ | 56,650 | $ | 63,264 | $ | 23,219 | ||||||
|
|
|
|
|
|
|
|||||||
To date, we have been engaged in two major research and development programs: the development of MDV3100 for the treatment of advanced prostate cancer and the development of dimebon for the treatment of Alzheimer’s disease and Huntington disease. Other research and development programs consist of earlier stage programs. Research and development costs are identified as either directly allocable to one of our research and development programs or as an indirect cost, with only direct costs being tracked by specific program. Direct costs consist primarily of clinical and preclinical study costs, cost of supplying drug substance and drug product for use in clinical and preclinical studies, personnel costs (including both cash costs and non-cash stock-based compensation costs), contract research organization fees, and other contracted services pertaining to specific clinical and preclinical studies. Indirect costs consist of corporate overhead costs and other administrative and support costs. The following table summarizes the direct costs attributable to each program and the total indirect costs for each respective period.
| Years Ended December 31, | ||||||||||||
| 2011 | 2010 | 2009 | ||||||||||
| Direct costs: |
||||||||||||
| MDV3100 |
$ | 40,545 | $ | 23,454 | $ | 27,046 | ||||||
| Dimebon |
11,749 | 35,327 | 47,148 | |||||||||
| Other |
14,088 | 9,258 | 9,461 | |||||||||
|
|
|
|
|
|
|
|||||||
| Total direct costs |
66,382 | 68,039 | 83,655 | |||||||||
| Indirect costs |
7,050 | 4,189 | 4,073 | |||||||||
|
|
|
|
|
|
|
|||||||
| Total |
$ | 73,432 | $ | 72,228 | $ | 87,728 | ||||||
|
|
|
|
|
|
|
|||||||
Our projects or intended projects may be subject to change from time to time as we evaluate our research and development priorities and available resources.
Based on the negative results in the Phase 3 CONNECTION, HORIZON and CONCERT trials, we and Pfizer have discontinued development of dimebon for all indications in 2012. In January 2012, Pfizer exercised its right to terminate the collaboration agreement as discussed in the section titled, “Management’s Discussion and Analysis of Financial Condition and Results of Operations—Subsequent Events—Termination of Pfizer Collaboration Agreement and Dimebon Program.”
In order to obtain the necessary regulatory approvals, we will need to establish to the satisfaction of the applicable regulatory authorities in the United States, Europe and other relevant countries that the applicable product candidate is both safe and effective for each of its intended indications. The process of conducting the preclinical and clinical testing required to establish safety and efficacy and obtain regulatory approvals is expensive, uncertain and takes many years. We are not able to reasonably estimate the time or cost required to obtain such regulatory approvals, and failure to receive the necessary regulatory approvals would prevent us from commercializing the product candidates affected. The length of time required for clinical development of a
45
Table of Contents
particular product candidate and our development costs for that product candidate may be impacted by the scope and timing of enrollment in clinical trials for the product candidate, unanticipated additional clinical trials that may be required, future decisions to develop a product candidate for subsequent indications, and whether in the future we decide to pursue development of the product candidate with a corporate partner or independently. For example, MDV3100 may have the potential to be approved for multiple indications, and we do not yet know how many of those indications we and our partner, Astellas, will pursue. The decision to pursue regulatory approval for subsequent indications will depend on several variables outside of our control, including the strength of the data generated in our prior and ongoing clinical and non-clinical studies and the willingness of our corporate partner to jointly fund such additional work. Furthermore, the scope and number of clinical studies required to obtain regulatory approval for each pursued indication is subject to the input of the applicable regulatory authorities; we have not yet sought such input for all potential indications that we and our collaboration partner may elect to pursue, and even after having given such input applicable regulatory authorities may subsequently require additional clinical studies prior to granting regulatory approval based on new data generated by us or other companies, or for other reasons outside of our control. Moreover, we or our current or potential future collaboration partners may decide to discontinue development of any development project at any time for regulatory, commercial, scientific or other reasons. To date, we have not commercialized any of our product candidates and in fact may never do so.
Selling, General and Administrative Expense
| Years Ended December 31, | ||||||||||||
| 2011 | 2010 | 2009 | ||||||||||
| Selling, general and administrative expense |
$ | 29,887 | $ | 23,005 | $ | 28,983 | ||||||
| Percentage change |
30 | % | (21 | %) | ||||||||
Selling, general and administrative, or SG&A, expense increased by $6.9 million, or 30%, to $29.9 million in the year ended December 31, 2011 from $23.0 million in the year ended December 31, 2010. This increase was due primarily to a $3.6 million increase in payroll-related expenses associated with increased headcount and higher bonus expense, a $1.7 million increase in legal fees primarily related to a litigation matter we initiated in the second quarter of 2011 against the licensor of our MDV3100 technology and the defense of the securities class action lawsuit following the negative results in our CONNECTION trial, for which we are subject to a $350,000 retention. The increase in SG&A expense was further impacted by an increase in sales and marketing expenses of $0.9 million as a result of increased market research and public relations expense due to the positive results of our Phase 3 AFFIRM trial discussed elsewhere in this Annual Report and recruitment expenses of $0.3 million as a result of increased headcount in 2011. These items were partially offset by restructuring costs and a lease termination fee in response to the negative results of our Phase 3 CONNECTION trial discussed elsewhere in this Annual Report.
SG&A expense decreased by $6.0 million, or 21%, in the year ended December 31, 2010 as compared to the prior year. This decrease was due primarily to decreases of $2.6 million in consulting and professional services fees, $2.5 million in payroll and other costs associated with lower headcount related to our SG&A activities and $0.9 million in sales and marketing expenses. These expense reductions were largely pursuant to the restructuring that we implemented in March 2010 in response to the negative results of our CONNECTION trial.
SG&A expenses represented 29%, 24% and 25% of total operating expenses in the years ended December 31, 2011, 2010 and 2009, respectively. We expect SG&A expenses to continue to increase as a percentage of our overall operating expenses as we hire additional staff and incur additional expenses in preparation for a potential commercial launch of MDV3100 in post-chemotherapy advanced prostate cancer.
46
Table of Contents
We recorded commercialization cost-sharing payments (to) from Astellas and Pfizer, and corresponding (increases) reductions in SG&A expenses, as follows:
| Years Ended December 31, | ||||||||||||
| 2011 | 2010 | 2009 | ||||||||||
| Commercialization cost-sharing payments (to) from Astellas |
$ | (472 | ) | $ | 520 | $ | 74 | |||||
| Commercialization cost-sharing payments (to) from Pfizer |
32 | (1,084 | ) | (720 | ) | |||||||
|
|
|
|
|
|
|
|||||||
| Total |
$ | (440 | ) | $ | (564 | ) | $ | (646 | ) | |||
|
|
|
|
|
|
|
|||||||
Other (Expense) Income, net
| Years Ended December 31, | ||||||||||||
| 2011 | 2010 | 2009 | ||||||||||
| Other (expense) income, net |
$ | (242 | ) | $ | 260 | $ | 976 | |||||
Other (expense) income, net, consists of interest income on our cash and short-term investment balances as well as the impact of changes in foreign exchange rates on our foreign exchange denominated payables. The impact of foreign exchange rates on our results of operations fluctuates period over period based upon our foreign currency exposures resulting from changes in applicable exchange rates associated with our foreign denominated payables.
Interest income decreased $0.2 million to $0.1 million for the year ended December 31, 2011 as compared to $0.3 million for the year ended December 31, 2010. The decrease was due to lower investment yields and average investment balances during 2011. We recognized foreign currency losses of $0.3 million during the year ended December 31, 2011 as compared to foreign currency losses of $0.1 million for the year ended December 31, 2010.
Interest income decreased $0.8 million to $0.3 million for the year ended December 31, 2010 as compared to $1.1 million for the year ended December 31, 2009. The decrease was due to lower investment yields and average investment balances in 2010. We recognized foreign currency losses of $0.1 million during the year ended December 31, 2010 as compared to foreign currency losses of $0.2 million for the year ended December 31, 2009.
Income Tax (Benefit) Expense
| Years Ended December 31, | ||||||||||||
| 2011 | 2010 | 2009 | ||||||||||
| Income tax (benefit) expense |
$ | (4,331 | ) | $ | 1,572 | $ | 8,272 | |||||
| Effective tax rate |
10.0 | % | (4.8 | %) | (17.9 | %) | ||||||
The income tax benefit for the year ended December 31, 2011 was $4.3 million and represents an effective tax rate of 10.0%. The income tax benefit mainly consists of a benefit due to the ability to carry back the current year Federal net operating loss, or NOL, to the prior two tax years. The difference in the effective tax rate for 2011 as compared to 2010 is primarily attributable to the benefit in 2011 due to the ability to carry back the current year Federal NOL to the prior two tax years.
The income tax expense for 2010 was approximately $1.6 million, which mainly consisted of Federal and state income tax and represents an effective tax rate of (4.8%). We incurred income tax liability for 2010 despite reporting a net loss in our consolidated financial statements primarily because we recognized for tax purposes in 2010 substantially all of the $110.0 million up-front payment and all of the $10.0 million development milestone payment previously received from Astellas. Due to the suspension of California net operating loss, or NOL,
47
Table of Contents
utilization for 2010, we were not able to utilize NOL carryforwards to offset state taxable income. The reduction in the effective tax rate for 2010 as compared to 2009 is primarily attributable to a California state income tax refund of $5.3 million recognized in 2010.
The income tax expense for 2009 was approximately $8.3 million, which mainly consisted of federal and state income tax and represents an effective tax rate of 17.9%. We incurred income tax liability for 2009 despite reporting a net loss for financial statement purposes primarily because we recognized for tax purposes in 2009 substantially all of the $225.0 million up-front payment previously received from our former collaboration partner Pfizer. Due to the suspension of California NOL utilization for 2009, we were not able to utilize NOL carryforwards to offset state taxable income.
A reconciliation of the Federal statutory income tax rate of 35% to our effective tax rate for each year presented is included in Note 9 , “Income Taxes,” to our consolidated financial statements included elsewhere in this Annual Report.
Liquidity and Capital Resources
Sources of Liquidity
We have incurred cumulative net losses of $250.3 million through December 31, 2011, and we expect to incur substantial additional losses for the foreseeable future as we pursue regulatory approval for, and, if approved, commercial launch of, MDV3100 and continue to finance clinical and preclinical studies of MDV3100 and potential future product candidates and our corporate overhead costs. We have not generated any revenue from product sales to date, and we may never generate any revenue from product sales. Our operations to date have been funded primarily through private and public offerings of our common stock and from the up-front, development milestone and cost-sharing payments from our collaboration partner Astellas and from our former collaboration partner, Pfizer.
As of December 31, 2011, we had cash, cash equivalents and short-term investments totaling $145.1 million available to fund our operations. Based upon our current expectations, we believe our capital resources at December 31, 2011 will be sufficient to fund our currently planned operations for the next 12 months. This estimate is based on a number of assumptions that may prove to be wrong and we could exhaust our available cash, cash equivalents and short-term investment earlier than presently anticipated. We may be required or choose to seek additional capital within the next 12 months to expand our clinical development activities for MDV3100 based on the positive results of our Phase 3 AFFIRM trial in post-chemotherapy advanced prostate cancer patients, to fund costs of planning for and executing a commercial launch of MDV3100, should it receive marketing approval, if we face challenges or delays in connection with our clinical trials or the potential approval and commercialization of MDV3100, or to maintain minimum cash balances that we deem reasonable and prudent. In addition, we intend to evaluate the capital markets from time to time to determine whether to raise additional capital in the form of equity, convertible debt or otherwise, depending on market conditions relative to our need for funds at such time, and we may seek to raise additional capital within the next 12 months should we conclude that such capital is available on terms that we consider to be in the best interests of our company and our stockholders.
Our current view of the worldwide capital markets is that they are extremely volatile with limited accessibility, and many biotechnology companies have had limited or no success in obtaining funding in this environment. Continuation of this challenging market climate may significantly limit our ability to raise funds, and there can be no assurance we will be able to raise additional funds on acceptable terms or at all. If we are unable to raise additional funds when needed, we could be required to delay, scale back or eliminate some or all of our development programs and other operations. We may seek to raise additional funds through public or private financing or other arrangements. Any additional equity financing would be dilutive to stockholders, and debt financing, if available, may involve restrictive covenants. Our failure to raise capital when needed may harm our business and operating results.
48
Table of Contents
Our future capital requirements will depend on many factors, many of which are wholly or partially outside of our control. Such factors include, without limitation:
| • | potential launch costs of MDV3100, should that agent receive marketing approval for post-chemotherapy patients; |
| • | the timing and magnitude of any potential sales of MDV3100, should that agent receive marketing approval for post-chemotherapy patients; |
| • | whether any changes are made to the scope of our ongoing clinical development activities; |
| • | the scope and results of our and our collaboration partner’s preclinical and clinical trials; |
| • | whether we experience delays in our preclinical and clinical development programs, including potential delays in recruiting, or inability to recruit, patients into our ongoing PREVAIL trial of MDV3100 in pre-chemotherapy advanced prostate cancer as a result of the availability of Zytiga (abiraterone acetate), which was approved by the FDA in April 2011 for post-chemotherapy patients, or other investigational and approved prostate cancer therapies, including MDV3100 itself should that agent be approved to treat post-chemotherapy patients, or slower than anticipated product development; |
| • | whether opportunities to acquire additional product candidates arise and the timing and costs of acquiring and developing those product candidates; |
| • | whether we are able to enter into additional third-party collaborative partnerships to develop and/or commercialize potential future product candidates on terms, including development cost share terms, that are acceptable to us; |
| • | the timing and requirements of, and the costs involved in, conducting studies required to obtain regulatory approvals for MDV3100 or potential future product candidates from the FDA and comparable foreign regulatory agencies; |
| • | the availability of third parties to perform the key development tasks for MDV3100 and potential future product candidates, including conducting preclinical and clinical studies and manufacturing our product candidates to be tested in those studies, and the associated costs of those services; |
| • | expenses associated with the pending purported securities class action lawsuits, as well as any other litigation; and |
| • | the costs involved in preparing, filing, prosecuting, maintaining, defending the validity of and enforcing patent claims and other costs related to patent rights and other intellectual property rights, including litigation costs and the results of such litigation. |
Astellas Collaboration Agreement
We entered into a collaboration agreement with Astellas in October 2009. Under the Astellas Collaboration Agreement, we and Astellas agreed to collaborate on the development of MDV3100 for the United States market, including associated regulatory filings with the FDA. In addition, if approved by the FDA, following such approval and the launch of MDV3100 in the United States, we and Astellas will co-promote MDV3100 in the United States. Astellas is responsible for development of, and seeking regulatory approval for, and commercialization of MDV3100 outside the United States. Astellas will be responsible for commercial manufacture of MDV3100 on a global basis. Both we and Astellas have agreed not to commercialize certain other products having a similar mechanism of action as MDV3100 for the treatment of specified indications for a specified time period, subject to certain exceptions.
We and Astellas share equally the costs of developing and commercializing MDV3100 for the United States market (subject to the exceptions noted below), and we and Astellas will share equally profits (or losses) resulting from the commercialization of MDV3100 in the United States. Costs of clinical trials supporting development in both the United States and in either Europe or Japan are borne two-thirds by Astellas and
49
Table of Contents
one-third by us. Both we and Astellas will be responsible for all costs incurred in fielding and supporting our respective MDV3100 sales forces, and each of us will be entitled to receive a fee for each detail made by our respective sales forces. Outside the United States, Astellas will bear all development and commercialization costs and will pay us tiered, double-digit royalties on the aggregate net sales of MDV3100.
The Astellas Collaboration Agreement establishes several joint committees consisting of an equal number of representatives from both parties that operate by consensus to oversee the collaboration. In the event that a joint committee is unable to reach consensus on a particular issue, then, depending on the issue, a dispute may be decided at the joint committee level by the party with the final decision on the issue or escalated to senior management of the parties. If a dispute is escalated to senior management and no consensus is reached, then the dispute may be decided by the party to whom the contract grants final decision on such issue. Other issues can only be decided by consensus of the parties, and unless and until the parties’ representatives reach agreement on such issue, no decision on such issue will be made, and the status quo will be maintained.
Under the Astellas Collaboration Agreement, Astellas paid us a non-refundable, up-front cash payment of $110.0 million in the fourth quarter of 2009. We are also eligible to receive up to $335.0 million in development milestone payments, plus up to an additional $320.0 million in commercial milestone payments. As of December 31, 2011, we had received an aggregate of $13.0 million in development milestone payments under the Astellas Collaboration Agreement. Should the FDA or European Medicines Agency accept for filing an NDA or a MAA, respectively, seeking approval of MDV3100 in post-chemotherapy patients based on the positive results from our AFFIRM trial, we would be entitled to a $10.0 million and a $5.0 million milestone payment, respectively, under the Astellas Collaboration Agreement. In addition, should the NDA or MAA be approved by the FDA or European regulators, respectively, we would be entitled to a $30.0 million and a $15.0 million milestone payment, respectively, from Astellas. We are required to share 10% of the up-front and development milestone payments received under the Astellas Collaboration Agreement with UCLA pursuant to the terms of our MDV3100 license agreement, which is discussed elsewhere in this Annual Report.
Each of Medivation and Astellas is permitted to terminate the Astellas Collaboration Agreement for an uncured material breach by the other party or for the insolvency of the other party. Astellas has a right to terminate the Astellas Collaboration Agreement unilaterally by advance written notice to us, but, except in certain specific circumstances, generally cannot exercise that termination right until the first anniversary of MDV3100’s first commercial sale. Following any termination of the Astellas Collaboration Agreement in its entirety, all rights to develop and commercialize MDV3100 will revert to us, and Astellas will grant a license to us to enable us to continue such development and commercialization. In addition, except in the case of a termination by Astellas for our uncured material breach, Astellas will supply MDV3100 to us during a specified transition period.
Former Collaboration Agreement with Pfizer
We entered into a collaboration agreement with Pfizer in October 2008. Under the agreement, we and Pfizer agreed to collaborate on the development and commercialization of dimebon for Alzheimer’s disease and Huntington disease for the United States market, including associated regulatory filings with the FDA. Pfizer paid us a non-refundable up-front cash payment of $225.0 million in the fourth quarter of 2008 pursuant to the terms of the agreement. Under the terms of the former collaboration agreement, we and Pfizer shared the costs and expenses of developing and commercializing dimebon for the United States market on a 60%/40% basis, with Pfizer assuming the larger share. Based on the negative results in the Phase 3 CONNECTION, HORIZON, and CONCERT trials, we and Pfizer have discontinued the development of dimebon for all indications in 2012. In January 2012, Pfizer exercised its right to terminate the collaboration agreement as discussed in “Management’s Discussion and Analysis of Financial Condition and Results of Operations—Subsequent Events—Termination of Pfizer Collaboration Agreement and Dimebon Program.”
50
Table of Contents
Cash Flow Analysis
| Years Ended December 31, | ||||||||||||
| 2011 | 2010 | 2009 | ||||||||||
| Net cash (used in) provided by: |
||||||||||||
| Operating activities |
$ | (75,961 | ) | $ | (75,064 | ) | $ | (7,585 | ) | |||
| Investing activities |
19,687 | 122,415 | (72,568 | ) | ||||||||
| Financing activities |
18,693 | 2,903 | 66,162 | |||||||||
|
|
|
|
|
|
|
|||||||
| Net change in cash and cash equivalents |
$ | (37,581 | ) | $ | 50,254 | $ | (13,991 | ) | ||||
|
|
|
|
|
|
|
|||||||
Operating Activities
Net cash used in operating activities totaled $76.0 million in 2011, which consisted of our net loss of $38.8 million and non-cash items of $46.1 million, partially offset by changes in operating assets and liabilities of $9.0 million. Non-cash items consisted primarily of the non-cash amortization of deferred revenue of $60.4 million, partially offset by non-cash stock-based compensation expense of $13.9 million. The increase in cash flows from changes in operating assets and liabilities was primarily driven by a $8.6 million decrease in receivables from our collaboration partner Astellas and our former collaboration partner Pfizer, an increase of $2.4 million in accounts payable arising in the ordinary course of business, an increase of $2.5 million in accrued expenses primarily driven by higher clinical and preclinical accruals and an increase in deferred revenue of $3.0 million related to a development milestone payment received from Astellas. These items were partially offset by a decrease in other current liabilities of $4.9 million as a result of a federal income tax payment of $4.5 million during 2011.
Net cash used in operating activities totaled $75.1 million in 2010, which consisted of our net loss of $34.1 million and non-cash items of $48.8 million, partially offset by changes in operating assets and liabilities of $7.8 million. Non-cash items consisted primarily of the non-cash amortization of deferred revenue of $62.5 million, partially offset by non-cash stock-based compensation expense of $13.5 million. The increase in cash flows from changes in operating assets and liabilities was primarily driven by a $10.0 million development milestone payment we received from Astellas, an increase in accounts payable and accrued expenses of $7.7 million arising in the ordinary course of business, and an increase in other current liabilities of $4.4 million, partially offset by increased receivables of $14.7 million from our collaboration partner Astellas and our former collaboration partner Pfizer.
Net cash used in operating activities totaled $7.6 million in 2009, which consisted of our net loss of $54.8 million and non-cash items of $59.3 million, partially offset by changes in operating assets and liabilities of $106.5 million. Non-cash items consisted primarily of the non-cash amortization of deferred revenue of $69.3 million, partially offset by non-cash stock-based compensation expense of $10.8 million. The increase in cash flows from changes in operating assets and liabilities was primarily driven by the non-refundable upfront payment of $110.0 million we received from Astellas and increased accounts payable and accrued expenses of $4.0 million arising in the ordinary course of business, partially offset by increased prepaid expenses of $5.5 million and increased receivables of $3.0 million from our collaboration partner Astellas and former collaboration partner Pfizer.
Investing Activities
Net cash provided by investing activities totaled $19.7 million in 2011, consisting primarily of net maturities of short-term investments of $25.1 million partially offset by an increase of $5.1 million in letters of credit collateralized by restricted cash to secure an operating lease for our future corporate headquarters.
Net cash provided by investing activities totaled $122.4 million in 2010, consisting primarily of net maturities of short-term investments.
51
Table of Contents
Net cash used in investing activities totaled $72.6 million in 2009, consisting primarily of net purchases of short-term investments.
Financing Activities
Net cash provided by financing activities totaled $18.7 million in the year ended December 31, 2011, consisting of $15.7 million in proceeds from the exercise of stock options and $3.0 million in net proceeds received in settlement of a short swing profit liability incurred by an unaffiliated stockholder.
Net cash provided by financing activities totaled $2.9 million in 2010, consisting primarily of $2.6 million in proceeds from the exercise of stock options and warrants.
Net cash provided by financing activities totaled $66.2 million in 2009, consisting primarily of net proceeds of approximately $62.1 million from the sale of our common stock in a registered offering and $3.4 million in proceeds from the exercise of stock options and warrants.
Commitments and Contingencies
The future minimum rentals under our non-cancelable operating leases at December 31, 2011 were as follows:
| Payment due by Period | ||||||||||||||||||||
| Total | Less than 1 Year |
1-3 Years | 4-5 Years | > 5 Years | ||||||||||||||||
| Operating lease obligations |
$ | 28,802 | $ | 3,599 | $ | 13,997 | $ | 6,651 | $ | 4,555 | ||||||||||
Additional information regarding our operating leases is included in Note 11, “Commitments and Contingencies,” to our consolidated financial statements included elsewhere in this Annual Report.
In February 2012, we entered into a lease agreement for 15,336 square feet of office space located in Oakbrook Terrace, Illinois, which is intended to serve as our commercial headquarters following construction and build-out. The term of the lease is seven years and seven months, with an anticipated commencement date of May 2012. We selected this location for our commercial headquarters based on its proximity to our collaboration partner, Astellas. The annual non-cancelable rent payments are approximately $0.3 million per year. In connection with the execution of the lease, we delivered to the lessor a letter of credit collateralized by restricted cash totaling $0.8 million. The future minimum lease payments under this lease are not included in the table above.
In addition to the contractual obligations disclosed in the table above, we have other potential obligations for which the timing and the extent of future payments are not known. We have described these potential obligations in the following paragraphs.
At December 31, 2011, $3.9 million of unrecognized tax benefits have been recorded as liabilities for uncertain income tax positions. The ultimate resolution of our uncertain income tax positions is dependent on uncontrollable factors such as law changes, new case law, and the willingness of the income tax authorities to settle, including the timing thereof, and other factors. Although we do not anticipate significant changes to our uncertain income tax positions in the next twelve months, items outside of our control could cause our uncertain income tax positions to change in the future. Such amounts have been included on our consolidated balance sheet at December 31, 2011, but have not been included in the table above.
We have a license agreement with UCLA under which we are required to pay UCLA an annual maintenance fee, up to $5.5 million in aggregate milestone payments upon the achievement of certain development and regulatory milestone events, and 10% of any up-front and development milestone payments we receive from
52
Table of Contents
sublicensees. We are also required to pay UCLA a single-digit royalty on sales of products falling within the scope of the patent rights licensed from UCLA. Should we receive marketing approval of MDV3100 in either the U.S. or Europe, we would be required to pay UCLA a single, one-time milestone payment of $2.0 million. Because the achievement of the development and regulatory milestones and the contingent royalty payments is neither probable nor reasonably estimable, such amounts have not been recorded on our consolidated balance sheet at December 31, 2011 and have not been included in the table above.
Off-Balance Sheet Arrangements
We have no material off-balance sheet arrangements as defined in Regulation S-K 303(a)(4)(ii).
| Item 7A. | Quantitative and Qualitative Disclosures About Market Risk. |
Our exposure to market risk for changes in interest rates relates to our cash equivalents on deposit in highly liquid money market accounts and short-term investments in highly liquid U.S. Treasury securities. The primary objective of our cash investment activities is to preserve principal. We do not use derivative financial instruments in our investment portfolio. Our cash and investments policy emphasizes liquidity and preservation of principal over other portfolio considerations. Our investment portfolio is subject to interest rate risk and will fall in value if market interest rates rise. There were no material changes to our market risk from those disclosed in our Annual Report on Form 10-K for the year ended December 31, 2010.
Interest Rate Risk
Our cash equivalents and short-term investments are exposed to the impact of interest rate changes and our interest income fluctuates as interest rates change. Due to the short-term nature of our investments in money market funds and U.S. Treasury securities, the carrying value of our cash equivalents and short-term investments approximate their fair value at December 31, 2011. Due to the short-term, highly liquid nature of our investments, we do not believe that we are subject to any material market risk exposure related to interest rates.
Foreign Currency Exchange Risk
We do not have any material exposure to foreign currency rate fluctuations as we operate primarily in the U.S. Although we conduct some research and development work with vendors outside the U.S., most of our transactions are denominated in U.S. dollars. However, certain of our ex-U.S. clinical development activities are pursuant to contracts denominated in foreign currencies. For the year ended December 31, 2011, we recorded $0.3 million in foreign currency exchange losses.
| Item 8. | Financial Statements and Supplementary Data. |
All information required by this item is included in Item 15 of Part IV of this Annual Report on Form 10-K and is incorporated into this item by reference.
| Item 9. | Changes in and Disagreements with Accountants on Accounting and Financial Disclosure. |
None.
| Item 9A. | Controls and Procedures. |
Evaluation of Disclosure Controls and Procedures
We maintain “disclosure controls and procedures,” as such term is defined in Rule 13a-15(e) under the Exchange Act, that are designed to ensure that information required to be disclosed by us in the reports that we file or submit under the Exchange Act is recorded, processed, summarized and reported within the time periods
53
Table of Contents
specified in the SEC rules and forms and that such information is communicated to our management, including our Chief Executive Officer (principal executive officer) and Chief Financial Officer (principal financial officer), as appropriate to allow timely decisions regarding required disclosure. In designing and evaluating our disclosure controls and procedures, management recognizes that disclosure controls and procedures, no matter how well designed and operated, can provide only reasonable, but not absolute, assurance that the objectives of the disclosure controls and procedures are met. Our disclosure controls and procedures have been designed to meet the reasonable assurance standards. Additionally, in designing disclosure controls and procedures, our management necessarily was required to apply its judgment in evaluating the cost-benefit relationship of possible disclosure controls and procedures. The design of any disclosure controls and procedures is also based in part upon certain assumptions about the likelihood of future events, and there can be no assurance that any design will succeed in achieving its stated goals under all potential future conditions.
As required by Rule 13a-15(b) or Rule 15d-15(b) of the Exchange Act, we carried out an evaluation, under the supervision and with the participation of our management, including our Chief Executive Officer (principal executive officer) and Chief Financial Officer (principal financial officer), of the effectiveness of the design and operation of our disclosure controls and procedures as of December 31, 2011. Based on the foregoing, our Chief Executive Officer (principal executive officer) and Chief Financial Officer (principal financial officer) concluded that our disclosure controls and procedures were effective as of December 31, 2011 at the reasonable assurance level.
Management’s Report on Internal Control over Financial Reporting
Our management is responsible for establishing and maintaining adequate internal control over financial reporting, as such term is defined in Exchange Act Rule 13a-15(f). Management, with the participation of our principal executive officer and principal financial officer, has conducted an evaluation of the effectiveness of our internal control over financial reporting based on the framework set forth in Internal Control—Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission. Based on our evaluation under the framework set forth in Internal Control—Integrated Framework, our management concluded that our internal control over financial reporting was effective as of December 31, 2011.
The effectiveness of our internal control over financial reporting as of December 31, 2011 has been audited by PricewaterhouseCoopers LLP, an independent registered public accounting firm, as stated in their report which appears elsewhere herein.
Changes in Internal Control Over Financial Reporting
There were no changes in internal control over financial reporting during the quarter ended December 31, 2011 that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
| Item 9B. | Other Information. |
None.
54
Table of Contents
PART III
The information required by Part III is omitted from this Annual Report on Form 10-K since we intend to file our definitive Proxy Statement for our 2012 Annual Meeting of Stockholders, pursuant to Regulation 14A of the Exchange Act, not later than 120 days after the end of the fiscal year covered by this Annual Report on Form 10-K, and certain information to be included in the Proxy Statement is incorporated herein by reference.
| Item 10. | Directors, Executive Officers and Corporate Governance. |
Information required by this item regarding directors and director nominees, executive officers, the board of directors and its committees, and certain corporate governance matters is incorporated by reference to the information set forth under the captions “Election of Directors,” “Information Regarding the Board of Directors and Corporate Governance” and “Executive Officers” in our Proxy Statement for the 2012 Annual Meeting of Stockholders. Information required by this item regarding compliance with Section 16(a) of the Exchange Act is incorporated by reference to the information set forth under the caption “Section 16(a) Beneficial Ownership Reporting Compliance” in our Proxy Statement for the 2012 Annual Meeting of Stockholders.
We have adopted a written code of business conduct and ethics that applies to our principal executive officer, principal financial officer, principal accounting officer or controller, or persons serving similar functions. The code of business conduct and ethics is available on our corporate website at www.medivation.com. If we make any substantive amendments to our code of business conduct and ethics or grant to any of our directors or executive officers any waiver, including any implicit waiver, from a provision of our code of business conduct and ethics, we will disclose the nature of the waiver or amendment on our website or in a Current Report on Form 8-K.
| Item 11. | Executive Compensation. |
Information required by this item regarding executive compensation is incorporated by reference to the information set forth under the captions “Executive Compensation,” “Director Compensation” and “Information Regarding the Board of Directors and Corporate Governance” in our Proxy Statement for the 2012 Annual Meeting of Stockholders.
| Item 12. | Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters. |
Information required by this item regarding security ownership of certain beneficial owners and management is incorporated by reference to the information set forth under the caption “Security Ownership of Certain Beneficial Owners and Management” in our Proxy Statement for the 2012 Annual Meeting of Stockholders. Information required by this item regarding securities authorized for issuance under our equity compensation plans is incorporated by reference to the information set forth under the caption “Equity Compensation Plan Information” in our Proxy Statement for the 2012 Annual Meeting of Stockholders.
| Item 13. | Certain Relationships and Related Transactions, and Director Independence. |
Information required by this item regarding certain relationships and related transactions is incorporated by reference to the information set forth under the caption “Transactions with Related Persons” in our Proxy Statement for the 2012 Annual Meeting of Stockholders. Information required by this item regarding director independence is incorporated by reference to the information set forth under the caption “Information Regarding the Board of Directors and Corporate Governance” in our Proxy Statement for the 2012 Annual Meeting of Stockholders.
| Item 14. | Principal Accounting Fees and Services. |
Information required by this item regarding principal accounting fees and services is incorporated by reference to the information set forth under the caption “Ratification of Selection of Independent Registered Public Accounting Firm” in our Proxy Statement for the 2012 Annual Meeting of Stockholders.
55
Table of Contents
PART IV
| Item 15. | Exhibits, Financial Statement Schedules. |
(a) The following documents are filed as part of this Annual Report on Form 10-K:
1. Financial Statements. Our consolidated financial statements and the Report of Independent Registered Public Accounting Firm, are included herein on the pages indicated:
2. Financial Statement Schedules: None.
3. Exhibits:
|
|
Incorporated By Reference | |||||||||||||||||
| Exhibit |
Exhibit Description |
Form |
File No. |
Exhibit | Filing Date |
Filed Herewith | ||||||||||||
| 3.1 | Amended and Restated Certificate of Incorporation. | 10-QSB | 000-20837 | 3.1(a) | 8/15/2005 | |||||||||||||
| 3.2 | Certificate of Amendment of Amended and Restated Certificate of Incorporation. | 10-QSB | 000-20837 | 3.1(b) | 8/15/2005 | |||||||||||||
| 3.3 | Certificate of Amendment to the Amended and Restated Certificate of Incorporation. | 10-QSB | 000-20837 | 3.1(c) | 8/15/2005 | |||||||||||||
| 3.4 | Certificate of Designations of the Series C Junior Participating Preferred Stock of Medivation, Inc. | 10-KSB | 001-32836 | 3.1(d) | 2/19/2008 | |||||||||||||
| 3.5 | Amended and Restated Bylaws of Medivation, Inc. | 10-K | 001-32836 | 3.2 | 3/16/2009 | |||||||||||||
| 4.1 | Common Stock Certificate. | SB-2/A | 333-03252 | 4.1 | 6/14/1996 | |||||||||||||
| 4.2 | Rights Agreement, dated as of December 4, 2006, between Medivation, Inc. and American Stock Transfer & Trust Company, as Rights Agent, which includes the form of Certificate of Designations of the Series C Junior Participating Preferred Stock of Medivation, Inc. as Exhibit A, the form of Right Certificate as Exhibit B and the Summary of Rights to Purchase Preferred Shares as Exhibit C. | 8-K | 001-32836 | 4.1 | 12/4/2006 | |||||||||||||
56
Table of Contents
|
|
Incorporated By Reference | |||||||||||||||||||
| Exhibit |
Exhibit Description |
Form |
File No. |
Exhibit | Filing Date |
Filed Herewith |
||||||||||||||
| 10.1 | Warrant to purchase Common Stock of Medivation Neurology, Inc. assumed by Orion Acquisition Corp. II issued to Joseph J. Grano, Jr., dated as of June 8, 2004. | SB-2 | 333-122431 | 10.5(a) | 1/31/2005 | |||||||||||||||
| 10.2* | Warrant to purchase Common Stock of Medivation Neurology, Inc. assumed by Orion Acquisition Corp. II issued to David T. Hung, M.D., dated as of November 16, 2004. | SB-2 | 333-122431 | 10.6 | 1/31/2005 | |||||||||||||||
| 10.3* | Amended and Restated 2004 Equity Incentive Award Plan. | 10-KSB | 001-32836 | 10.4(a) | 2/19/2008 | |||||||||||||||
| 10.4* | Form of Stock Option Agreement under the 2004 Equity Incentive Award Plan. | 10-KSB | 000-20837 | 10.7(b) | 2/11/2005 | |||||||||||||||
| 10.5* | Form of Stock Option Agreement for Early Exercisable Options under the 2004 Equity Incentive Award Plan. | 10-KSB | 000-20837 | 10.7(c) | 2/11/2005 | |||||||||||||||
| 10.6** | Amended and Restated Collaboration Agreement, dated as of October 20, 2008, between Medivation, Inc. and Pfizer Inc. | 10-Q | 001-32836 | 10.8 | 11/10/2008 | |||||||||||||||
| 10.7* | Change of Control Severance Benefits Agreement, dated as of February 2, 2009, between Medivation, Inc. and David Hung, M.D. | 10-K | 001-32836 | 10.11 | 3/16/2009 | |||||||||||||||
| 10.8* | Form of Medivation, Inc. Change of Control Severance Benefits Agreement. | 10-K | 001-32836 | 10.13 | 3/16/2009 | |||||||||||||||
| 10.9** | Collaboration Agreement, dated as of October 26, 2009, by and between Medivation, Inc. and Astellas US LLC. | 10-K | 001-32836 | 10.15 | 3/15/2010 | |||||||||||||||
| 10.10** | Amendment No. 1 to Collaboration Agreement, dated January 1, 2010, by and among Medivation, Inc., Astellas Pharma Inc. and Astellas US LLC | 10-Q | 001-32836 | 10.1 | 8/9/2011 | |||||||||||||||
| 10.11** | Amendment No. 2 to Collaboration Agreement, dated May 13, 2011, by and among Medivation, Inc., Astellas Pharma Inc. and Astellas US LLC | 10-Q | 001-32836 | 10.2 | 8/9/2011 | |||||||||||||||
| 10.12 | Office Lease Agreement, dated as of November 2, 2009, by and between Medivation, Inc. and PPF OFF 345 Spear Street, LP. | 10-K | 001-32836 | 10.16 | 3/15/2010 | |||||||||||||||
| 10.13* | Compensation Information for Non-Employee Directors. | X | ||||||||||||||||||
57
Table of Contents
|
|
Incorporated By Reference | |||||||||||||||||||
| Exhibit |
Exhibit Description |
Form |
File No. |
Exhibit | Filing Date |
Filed Herewith |
||||||||||||||
| 10.14** | Exclusive License Agreement, dated as of August 12, 2005, as amended through October 21, 2009, by and between Medivation, Inc. and The Regents of the University of California. | 10-Q/A | 001-32836 | 10.18 | 8/20/2010 | |||||||||||||||
| 10.15 | Office Lease, dated April 18, 2007, by and between CREA Spear Street Terrace LLC and Medivation, Inc. | 10-K | 001-32836 | 10.16 | 3/16/2011 | |||||||||||||||
| 10.16 | Sublease, dated November 10, 2008, by and between MacFarlane Partners Investment Management, LLC and Medivation, Inc. | 10-K | 001-32836 | 10.17 | 3/16/2011 | |||||||||||||||
| 10.17 | First Amendment to Lease, dated September 16, 2009, by and between CREA Spear Street Terrace LLC and Medivation, Inc. | 10-K | 001-32836 | 10.18 | 3/16/2011 | |||||||||||||||
| 10.18 | Second Amendment to Lease, dated November 30, 2010, by and between CREA Spear Street Terrace LLC and Medivation, Inc. | 10-K | 001-32836 | 10.19 | 3/16/2011 | |||||||||||||||
| 10.19* | Bonuses for Fiscal Year 2010 and Base Salaries for Fiscal Year 2011 for Certain Executive Officers. | 10-K | 001-32836 | 10.20 | 3/16/2011 | |||||||||||||||
| 10.20* | Medivation, Inc. 2011 Bonus Plan Summary. | 10-K | 001-32836 | 10.21 | 3/16/2011 | |||||||||||||||
| 10.21* | Form of Restricted Stock Unit Grant Notice and Agreement under the 2004 Equity Incentive Award Plan. | 10-K | 001-32836 | 10.22 | 3/16/2011 | |||||||||||||||
| 10.22* | Separation Agreement, dated as of September 21, 2011, between Medivation, Inc. and Rohan Palekar. | 8-K | 001-32836 | 10.1 | 9/27/2011 | |||||||||||||||
| 10.23* | Bonuses for Fiscal Year 2011 and Base Salaries for Fiscal Year 2012 for Certain Executive Officers | 8-K | 001-32836 | 10.1 | 12/15/2011 | |||||||||||||||
| 10.24* | Medivation, Inc. 2012 Bonus Plan Summary | 8-K | 001-32836 | 10.2 | 12/15/2011 | |||||||||||||||
| 10.25* | Form of Indemnification Agreement for directors and officers | X | ||||||||||||||||||
| 10.26* | Offer Letter, dated August 31, 2011, by and between Medivation, Inc. and Cheryl Cohen. | X | ||||||||||||||||||
| 10.27 | Office Lease, dated as of December 28, 2011, by and between Knickerbocker Properties, Inc. XXXIII and Medivation, Inc. | X | ||||||||||||||||||
| 10.28* | Form of Stock Appreciation Right Grant Notice and Agreement under the 2004 Equity Incentive Award Plan | X | ||||||||||||||||||
58
Table of Contents
|
|
Incorporated By Reference | |||||||||||||
| Exhibit |
Exhibit Description |
Form |
File No. |
Exhibit | Filing Date |
Filed Herewith |
||||||||
| 10.29* | Form of Performance Share Award Grant Notice and Agreement under the 2004 Equity Incentive Award Plan | X | ||||||||||||
| 21.1 | Subsidiaries of Medivation, Inc. | X | ||||||||||||
| 23.1 | Consent of Independent Registered Public Accounting Firm. | X | ||||||||||||
| 24.1 | Power of Attorney (contained on signature page). | X | ||||||||||||
| 31.1 | Certification pursuant to Rule 13a-14(a)/15d-14(a). | X | ||||||||||||
| 31.2 | Certification pursuant to Rule 13a-14(a)/15d-14(a). | X | ||||||||||||
| 32.1† | Certifications of Chief Executive Officer and Chief Financial Officer. | X | ||||||||||||
| 101.INS# | XBRL Instance Document. | X | ||||||||||||
| 101.SCH# | XBRL Taxonomy Extension Schema Document. | X | ||||||||||||
| 101.CAL# | XBRL Taxonomy Extension Calculation Linkbase Document | X | ||||||||||||
| 101.DEF# | XBRL Taxonomy Extension Definition Linkbase Document | X | ||||||||||||
| 101.LAB# | XBRL Taxonomy Extension Labels Linkbase Document | X | ||||||||||||
| 101.PRE# | XBRL Taxonomy Extension Presentation Linkbase Document | X | ||||||||||||
| * | Indicates management contract or compensatory plan or arrangement. |
| ** | Confidential treatment has been granted with respect to certain portions of this exhibit. |
| † | The certifications attached as Exhibit 32.1 accompany this Annual Report on Form 10-K, are not deemed filed with the Securities and Exchange Commission and are not to be incorporated by reference into any filing of Medivation, Inc., under the Securities Act of 1933, as amended, or the Securities Exchange Act of 1934, as amended, whether made before or after the date of this Annual Report on Form 10-K, irrespective of any general incorporation language contained in such filing. |
| # | Pursuant to applicable securities laws and regulations, the Registrant is deemed to have complied with the reporting obligation related to the submission of interactive data files in such exhibits and is not subject to liability under any anti-fraud provisions of the federal securities laws as long as the Registrant has made a good faith attempt to comply with the submission requirements and promptly amends the interactive data files after becoming aware that the interactive data files fails to comply with the submission requirements. These interactive data files are deemed not filed or part of a registration statement or prospectus for purposes of sections 11 or 12 of the Securities Act, are deemed not filed for the purposes of section 18 of the Exchange Act and otherwise are not subject to liability under those sections. |
59
Table of Contents
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the Registrant has duly caused this Report to be signed on its behalf by the undersigned, thereunto duly authorized.
| MEDIVATION, INC. | ||
| /S/ C. PATRICK MACHADO | ||
| C. Patrick Machado Chief Business Officer and Chief Financial Officer (Duly Authorized and Principal Financial and Accounting Officer) | ||
Dated: February 29, 2012
POWER OF ATTORNEY
KNOW ALL PERSONS BY THESE PRESENTS, that each person whose signature appears below constitutes and appoints David T. Hung, M.D. and C. Patrick Machado, and each of them, as his true and lawful attorneys-in-fact and agents, with full power of substitution and resubstitution, for him and in his name, place, and stead, in any and all capacities, to sign any and all amendments to this Report, and to file the same, with all exhibits thereto, and other documents in connection therewith, with the Securities and Exchange Commission, granting unto said attorneys-in-fact and agents, and each of them, full power and authority to do and perform each and every act and thing requisite and necessary to be done in connection therewith, as fully to all intents and purposes as he might or could do in person, hereby ratifying and confirming that all said attorneys-in-fact and agents, or any of them or their or his substitute or substitutes, may lawfully do or cause to be done by virtue hereof.
Pursuant to the requirements of the Securities Exchange Act of 1934, this Report has been signed below by the following persons on behalf of the Registrant and in the capacities and on the dates indicated:
| /S/ DAVID T. HUNG, M.D. David T. Hung, M.D. |
President, Chief Executive Officer and Director (Principal Executive Officer) |
February 29, 2012 | ||
| /S/ C. PATRICK MACHADO C. Patrick Machado |
Chief Business Officer and Chief Financial Officer (Principal Financial and Accounting Officer) | February 29, 2012 | ||
| /S/ DANIEL D. ADAMS Daniel D. Adams |
Director | February 29, 2012 | ||
| /S/ GREGORY H. BAILEY Gregory H. Bailey |
Director | February 29, 2012 | ||
| /S/ KIM D. BLICKENSTAFF Kim D. Blickenstaff |
Director | February 29, 2012 | ||
|
W. Anthony Vernon |
Director | |||
60
Table of Contents
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Board of Directors and Stockholders of
Medivation, Inc.
In our opinion, the accompanying consolidated balance sheets and the related consolidated statements of operations, of cash flows and of stockholders’ equity present fairly, in all material respects, the financial position of Medivation, Inc. and its subsidiaries (the “Company”) at December 31, 2011 and December 31, 2010 and the results of their operations and their cash flows for each of the three years in the period ended December 31, 2011 in conformity with accounting principles generally accepted in the United States of America. Also in our opinion, the Company maintained, in all material respects, effective internal control over financial reporting as of December 31, 2011, based on criteria established in Internal Control—Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (COSO). The Company’s management is responsible for these financial statements, for maintaining effective internal control over financial reporting and for its assessment of the effectiveness of internal control over financial reporting, included in Management’s Report on Internal Control over Financial Reporting appearing under Item 9A. Our responsibility is to express opinions on these financial statements and on the Company’s internal control over financial reporting based on our integrated audits. We conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audits to obtain reasonable assurance about whether the financial statements are free of material misstatement and whether effective internal control over financial reporting was maintained in all material respects. Our audits of the financial statements included examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements, assessing the accounting principles used and significant estimates made by management, and evaluating the overall financial statement presentation. Our audit of internal control over financial reporting included obtaining an understanding of internal control over financial reporting, assessing the risk that a material weakness exists, and testing and evaluating the design and operating effectiveness of internal control based on the assessed risk. Our audits also included performing such other procedures as we considered necessary in the circumstances. We believe that our audits provide a reasonable basis for our opinions.
A company’s internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A company’s internal control over financial reporting includes those policies and procedures that (i) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (ii) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only in accordance with authorizations of management and directors of the company; and (iii) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of the Company’s assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
/s/ PricewaterhouseCoopers LLP
San Jose, California
February 29, 2012
61
Table of Contents
CONSOLIDATED BALANCE SHEETS
(in thousands, except share and per share data)
| December 31, 2011 |
December 31, 2010 |
|||||||
| ASSETS |
||||||||
| Current assets: |
||||||||
| Cash and cash equivalents |
$ | 70,136 | $ | 107,717 | ||||
| Short-term investments |
74,996 | 100,039 | ||||||
| Receivable from collaboration partners |
12,545 | 21,188 | ||||||
| Prepaid expenses and other current assets |
10,512 | 8,067 | ||||||
|
|
|
|
|
|||||
| Total current assets |
168,189 | 237,011 | ||||||
| Property and equipment, net |
720 | 862 | ||||||
| Restricted cash |
5,489 | 843 | ||||||
| Other non-current assets |
719 | 887 | ||||||
|
|
|
|
|
|||||
| Total assets |
$ | 175,117 | $ | 239,603 | ||||
|
|
|
|
|
|||||
| LIABILITIES AND STOCKHOLDERS’ EQUITY |
||||||||
| Current liabilities: |
||||||||
| Accounts payable |
$ | 5,588 | $ | 3,229 | ||||
| Accrued expenses |
24,014 | 21,399 | ||||||
| Deferred revenue |
59,762 | 59,153 | ||||||
| Other current liabilities |
270 | 5,193 | ||||||
|
|
|
|
|
|||||
| Total current liabilities |
89,634 | 88,974 | ||||||
| Deferred revenue, net of current |
83,509 | 141,507 | ||||||
| Other non-current liabilities |
653 | 1,438 | ||||||
|
|
|
|
|
|||||
| Total liabilities |
173,796 | 231,919 | ||||||
| Commitments and contingencies (Note 11) |
||||||||
| Stockholders’ equity: |
||||||||
| Preferred stock, $0.01 par value per share; 1,000,000 shares authorized; no shares issued and outstanding |
— | — | ||||||
| Common stock, $0.01 par value per share; 50,000,000 shares authorized; issued and outstanding shares of 35,731,838 at December 31, 2011 and 34,573,829 at December 31, 2010 |
357 | 346 | ||||||
| Additional paid-in capital |
251,242 | 218,786 | ||||||
| Accumulated other comprehensive gain |
13 | 2 | ||||||
| Accumulated deficit |
(250,291 | ) | (211,450 | ) | ||||
|
|
|
|
|
|||||
| Total stockholders’ equity |
1,321 | 7,684 | ||||||
|
|
|
|
|
|||||
| Total liabilities and stockholders’ equity |
$ | 175,117 | $ | 239,603 | ||||
|
|
|
|
|
|||||
SEE ACCOMPANYING NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
62
Table of Contents
CONSOLIDATED STATEMENTS OF OPERATIONS
(in thousands, except per share data)
| Years ended December 31, | ||||||||||||
| 2011 | 2010 | 2009 | ||||||||||
| Collaboration revenue |
$ | 60,389 | $ | 62,508 | $ | 69,254 | ||||||
| Operating expenses: |
||||||||||||
| Research and development |
73,432 | 72,228 | 87,728 | |||||||||
| Selling, general and administrative |
29,887 | 23,005 | 28,983 | |||||||||
|
|
|
|
|
|
|
|||||||
| Total operating expenses |
103,319 | 95,233 | 116,711 | |||||||||
|
|
|
|
|
|
|
|||||||
| Loss from operations |
(42,930 | ) | (32,725 | ) | (47,457 | ) | ||||||
| Other (expense) income, net |
(242 | ) | 260 | 976 | ||||||||
|
|
|
|
|
|
|
|||||||
| Net loss before income tax (benefit) expense |
(43,172 | ) | (32,465 | ) | (46,481 | ) | ||||||
| Income tax (benefit) expense |
(4,331 | ) | 1,572 | 8,272 | ||||||||
|
|
|
|
|
|
|
|||||||
| Net loss |
$ | (38,841 | ) | $ | (34,037 | ) | $ | (54,753 | ) | |||
|
|
|
|
|
|
|
|||||||
| Basic and diluted net loss per common share |
$ | (1.11 | ) | $ | (0.99 | ) | $ | (1.71 | ) | |||
|
|
|
|
|
|
|
|||||||
| Weighted-average common shares used in the calculation of basic and diluted net loss per common share |
34,960 | 34,290 | 32,094 | |||||||||
|
|
|
|
|
|
|
|||||||
SEE ACCOMPANYING NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
63
Table of Contents
CONSOLIDATED STATEMENTS OF CASH FLOWS
(in thousands)
| Years ended December 31, | ||||||||||||
| 2011 | 2010 | 2009 | ||||||||||
| Cash flows from operating activities: |
||||||||||||
| Net loss |
$ | (38,841 | ) | $ | (34,037 | ) | $ | (54,753 | ) | |||
| Adjustments to reconcile net loss to net cash flows used in operating activities: |
||||||||||||
| Amortization of deferred revenue |
(60,389 | ) | (62,508 | ) | (69,254 | ) | ||||||
| Stock-based compensation |
13,885 | 13,530 | 10,726 | |||||||||
| Depreciation and amortization |
430 | 465 | 311 | |||||||||
| Accretion of discount on securities |
(65 | ) | (281 | ) | (1,081 | ) | ||||||
| Changes in operating assets and liabilities: |
||||||||||||
| Receivable from collaboration partners |
8,643 | (14,698 | ) | (2,968 | ) | |||||||
| Prepaid expenses and other current assets |
(1,945 | ) | (224 | ) | (5,450 | ) | ||||||
| Other assets |
166 | (322 | ) | 78 | ||||||||
| Accounts payable |
2,359 | (1,611 | ) | (2,326 | ) | |||||||
| Accrued expenses |
2,504 | 9,345 | 6,282 | |||||||||
| Other current liabilities |
(4,923 | ) | 4,393 | 706 | ||||||||
| Deferred revenue |
3,000 | 10,000 | 110,000 | |||||||||
| Other non-current liabilities |
(785 | ) | 884 | 144 | ||||||||
|
|
|
|
|
|
|
|||||||
| Net cash used in operating activities |
(75,961 | ) | (75,064 | ) | (7,585 | ) | ||||||
|
|
|
|
|
|
|
|||||||
| Cash flows from investing activities: |
||||||||||||
| Purchase of short-term investments |
(74,956 | ) | (209,888 | ) | (342,437 | ) | ||||||
| Maturities of short-term investments |
100,075 | 331,000 | 272,000 | |||||||||
| Purchase of property and equipment |
(286 | ) | (197 | ) | (631 | ) | ||||||
| Change in restricted cash |
(5,146 | ) | 1,500 | (1,500 | ) | |||||||
|
|
|
|
|
|
|
|||||||
| Net cash provided by (used in) investing activities |
19,687 | 122,415 | (72,568 | ) | ||||||||
|
|
|
|
|
|
|
|||||||
| Cash flows from financing activities: |
||||||||||||
| Proceeds from stock option and warrant exercises |
15,734 | 2,625 | 3,389 | |||||||||
| Net proceeds from stockholder securities law settlement |
2,959 | — | — | |||||||||
| Proceeds from issuance of common stock, net of issuance costs |
— | — | 62,059 | |||||||||
| Excess tax benefits from stock-based compensation |
— | 278 | 714 | |||||||||
|
|
|
|
|
|
|
|||||||
| Net cash provided by financing activities |
18,693 | 2,903 | 66,162 | |||||||||
|
|
|
|
|
|
|
|||||||
| Net (decrease) increase in cash and cash equivalents |
(37,581 | ) | 50,254 | (13,991 | ) | |||||||
| Cash and cash equivalents at beginning of year |
107,717 | 57,463 | 71,454 | |||||||||
|
|
|
|
|
|
|
|||||||
| Cash and cash equivalents at end of year |
$ | 70,136 | $ | 107,717 | $ | 57,463 | ||||||
|
|
|
|
|
|
|
|||||||
| Supplemental disclosures of cash flow information: |
||||||||||||
| Income taxes paid |
$ | 5,358 | $ | — | $ | 8,400 | ||||||
| Receivable from stock option exercises |
$ | — | $ | — | $ | 436 | ||||||
SEE ACCOMPANYING NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
64
Table of Contents
CONSOLIDATED STATEMENTS OF STOCKHOLDERS’ EQUITY
(in thousands, except share data)
| COMMON STOCK | ADDITIONAL PAID-IN CAPITAL |
ACCUMULATED OTHER COMPREHENSIVE INCOME |
ACCUMULATED DEFICIT |
TOTAL STOCKHOLDERS’ EQUITY |
||||||||||||||||||||
| SHARES | AMOUNT | |||||||||||||||||||||||
| Balances at January 1, 2009 |
30,088,390 | $ | 301 | $ | 125,074 | $ | 693 | $ | (122,660 | ) | $ | 3,408 | ||||||||||||
| Common stock issued: |
||||||||||||||||||||||||
| In the June 2009 financing |
3,162,500 | 32 | 62,396 | — | — | 62,428 | ||||||||||||||||||
| Upon exercise of stock options and warrants |
553,006 | 5 | 3,820 | — | — | 3,825 | ||||||||||||||||||
| Upon vesting of restricted stock units |
19,166 | — | — | — | — | — | ||||||||||||||||||
| Offering expenses |
— | — | (369 | ) | — | — | (369 | ) | ||||||||||||||||
| Stock-based compensation expense |
— | — | 10,726 | — | — | 10,726 | ||||||||||||||||||
| Excess tax benefit from employee stock plan awards |
— | — | 714 | — | — | 714 | ||||||||||||||||||
| Net loss |
— | — | — | — | (54,753 | ) | (54,753 | ) | ||||||||||||||||
| Change in unrealized gain (loss) on available-for-sale securities |
— | — | — | (705 | ) | — | (705 | ) | ||||||||||||||||
|
|
|
|||||||||||||||||||||||
| Comprehensive loss |
— | — | — | — | — | (55,458 | ) | |||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
| Balances at December 31, 2009 |
33,823,062 | 338 | 202,361 | (12 | ) | (177,413 | ) | 25,274 | ||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
| Common stock issued: |
||||||||||||||||||||||||
| Upon exercise of stock options and warrants |
740,767 | 8 | 2,617 | — | — | 2,625 | ||||||||||||||||||
| Upon vesting of restricted stock units |
10,000 | — | — | — | — | — | ||||||||||||||||||
| Stock-based compensation expense |
— | — | 13,530 | — | — | 13,530 | ||||||||||||||||||
| Excess tax benefit from employee stock plan awards |
— | — | 278 | — | — | 278 | ||||||||||||||||||
| Net loss |
— | — | — | — | (34,037 | ) | (34,037 | ) | ||||||||||||||||
| Change in unrealized gain (loss) on available-for-sale securities |
— | — | — | 14 | — | 14 | ||||||||||||||||||
|
|
|
|||||||||||||||||||||||
| Comprehensive loss |
— | — | — | — | — | (34,023 | ) | |||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
| Balances at December 31, 2010 |
34,573,829 | 346 | 218,786 | 2 | (211,450 | ) | 7,684 | |||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
| Common stock issued: |
||||||||||||||||||||||||
| Upon exercise of stock options |
1,106,403 | 11 | 15,723 | — | — | 15,734 | ||||||||||||||||||
| Upon vesting of restricted stock units |
51,606 | — | — | — | — | — | ||||||||||||||||||
| Stock-based compensation expense |
— | — | 13,774 | — | — | 13,774 | ||||||||||||||||||
| Net proceeds from stockholder securities law settlement |
— | — | 2,959 | — | — | 2,959 | ||||||||||||||||||
| Net loss |
— | — | — | — | (38,841 | ) | (38,841 | ) | ||||||||||||||||
| Change in unrealized gain on available-for-sale securities |
— | — | — | 11 | — | 11 | ||||||||||||||||||
|
|
|
|||||||||||||||||||||||
| Comprehensive loss |
— | — | — | — | — | (38,830 | ) | |||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
| Balances at December 31, 2011 |
35,731,838 | $ | 357 | $ | 251,242 | $ | 13 | $ | (250,291 | ) | $ | 1,321 | ||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
SEE ACCOMPANYING NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
65
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
December 31, 2011
1. DESCRIPTION OF BUSINESS
Medivation, Inc. (the “Company” or “Medivation”) is a biopharmaceutical company focused on the rapid development of novel small molecule drugs to treat serious diseases for which there are limited treatment options. Together with its collaboration partner, Astellas Pharma, Inc., or Astellas, the Company is developing MDV3100 for multiple stages of advanced prostate cancer. The Company has completed the Phase 3 AFFIRM trial in the latest stage patients – those who have already failed docetaxel-based chemotherapy – and is conducting an additional Phase 3 and two Phase 2 trials in men with earlier stages of advanced prostate cancer.
The Company has not generated any revenue from product sales to date. The Company has funded its operations primarily through private and public offerings of its common stock and from the up-front, development milestone and cost-sharing payments under its collaboration agreement with Astellas and its former collaboration agreement with Pfizer Inc., or Pfizer. The Company has incurred cumulative net losses of $250.3 million through December 31, 2011 and it expects to incur substantial additional losses for the foreseeable future as it pursues regulatory approval for, and, if approved, commercial launch of MDV3100 and continues to finance clinical and preclinical studies of its existing and potential future product candidates and its corporate overhead costs.
(a) Astellas Collaboration Agreement
The Company entered into a collaboration agreement with Astellas in October 2009, pursuant to which it received a non-refundable up-front cash payment of $110.0 million in the fourth quarter of 2009. The Company subsequently received development milestone payments of $10.0 million and $3.0 million in the fourth quarter of 2010 and the second quarter of 2011, respectively. The Company recorded development and commercialization cost-sharing payments from Astellas totaling $43.8 million, $34.6 million and $2.9 million during the years ended December 31, 2011, 2010 and 2009, respectively, pursuant to the terms of the Astellas Collaboration Agreement, which is discussed further in Note 3, “Collaboration Agreements.” The Company refers to its collaboration agreement with Astellas as the Astellas Collaboration Agreement.
In November 2011, the Company reported positive results from a planned interim analysis of its Phase 3 AFFIRM trial in advanced prostate cancer patients previously treated with docetaxel-based chemotherapy. In February 2012, the Company reported further positive results from the AFFIRM trial at the American Society of Clinical Oncology’s 2012 Genitourinary Cancers Symposium. Based on the positive results from the AFFIRM trial, the Company elected to exercise its right under the Astellas Collaboration Agreement to co-promote MDV3100 in the U.S. market. Should MDV3100 receive marketing approval, the Company will provide 50% of the sales and medical affairs field forces supporting MDV3100 in the U.S. market.
(b) Former Collaboration Agreement with Pfizer
The Company entered into a collaboration agreement with Pfizer in October 2008, pursuant to which it received a non-refundable up-front cash payment of $225.0 million in the fourth quarter of 2008. The Company recorded development and commercialization cost-sharing payments from Pfizer totaling $12.4 million, $28.1 million and $19.7 million during the years ended December 31, 2011, 2010 and 2009, respectively, pursuant to the terms of the collaboration agreement, which is discussed further in Note 3, “Collaboration Agreements.”
In January 2012, the Company reported negative top line results from its Phase 3 CONCERT trial of dimebon in patients with mild-to-moderate Alzheimer’s disease. The Company previously had reported negative top line results from its Phase 3 CONNECTION trial of dimebon in patients with mild-to-moderate Alzheimer’s
66
Table of Contents
disease and its Phase 3 HORIZON trial of dimebon in patients with Huntington disease. In January 2012, Pfizer exercised its right to terminate the collaboration agreement and the Company and Pfizer discontinued the development of dimebon for all indications in 2012 as discussed further in Note 14, “Subsequent Events.” The Company refers to its collaboration agreement with Pfizer as the former collaboration agreement with Pfizer.
2. SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
(a) Basis of Consolidation; Business Segments
The consolidated financial statements incorporate the accounts of Medivation, Inc. and its wholly-owned subsidiaries. All significant inter-company transactions and balances have been eliminated in consolidation. The Company operates in one business segment.
All tabular disclosures of dollar amounts are presented in thousands. All share and per share amounts are presented at their actual amounts.
(b) Use of Estimates
The preparation of consolidated financial statements in conformity with accounting principles generally accepted in the United States, or U.S. GAAP, requires that management make estimates and assumptions in certain circumstances that affect the reported amounts of assets and liabilities and disclosures of contingent assets and liabilities at the date of the financial statements and the reported amounts of revenues and expenses during the reporting period. Estimates and assumptions used by management principally relate to the performance periods of the Company’s deliverables under its collaboration agreement with Astellas and its former collaboration agreement with Pfizer; services performed by third parties but not yet invoiced; the fair value and forfeiture rates of stock awards granted to employees, directors and consultants and the probability of attaining the performance objectives of performance share awards; the probability and potential magnitude of contingent liabilities; and deferred income taxes, income tax provisions and accruals for uncertain income tax positions.
Although management believes that estimates used in the preparation of the consolidated financial statements are reasonable under the circumstances, actual future results could differ from those estimates. In addition, had different estimates and assumptions been used, the consolidated financial statements could have differed materially from that which is presented.
(c) Risks and Uncertainties
The Company has incurred cumulative net losses of $250.3 million through December 31, 2011 and it expects to incur substantial additional losses for the foreseeable future as it pursues regulatory approval for, and, if approved, commercial launch of MDV3100 and continues to finance clinical and preclinical studies of its existing and potential future product candidates and its corporate overhead costs. At December 31, 2011, the Company had cash, cash equivalents and short-term investments totaling $145.1 million to fund its operations. The Company expects that its current resources will be sufficient to fund the presently budgeted costs of executing its current development and commercialization plan for the next 12 months. However, this statement is subject to significant risks and uncertainty and is based on a number of assumptions that may prove to be wrong and the Company could exhaust its available cash, cash equivalents and short-term investments earlier than presently anticipated. The Company may be required or choose to seek additional capital within the next 12 months to expand its clinical development activities for MDV3100 based on the positive results of the Company’s Phase 3 AFFIRM trial in post-chemotherapy advanced prostate cancer patients, to fund costs of planning for and executing a commercial launch of MDV3100, should it receive marketing approval, if the Company faces challenges or delays in connection with its clinical trials or the potential approval and commercialization of MDV3100, or to maintain cash balances that the Company deems reasonable and prudent. The Company’s ability to raise capital on acceptable terms, or at all, is subject to significant risks, many of which are beyond its control.
67
Table of Contents
(d) Cash Equivalents
Cash and cash equivalents are stated at cost, which approximates fair market value. The Company considers all highly liquid investments with a remaining maturity of three months or less at the time of acquisition to be cash equivalents.
(e) Short-Term Investments
The Company considers all highly liquid investments with a remaining maturity at the time of acquisition of more than three months but no longer than 12 months to be short-term investments. The Company classifies its short-term investments as available-for-sale securities and reports them at fair value with related unrealized gains and losses included as a component of stockholders’ equity. The amortized cost of debt securities in this category is adjusted for amortization of premiums and accretion of discounts to maturity, which is included in other (expense) income, net, on the consolidated statements of operations. Realized gains and losses and declines in value judged to be other-than-temporary, if any, on available-for-sale securities are included in other (expense) income, net. The cost of securities sold is based on the specific identification method. Interest and dividends on securities classified as available-for-sale are included in other (expense) income, net.
(f) Restricted Cash
Restricted cash represents certificates of deposit held in the Company’s name with a major financial institution to secure the Company’s contingent obligations under irrevocable letters of credit issued to the lessors of the Company’s office facilities.
(g) Fair Value of Financial Instruments
The fair value of the Company’s cash equivalents and short-term investments is based on quoted market prices. Other financial instruments, including receivables from collaboration partners, accounts payable and accrued expenses, are carried at cost, which the Company believes approximates fair value because of the short-term maturities of these instruments.
(h) Concentration of Credit Risk
Financial instruments that potentially subject the Company to credit risk consist of cash, cash equivalents, short-term investments and receivables from collaboration partners. The Company’s current investment policy is to invest only in a) debt securities issued by, or backed by the full faith and credit of, the U.S. government, b) repurchase agreements that are fully collateralized by such debt securities, and c) money market funds invested exclusively in the types of securities described in a) and b) above. Given this investment policy, the Company does not believe its exposure to credit risk with respect to the issuers of the securities in which it invests is material, and accordingly has no formal policy for mitigating such risk. The Company’s cash and cash equivalents are primarily invested in deposits and money market accounts with one major financial institution in the United States. Deposits in this financial institution may exceed the amount of insurance provided on such deposits.
(i) Property and Equipment
Property and equipment are recorded at cost, less accumulated depreciation and amortization. Repairs and maintenance costs are expensed in the period incurred. Property and equipment is depreciated on a straight-line basis over the estimated useful lives of the assets as follows:
| Description |
Estimated Useful Life | |
| Office equipment and furniture |
3 years | |
| Software and computer equipment |
3-5 years | |
| Laboratory equipment |
5 years | |
| Leasehold improvements and fixtures |
Lesser of estimated useful life or life of lease |
68
Table of Contents
(j) Leases
At the inception of a lease, the Company evaluates the lease agreement to determine whether the lease is an operating or a capital lease using the criteria in the Financial Accounting Standards Board, or FASB, Accounting Standards Codification, or ASC, 840, “Leases.” For operating leases, the Company recognizes rent expense on a straight-line basis over the non-cancellable lease term and records the difference between cash rent payments and the recognition of rent expense as a deferred liability. Where lease agreements contain rent escalation clauses, rent abatements and/or concessions, such as rent holidays and tenant improvement allowances, the Company applies them in the determination of straight-line expense over the lease term. The Company currently has no capital leases. Certain lease agreements also require the Company to make additional payments under the lease term for taxes, insurance, and other operating expenses incurred during the lease period, which are expensed as incurred.
(k) Litigation
The Company records an accrual for exposures to various litigation matters as a charge to its consolidated statements of operations when it becomes probable and can be reasonably estimated. The exposure to legal matters is evaluated and estimated, if possible, following consultation with legal counsel. Such estimates are based on currently available information and, given the subjective nature and complexities inherent in making these estimates, the ultimate outcome of the Company’s legal matters may be different than the amounts estimated, if any.
(l) Collaboration Agreement Payments
The Company accounts for the various payment flows under its collaboration agreement with Astellas and its former collaboration agreement with Pfizer in a consistent manner, as follows:
Estimated Performance Periods
Both the Astellas Collaboration Agreement and the former collaboration agreement with Pfizer contain multiple elements and deliverables, and required evaluation pursuant to ASC 605-25, “Revenue Recognition—Multiple-Element Arrangements.” The Company evaluated the facts and circumstances of the collaboration agreements to determine whether it had obligations constituting deliverables under ASC 605-25. The Company concluded that it had multiple deliverables under both collaboration agreements, including deliverables relating to grants of technology licenses, and performance of manufacturing, regulatory and clinical development activities in the U.S. In the case of the Astellas Collaboration Agreement, the period in which the Company performs its deliverables began in the fourth quarter of 2009 and at December 31, 2011, management estimated that it would be completed in the fourth quarter of 2014. In the case of the former collaboration agreement with Pfizer, the period in which the Company performs its deliverables began in the fourth quarter of 2008 and at December 31, 2011, management estimated that it would be completed in the fourth quarter of 2013. The Company also concluded that its deliverables under each collaboration agreement should be accounted for as a single unit of accounting under ASC 605-25.
Estimation of the performance periods of the Company’s deliverables requires the use of management’s judgment. Significant factors considered in management’s evaluation of the estimated performance periods include, but are not limited to, the Company’s experience, along with Astellas’ and Pfizer’s experience, in conducting clinical development and regulatory activities. The Company reviews the estimated duration of its performance periods under both collaborations on a quarterly basis and makes any appropriate adjustments on a prospective basis.
During the year ended December 31, 2010, the Company extended the estimated completion date of its performance period under the former collaboration agreement with Pfizer from the second quarter of 2012 to the fourth quarter of 2013, based on the failure of the CONNECTION trial and the resulting longer period required to
69
Table of Contents
complete the clinical trials evaluating dimebon’s potential safety and efficacy as a treatment for mild-to-moderate Alzheimer’s disease.
In January 2012, Pfizer exercised its right to terminate the collaboration agreement. As a result, during the first quarter of 2012, the Company revised its estimate of the remaining performance period under the collaboration agreement. The Company expects to complete its remaining performance obligations pursuant to the terms of the former collaboration agreement with Pfizer in the first half of 2012.
Future changes in estimates of the performance periods under the collaboration agreements could significantly impact the timing of future revenue recognized under the applicable collaboration agreement.
Up-Front Payments
The Company has received non-refundable up-front cash payments of $110.0 million and $225.0 million under its collaboration agreement with Astellas and its former collaboration agreement with Pfizer, respectively. The Company recognizes these payments as collaboration revenue on a straight-line basis over the applicable estimated performance period.
Milestone Payments
The Company is eligible to receive milestone payments under the Astellas Collaboration Agreement based on achievement of specified development, regulatory and commercial events. Management evaluated the nature of the events triggering these contingent payments, and concluded that these events—except for (a) those relating to regulatory activities in Europe, development and regulatory activities in Japan, and commercial activities, all of which are areas in which the Company has no pertinent contractual responsibilities, and (b) the initiation of the Phase 3 PREVAIL trial, an event which management deemed to be reasonably assured at the inception of the Astellas collaboration—constituted substantive milestones. This conclusion was based primarily on the facts that (i) each triggering event represents a specific outcome that can be achieved only through successful performance by the Company of one or more of its deliverables, (ii) achievement of each triggering event was subject to inherent risk and uncertainty and would result in additional payments becoming due to the Company, (iii) each of these milestones was substantive, based primarily on the facts that the payments they trigger are non-refundable, (iv) achievement of the milestone entails risk and was not reasonably assured at inception of the collaboration agreement, (v) substantial effort is required to complete each milestone, (vi) the amount of each milestone payment is reasonable in relation to the value created in achieving the milestone, (vii) a substantial amount of time is expected to pass between the up-front payment and the potential milestone payments, and (viii) the milestone payments relate solely to past performance. Based on the foregoing, the Company will recognize any revenue from these milestone payments under the substantive milestone method in the period in which the underlying triggering event occurs.
For the contingent payments triggered by events that do not constitute substantive milestones, management concluded that the appropriate revenue recognition treatment depends on whether the triggering event occurs during or after the performance period. Where the triggering event occurs during the applicable performance period, the Company will amortize any revenue from this event on a straight-line basis over the applicable performance period. Where the triggering event occurs after the applicable performance period, the Company will recognize the associated revenue in the period in which the event occurs.
Royalties and Profit Sharing Payments
Under the Astellas Collaboration Agreement, the Company is eligible to receive profit sharing payments on sales of products in the U.S. and royalties on sales of products outside the U.S. The Company will recognize any revenue from these events based on the revenue recognition criteria set forth in ASC 605-10-25-1, “Revenue Recognition.” Based on those criteria, the Company considers these potential payments to be contingent revenues, and will recognize them as revenue in the period in which the applicable contingency is resolved.
70
Table of Contents
Cost-Sharing Payments
Under both the Astellas Collaboration Agreement and the former collaboration agreement with Pfizer, the Company and its collaboration partners share certain development and commercialization costs in the U.S. The parties make quarterly cost-sharing payments to one another in amounts necessary to ensure that each party bears its contractual share of the overall development and commercialization costs incurred. The Company’s policy is to account for cost-sharing payments to its collaboration partners as increases in expense in its consolidated statements of operations, while cost sharing payments by its collaboration partners to the Company are presented as reductions in expense. Cost-sharing payments related to development activities and commercialization activities are recorded in research and development expenses and selling, general and administrative expenses, respectively.
(m) Research and Development
Research and development expenses include personnel and facility-related expenses, outside contracted services including clinical trial costs, manufacturing and process development costs, research costs and other consulting services. Research and development costs are expensed as incurred. In instances where the Company enters into agreements with third parties to provide research and development services to it, costs are expensed as services are performed. Amounts due under such arrangements may be either fixed fee or fee for service, and may include upfront payments, monthly payments, and payments upon the completion of milestones or receipt of deliverables.
The Company’s cost accruals for clinical trials and other research and development activities are based on estimates of the services received and efforts expended pursuant to contracts with numerous clinical trial centers and contract research organizations. In the normal course of business the Company contracts with third parties to perform various research and development activities in the on-going development of its product candidates, including without limitation, third party clinical trial centers and contract research organizations that perform and administer the Company’s clinical trials on its behalf. The financial terms of these agreements are subject to negotiation and vary from contract to contract and may result in uneven payment flows. Payments under these agreements depend on factors such as the achievement of certain events, the successful enrollment of patients, and the completion of portions of the clinical trial or similar conditions. The objective of the Company’s accrual policy is to match the recording of expenses in its consolidated financial statements to the actual services received and efforts expended. As such, expense accruals related to clinical trials and other research and development activities are recognized based on the Company’s estimate of the degree of completion of the event or events specified in the specific agreement.
The Company’s estimates are dependent upon the time lines and accuracy of data provided by third parties regarding the status and cost of studies, and may not match the actual services performed by the organizations. This could result in adjustment to the Company’s research and development expense in future periods. To date, the Company has had no significant adjustments.
(n) Stock-Based Compensation
The Company has granted stock options, restricted stock units, performance share awards and stock appreciation rights pursuant to the terms of its Amended and Restated 2004 Equity Incentive Award Plan. The Company accounts for stock-based compensation awards granted to employees and directors in accordance with ASC 718, “Stock Compensation,” and has applied the provisions of the Securities and Exchange Commission, or SEC, Staff Accounting Bulletin, or SAB, No. 107 and No. 110, in its application of ASC 718. Stock compensation arrangements with consultants are accounted for in accordance with ASC 718 and ASC 505-50, “Equity-Based Payments to Non-Employees,” using a fair value approach. The compensation costs of these arrangements are subject to remeasurement over the vesting terms as earned.
71
Table of Contents
The Company determines stock-based compensation expense associated with stock options based on the estimated grant date fair value using the Black-Scholes valuation model, which requires the use of subjective assumptions related to expected stock price volatility, expected option term, expected dividend yield and risk-free interest rate. For employee and director stock options, the Company recognizes compensation expense over the vesting period of the awards that are ultimately expected to vest. Stock options granted to consultants are valued at their respective measurement dates and recognized as expense based on the portion of the total consulting services provided during the applicable period. As further consulting services are provided in each period, the Company revalues the associated awards and recognizes additional expense based on their then-current fair values.
Stock-based compensation expense associated with restricted stock units is based on the fair value of the Company’s common stock on the grant date, which equals the closing market price of the Company’s common stock on the grant date of the award. For restricted stock units, the company recognizes compensation expense over the vesting period of the awards that are ultimately expected to vest.
Performance share awards allow the recipients of such awards to earn fully vested shares of the Company’s common stock based on the achievement of pre-established performance objectives specified in the awards. Stock-based compensation expense associated with performance share awards is based on the estimated grant date fair value of the Company’s common stock using the Black Scholes valuation model and is recognized based upon the Company’s best estimates of the achievement of the performance objectives specified in such awards and the resulting number of shares that are expected to be earned. The Company evaluates on a quarterly basis the probability of achieving the performance criteria. The cumulative effect on current and prior periods of a change in the estimated number of performance share awards expected to be earned is recognized as compensation expense or as reduction of previously recognized compensation expense in the period of the revised estimate.
The fair value of stock-settled and cash-settled stock appreciation rights is initially measured on the grant date using the Black-Scholes valuation model, which requires the use of subjective assumptions related to the expected stock price volatility, expected term, expected dividend yield and risk-free interest rate. Similar to stock options, compensation expense for stock-settled stock appreciation rights is recognized over the vesting period of the awards that are ultimately expected to vest based on the grant-date fair value. Cash-settled stock appreciation rights are liability-classified awards for which compensation expense and the liability are remeasured at each reporting date through the date of settlement based on the portion of the requisite service period rendered.
The Company applies a forfeiture rate when determining stock-based compensation expense to account for an estimate of the granted awards not expected to vest. If actual forfeitures differ from the expected rate, the Company may be required to make additional adjustments to compensation expense in future periods.
(o) Promotional and Advertising Costs
Promotional and advertising costs are classified as selling, general and administrative expenses, and are expensed as incurred. Promotional and advertising expenses consist primarily of the costs of designing, producing and distributing materials promoting the Company or its product candidates, including its corporate website. Promotional and advertising expenses were insignificant in the years ended December 31, 2011, 2010 and 2009.
(p) Income Taxes
The Company accounts for income taxes using an asset and liability approach in ASC 740-10, “Accounting for Income Taxes,” which requires the recognition of taxes payable or refundable for the current year and deferred tax assets and liabilities for the future tax consequences of events that have been recognized in the consolidated financial statements or tax returns. The measurement of current and deferred tax assets and
72
Table of Contents
liabilities is based on provisions of the enacted tax law; the effects of future changes in tax laws or rates are not anticipated. The measurement of deferred tax assets is reduced, if necessary, by the amount of any tax benefits that, based on available evidence, is not expected to be realized.
The Company records a valuation allowance to reduce its deferred tax assets for the amount that it believes is more likely than not to be realized. Due to the Company’s lack of earnings history, the Company intends to maintain a full valuation allowance on the U.S. deferred tax assets until sufficient positive evidence exists to support reversal of the valuation allowance.
The Company establishes reserves for uncertain income tax positions based on the guidance in ASC 740-10-25, “Accounting for Uncertainty in Income Taxes.” When establishing reserves for uncertain income tax positions, the Company considers the technical support for the positions, past audit experience with similar situations and potential interest and penalties related to the matters. Recorded reserves represent the Company’s best estimate of the amount, if any, that it may ultimately be required to pay to settle such matters. The resolution of uncertain income tax positions is dependent on uncontrollable factors such as law changes, new case law and the willingness of income tax authorities to settle, including the timing thereof and other factors. The Company does not expect significant changes to its uncertain income tax positions in the next 12 months. However, items outside of the Company’s control could cause its uncertain income tax positions to change in the future, which would be recorded in the consolidated statements of operations. The Company’s practice is to recognize interest and/or penalties related to income tax matters in income tax expense as incurred.
(q) Basic and Diluted Net Loss per Common Share
Basic net loss per common share is computed by dividing the net loss by the weighted-average number of common shares outstanding during the period. Diluted net loss per common share is computed similarly to basic net loss per common share, except that the denominator is increased to include all potentially dilutive common shares, including outstanding options, warrants, restricted stock units, performance share awards and stock appreciation rights. Potentially dilutive common shares have been excluded from the diluted net loss per common share computations in all periods presented because such securities have an anti-dilutive effect on net loss per common share due to the Company’s net loss in each of the years presented.
Potentially dilutive securities were as follows:
| December 31, | ||||||||||||
| 2011 | 2010 | 2009 | ||||||||||
| Stock options |
4,301,808 | 5,041,303 | 5,603,960 | |||||||||
| Restricted stock units |
101,617 | 173,119 | 10,834 | |||||||||
| Warrants |
22,904 | 22,904 | 100,323 | |||||||||
| Stock appreciation rights |
262,800 | — | — | |||||||||
| Performance shares eligible to be earned |
62,500 | — | — | |||||||||
|
|
|
|
|
|
|
|||||||
| Total |
4,751,629 | 5,237,326 | 5,715,117 | |||||||||
|
|
|
|
|
|
|
|||||||
(r) Recently Issued Accounting Pronouncements Impacting the Company
In June 2011, the FASB issued Accounting Standards Update, or ASU, No. 2011-05, “Comprehensive Income (Topic 220): Presentation of Comprehensive Income,” with an objective of increasing the prominence of items reported in other comprehensive income, or OCI. This amendment provides companies with the option to present the total of comprehensive income, the components of net income and the components of OCI in either a single continuous statement of comprehensive income or in two separate but continuous statements. In addition, companies must present on the face of the financial statements, items reclassified from OCI to net income in the section of the financial statements where the components of net income and OCI are presented, regardless of the option selected to present comprehensive income. The amended guidance related to the presentation of
73
Table of Contents
comprehensive income is applicable retrospectively and is effective for fiscal years and interim periods within those fiscal years, beginning after December 15, 2011 and will be effective for the Company in the first quarter of fiscal 2012. In December 2011, the FASB issued ASU No. 2011-12, which defers changes in ASU No. 2011-05 that relate to the presentation of reclassification adjustments out of accumulated OCI to enable the FASB time to re-deliberate the presentation requirements. The adoption of this amendment concerns disclosure only and the Company does not expect it to have an impact on its consolidated financial position, results of operations or cash flows.
In May 2011, the FASB issued ASU No. 2011-04, “Fair Value Measurement (Topic 820): Amendments to Achieve Common Fair Value Measurement and Disclosure Requirements in U.S. GAAP and IFRSs.” This amendment is intended to result in convergence between U.S. GAAP and International Financial Reporting Standards requirements for measurement of and disclosures about fair value. This guidance clarifies the application of existing fair value measurements and disclosures, and changes certain principles or requirements for fair value measurements and disclosures. The amendment will be effective for the Company in the second quarter of fiscal year 2012. The Company is currently assessing the potential impact, if any, this amendment may have on its consolidated financial position, results of operations and cash flows.
In March 2010, the FASB ratified Emerging Issues Task Force, or EITF, Issue No. 08-9, “Milestone Method of Revenue Recognition” (Issue 08-9). The ASU, resulting from Issue 08-9 amends ASC 605-28. The Task Force concluded that the milestone method is a valid application of the proportional performance model when applied to research or development arrangements. Accordingly, the consensus states that an entity can make an accounting policy election to recognize a payment that is contingent upon the achievement of a substantive milestone in its entirety in the period in which the milestone is achieved. A milestone is defined in the consensus as an event: (1) that can only be achieved based in whole or in part on either (a) the entity’s performance or (b) on the occurrence of a specific outcome resulting from the entity’s performance; (2) for which there is substantive uncertainty at the date the arrangement is entered into that the event will be achieved; and (3) that would result in additional payments being due to the entity. Issue 08-9 is effective for fiscal years, and interim periods within those years, beginning on or after June 15, 2010, and may be applied either prospectively to milestones achieved after the adoption date, or; retrospectively for all periods presented. The Company adopted the new guidance effective January 1, 2011, analyzed its impact on its consolidated financial statements, and concluded there was no such impact.
In September 2009, the FASB amended ASC 605 as summarized in ASU 2009-13, “Revenue Recognition: Multiple-Deliverable Revenue Arrangements.” Guidance in ASC 605-25 on revenue arrangements with multiple deliverables has been amended to require an entity to allocate revenue to deliverables in an arrangement using its best estimate of selling prices if the vendor does not have vendor-specific objective evidence or third-party evidence of selling prices, and to eliminate the use of the residual method and require the entity to allocate revenue using the relative selling price method. The new guidance also requires expanded quantitative and qualitative disclosures about revenue from arrangements with multiple deliverables. The update is effective for fiscal years beginning on or after June 15, 2010, with early adoption permitted. Adoption may either be on a prospective basis for new revenue arrangements entered into or existing arrangements modified, after adoption of the update, or by retrospective application. The Company adopted this guidance on a prospective basis effective January 1, 2011, analyzed its impact on its consolidated financial statements, and concluded there was no such impact.
3. COLLABORATION AGREEMENTS
(a) Collaboration Agreement with Astellas
The Company entered into a collaboration agreement with Astellas in October 2009. Under the Astellas Collaboration Agreement, the Company and Astellas agreed to collaborate on the development of MDV3100 for the United States market, including associated regulatory filings with the U.S. Food and Drug Administration, or
74
Table of Contents
the FDA. Based on the positive results from the AFFIRM trial, the Company elected to exercise its right under the Astellas Collaboration Agreement to co-promote MDV3100 in the U.S. market. Should MDV3100 receive marketing approval by the FDA, following such approval and the launch of MDV3100 in the United States, the Company and Astellas will co-promote MDV3100 in the United States and each will provide 50% of the sales and medical affairs field forces supporting MDV3100 in the U.S. market. Astellas is responsible for development of, seeking regulatory approval for and commercialization of MDV3100 outside the United States. Astellas will be responsible for commercial manufacture of MDV3100 on a global basis. Both Medivation and Astellas have agreed not to commercialize certain other products having a similar mechanism of action as MDV3100 for the treatment of specified indications for a specified time period, subject to certain exceptions.
The Company and Astellas share equally the costs and expenses of developing and commercializing MDV3100 for the United States market, except that (a) development costs for studies useful in both the United States market and either Europe or Japan are shared two-thirds by Astellas and one-third by the Company, and (b) both the Company and Astellas will be responsible for all commercialization costs incurred in fielding and supporting its respective MDV3100 sales forces, and each of Astellas and the Company will be entitled to receive a fee for each detail made by its respective sales forces. The Company and Astellas will share profits (or losses) resulting from the commercialization of MDV3100 in the United States equally. Outside the United States, Astellas will bear all development and commercialization costs and will pay the Company tiered, double-digit royalties on the aggregate net sales of MDV3100.
The agreement establishes several joint committees consisting of an equal number of representatives from both parties that operate by consensus to oversee the collaboration. In the event that a joint committee is unable to reach consensus on a particular issue, then, depending on the issue, a dispute may be decided at the joint committee level by the party with the final decision on the issue or escalated to senior management of the parties. If a dispute is escalated to senior management and no consensus is reached, then the dispute may be decided by the party to whom the contract grants final decision on such issue. Other issues can only be decided by consensus of the parties, and unless and until the parties’ representatives reach agreement on such issue, no decision on such issue will be made, and the status quo will be maintained.
Under the Astellas Collaboration Agreement, Astellas paid the Company a non-refundable, up-front cash payment of $110.0 million in the fourth quarter of 2009. The Company is also eligible to receive up to $335.0 million in development milestone payments, plus up to an additional $320.0 million in commercial milestone payments. As of December 31, 2011, the Company had received an aggregate of $13.0 million in development milestone payments under the Astellas Collaboration Agreement. Should the FDA or European Medicines Agency accept for filing a new drug application or marketing authorization application, respectively, seeking approval of MDV3100 in post-chemotherapy advanced prostate cancer patients based on the positive results from the AFFIRM trial, the Company would be entitled to a $10.0 million and a $5.0 million milestone payment, respectively, under the Astellas Collaboration Agreement. In addition, should the new drug application be approved by the FDA or the marketing authorization application be approved by the European regulators, the Company would be entitled to a $30.0 million and a $15.0 million milestone payment, respectively, under the Astellas Collaboration Agreement. The Company is required to share 10% of the up-front and development milestone payments received under the Astellas Collaboration Agreement with The Regents of the University of California, or UCLA, pursuant to the terms of its MDV3100 license agreement. The Company and Astellas each are permitted to terminate the Astellas Collaboration Agreement for an uncured material breach by, or the insolvency of, the other party. Astellas has a right to terminate the Astellas Collaboration Agreement unilaterally by advance written notice to the Company, but except in certain specific circumstances, generally cannot exercise that termination right until the first anniversary of MDV3100’s first commercial sale. Following any termination of the Astellas Collaboration Agreement in its entirety, all rights to develop and commercialize MDV3100 will revert to the Company, and Astellas will grant a license to the Company to enable the Company to continue such development and commercialization. In addition, except in the case of a termination by Astellas for an uncured material breach, Astellas will supply MDV3100 to the Company during a specified transition period.
75
Table of Contents
(b) Former Collaboration Agreement with Pfizer
The Company entered into a collaboration agreement with Pfizer in October 2008. Under the agreement, the Company and Pfizer agreed to collaborate on the development of dimebon for Alzheimer’s disease and Huntington disease for the United States market, including associated regulatory filings with the FDA. Pfizer paid the Company a non-refundable up-front cash payment of $225.0 million in the fourth quarter of 2008 pursuant to the terms of the agreement. Under the terms of the former collaboration agreement with Pfizer, the Company and Pfizer shared the costs and expenses of developing and commercializing dimebon for the United States market on a 60%/40% basis, with Pfizer assuming the larger share.
Based on the negative results in the Phase 3 CONNECTION, HORIZON and CONCERT trials, the Company and Pfizer have discontinued development of dimebon for all indications in 2012. In January 2012, Pfizer exercised its right to terminate the collaboration agreement as discussed in Note 14, “Subsequent Events.”
(c) Deferred Revenue and Collaboration Revenue
Through December 31, 2011, the Company has received an aggregate of $123.0 million of up-front and development milestone payments under the Astellas Collaboration Agreement and $225.0 million of up-front payments under its former collaboration agreement with Pfizer.
Deferred revenue related to the Astellas Collaboration Agreement and the former collaboration agreement with Pfizer consisted of the following:
| December 31, | ||||||||
| 2011 | 2010 | |||||||
| Current portion: |
||||||||
| Deferred revenue from Astellas |
$ | 23,747 | $ | 23,138 | ||||
| Deferred revenue from Pfizer |
36,015 | 36,015 | ||||||
|
|
|
|
|
|||||
| Total |
$ | 59,762 | $ | 59,153 | ||||
|
|
|
|
|
|||||
| Long-term portion: |
||||||||
| Deferred revenue from Astellas |
$ | 47,494 | $ | 69,477 | ||||
| Deferred revenue from Pfizer |
36,015 | 72,030 | ||||||
|
|
|
|
|
|||||
| Total |
$ | 83,509 | $ | 141,507 | ||||
|
|
|
|
|
|||||
Collaboration revenue recognized with respect to the Astellas Collaboration Agreement and the former collaboration agreement with Pfizer consisted of the following:
| Years Ended December 31, | ||||||||||||
| 2011 | 2010 | 2009 | ||||||||||
| Collaboration revenue from Astellas |
$ | 24,374 | $ | 23,492 | $ | 3,893 | ||||||
| Collaboration revenue from Pfizer |
36,015 | 39,016 | 65,361 | |||||||||
|
|
|
|
|
|
|
|||||||
| Total |
$ | 60,389 | $ | 62,508 | $ | 69,254 | ||||||
|
|
|
|
|
|
|
|||||||
76
Table of Contents
(d) Cost-Sharing Payments
The Company recorded development cost-sharing payments from Astellas and Pfizer, and corresponding reductions in research and development expense as follows:
| Years Ended December 31, | ||||||||||||
| 2011 | 2010 | 2009 | ||||||||||
| Development cost-sharing payments from Astellas |
$ | 44,285 | $ | 34,125 | $ | 2,784 | ||||||
| Development cost-sharing payments from Pfizer |
12,365 | 29,139 | 20,435 | |||||||||
|
|
|
|
|
|
|
|||||||
| Total |
$ | 56,650 | $ | 63,264 | $ | 23,219 | ||||||
|
|
|
|
|
|
|
|||||||
The Company recorded commercialization cost-sharing payments (to) from Astellas and Pfizer, and corresponding (increases) reductions in selling, general and administrative expenses as follows:
| Years Ended December 31, | ||||||||||||
| 2011 | 2010 | 2009 | ||||||||||
| Commercialization cost-sharing payments (to) from Astellas |
$ | (472 | ) | $ | 520 | $ | 74 | |||||
| Commercialization cost-sharing payments from (to) Pfizer |
32 | (1,084 | ) | (720 | ) | |||||||
|
|
|
|
|
|
|
|||||||
| Total |
$ | (440 | ) | $ | (564 | ) | $ | (646 | ) | |||
|
|
|
|
|
|
|
|||||||
At December 31, 2011 and 2010, development and commercialization cost-sharing payments receivable from Astellas were $10.6 million and $11.6 million, respectively, and at December 31, 2011 and 2010, development and commercialization cost-sharing payments receivable from Pfizer were $2.0 million and $9.6 million, respectively. The amounts receivable at December 31, 2011 from Astellas and Pfizer were received in the first quarter of 2012.
4. SHORT-TERM INVESTMENTS
At December 31, 2011, the amortized cost, gross unrealized gain and estimated fair value of available-for-sale securities, consisting of United States treasury bills maturing in January 2012 and May 2012, were $75.0 million, $0.0 million and $75.0 million, respectively.
At December 31, 2010, the amortized cost, gross unrealized gain and estimated fair value of available-for-sale securities, consisting of a United States treasury note maturing in April 2011 was $100.0 million, $0.0 million and $100.0 million, respectively.
5. PROPERTY AND EQUIPMENT, NET
The components of the Company’s property and equipment, net, consisted of the following:
| December 31, | ||||||||
| 2011 | 2010 | |||||||
| Furniture and fixtures |
$ | 229 | $ | 221 | ||||
| Leasehold improvements |
630 | 630 | ||||||
| Computer equipment and software |
747 | 632 | ||||||
| Laboratory equipment |
371 | 349 | ||||||
| Construction in progress |
164 | 22 | ||||||
|
|
|
|
|
|||||
| 2,141 | 1,854 | |||||||
| Less: accumulated depreciation and amortization |
(1,421 | ) | (992 | ) | ||||
|
|
|
|
|
|||||
| Total |
$ | 720 | $ | 862 | ||||
|
|
|
|
|
|||||
77
Table of Contents
6. ACCRUED EXPENSES
Accrued expenses consisted of the following:
| December 31, | ||||||||
| 2011 | 2010 | |||||||
| Payroll and payroll related |
$ | 1,112 | $ | 617 | ||||
| Preclinical and clinical trials |
21,453 | 19,190 | ||||||
| Other |
1,449 | 1,592 | ||||||
|
|
|
|
|
|||||
| Total |
$ | 24,014 | $ | 21,399 | ||||
|
|
|
|
|
|||||
7. STOCKHOLDERS’ EQUITY
(a) Stock Purchase Rights
All shares of the Company’s common stock, if issued prior to the termination by the Company of its rights agreement, dated as of December 4, 2006, include stock purchase rights. The rights are exercisable only if a person or group acquires twenty percent or more of the Company’s common stock or announces a tender or exchange offer which would result in ownership of twenty percent or more of the Company’s common stock. Following the acquisition of twenty percent or more of the Company’s common stock, the holders of the rights, other than the acquiring person or group, may purchase Medivation common stock at half of its fair market value. In the event of a merger or other acquisition of the Company, the holders of the rights, other than the acquiring person or group, may purchase shares of the acquiring entity at half of their fair market value. The rights were not exercisable at December 31, 2011.
(b) Medivation Equity Incentive Plan
The Medivation Amended and Restated 2004 Equity Incentive Award Plan, or the Medivation Equity Incentive Plan, which is stockholder-approved, provides for the issuance of options and other stock-based awards, including restricted stock units, performance share awards and stock appreciation rights, covering up to 7,500,000 shares of Medivation’s common stock. Shares issued upon exercise of stock-based awards are new shares that have been reserved for issuance under the Medivation Equity Incentive Plan. The amendment and restatement of the Medivation Equity Incentive Plan was approved by the Company’s Board of Directors, or Board, and by the Company’s stockholders in March and May 2007, respectively.
The Medivation Equity Incentive Plan is administered by the Board, or a committee appointed by the Board, which determines recipients and types of awards to be granted, including the number of shares subject to the awards, the exercise price and the vesting schedule. The vesting of all outstanding awards under the Medivation Equity Incentive Plan, including all outstanding options, restricted stock units, performance share awards and stock appreciation rights will accelerate and become immediately exercisable upon a “change of control” of Medivation, as defined in the Medivation Equity Incentive Plan.
Stock options
The Company has granted stock options to employees, directors and consultants pursuant to the terms of the Medivation Equity Incentive Plan. The terms of stock options granted under the Medivation Equity Incentive Plan cannot exceed ten years. Options generally have an exercise price equal to the fair market value of the Company’s common stock on the grant date, and generally vest over a period of four years.
The Company estimates the fair value of each option granted to employees and directors on the grant date using the Black-Scholes valuation model, which requires the use of subjective assumptions related to the expected stock price volatility, expected stock option term, expected dividend yield and expected risk-free rates
78
Table of Contents
of return. The following table presents the assumptions used to estimate the fair value of employee and director stock options granted during the years ended December 31, 2011, 2010 and 2009. No significant option grants were made to consultants during the periods presented.
| Years Ended December 31, | ||||||||||||
| 2011 | 2010 | 2009 | ||||||||||
| Risk-free interest rate |
1.02-2.34% | 1.48-2.39% | 1.71-2.87% | |||||||||
| Estimated term (in years) |
5.6-6.0 | 6.0 | 6.0 | |||||||||
| Estimated volatility |
71-86% | 71-72% | 72–88% | |||||||||
| Estimated dividend yield |
None | None | None | |||||||||
| Weighted-average grant-date fair value per share of options granted |
$ | 13.75 | $ | 9.64 | $ | 23.04 | ||||||
Beginning with the first quarter of 2011, the Company estimates volatility based on the historical price volatility of its common stock and implied volatility of its common stock inherent in the market price of publicly traded options in its common stock. For periods prior to the first quarter of 2011, the Company also considered the historical price volatility of comparable companies’ common stock.
Prior to the first quarter of 2010, due to its limited history of stock option exercise behavior, the Company used the simplified method of estimating stock option term provided for in SAB No. 107 and No. 110 for options granted to employees and directors. Beginning in the first quarter of 2010, the Company changed to a method based on its actual exercise experience and an assumption that unexercised options will remain outstanding for a period equal to the midpoint between the date the option vests in full and the contractual option termination date.
The Company has no history or expectation of paying cash dividends on its common stock. The risk-free interest rate is based on the U.S. Treasury yield for a term consistent with the expected term of the awards at the time of grant.
The use of different estimates of expected stock price volatility, expected stock option term, expected dividend yield and risk-free interest rates could materially change the fair value of an option and the resulting non-cash compensation expense.
The following table summarizes stock option activity during the year ended December 31, 2011:
| Number of Shares |
Weighted- Average Exercise Price |
Weighted- Average Remaining Contractual Term (in years) |
Aggregate Intrinsic Value (1) |
|||||||||||||
| Options outstanding at December 31, 2010 |
5,041,303 | $ | 17.39 | |||||||||||||
| Granted |
595,364 | $ | 19.71 | |||||||||||||
| Exercised |
(1,106,403 | ) | $ | 14.22 | ||||||||||||
| Forfeited |
(228,456 | ) | $ | 20.79 | ||||||||||||
|
|
|
|||||||||||||||
| Options outstanding at December 31, 2011 |
4,301,808 | $ | 18.34 | 6.9 | $ | 119.4 | ||||||||||
|
|
|
|||||||||||||||
| Vested and exercisable as of December 31, 2011 |
2,739,718 | $ | 16.72 | 5.9 | $ | 80.5 | ||||||||||
|
|
|
|||||||||||||||
| (1) | The aggregate intrinsic value is calculated as the pre-tax difference between the exercise price of the underlying awards and the closing stock price per share of $46.11 of the Company’s common stock on December 30, 2011. The amount is presented in millions. |
79
Table of Contents
Additional information regarding stock options is set forth below:
| Years Ended December 31, | ||||||||||||
| 2011 | 2010 | 2009 | ||||||||||
| Intrinsic value of options exercised |
$ | 22,238 | $ | 8,584 | $ | 6,208 | ||||||
| Grant-date fair value of options vested |
$ | 12,608 | $ | 13,730 | $ | 11,653 | ||||||
Restricted Stock Units
The Company has granted restricted stock units to employees pursuant to the terms of the Medivation Equity Incentive Plan. A restricted stock unit award is an agreement to issue shares of the Company’s common stock at the time of vesting. All restricted stock units granted to date by the Company vest in three equal installments on the first, second and third anniversaries of the grant date. The fair value of restricted stock units equals the closing market price of the Company’s common stock on the grant date.
The following table summarizes restricted stock unit activity:
| Number of Shares |
Weighted- Average Grant-Date Fair Value |
|||||||
| Unvested at December 31, 2010 |
173,119 | $ | 14.23 | |||||
| Vested |
(51,606 | ) | $ | 14.37 | ||||
| Forfeited |
(19,896 | ) | $ | 13.29 | ||||
|
|
|
|||||||
| Unvested at December 31, 2011 |
101,617 | $ | 14.35 | |||||
|
|
|
|||||||
The total fair value of restricted stock units that vested during the years ended December 31, 2011, 2010 and 2009 were $2.4 million, $0.2 million and $0.4 million, respectively.
Performance Share Awards
The Company granted performance share awards in 2011 to certain employees pursuant to the terms of the Medivation Equity Incentive Plan. The terms of the performance share awards provide for base case and upside case numbers of shares eligible to be earned based on the level of achievement of specified performance objectives relating to commercial product sales and timelines.
The performance shares under the awards will be earned, if at all, upon determination by the Compensation Committee of the Board of Directors, or the Committee, of actual achievement of performance objectives, subject to specified change of control exceptions. Each recipient of a performance share award must remain an employee of the Company through the date the Committee determines actual performance has been achieved in order to earn the performance shares eligible under the award. Each performance share award is convertible into one share of the Company’s common stock upon the achievement of the performance objective. The base and upside case numbers of shares eligible to be earned under the performance share awards are set forth below. The number of performance shares eligible to be earned at the upside case level is based on achievement of the applicable performance objectives by specified dates, and the number of performance shares eligible to be earned at the base case level is based on the achievement of the performance objectives during the ten year term of the performance share awards. The following summarizes the performance share awards outstanding at December 31, 2011:
| Base Case Achievement | Upside Case Achievement | |||||||||||||||
| Minimum | Maximum | Minimum | Maximum | |||||||||||||
| Performance shares eligible to be earned |
10,419 | 31,254 | 20,834 | 62,500 | ||||||||||||
80
Table of Contents
Stock-based compensation expense associated with performance share awards is based on the estimated grant date fair value of the Company’s common stock using the Black Scholes valuation model, and is recognized based on the probable outcome of the performance conditions, which are evaluated quarterly.
Stock Appreciation Rights
The Company granted stock appreciation rights in 2011 to certain employees pursuant to the terms of the Medivation Equity Incentive Plan. Stock appreciation rights give the holder the right, upon exercise, to receive the difference between the price per share of the Company’s common stock at the time of exercise and the exercise price of the stock appreciation right. The exercise price of the stock appreciation rights is equal to the market price of the Company’s common stock at the date of grant. One-fourth of the shares subject to the stock appreciation rights vest and become exercisable on the first anniversary of the grant date, and the remaining three-fourths of the shares vest monthly over the three years thereafter. The term of the stock appreciation rights is ten years.
Until such time, if any, that the Company’s stockholders approve an increase to the number of shares of common stock available pursuant to the Medivation Equity Incentive Plan, the stock appreciation rights may be settled only in cash; following such approval, if it occurs, the stock appreciation rights may be settled only in the Company’s common stock.
The Company estimates the fair value of each stock appreciation right on the grant date using the Black-Scholes valuation model. The following grant date assumptions were used to estimate the fair value of stock appreciation rights granted during the year ended December 31, 2011:
| Risk-free interest rate |
1.07 | % | ||
| Estimated term (in years) |
5.6 | |||
| Estimated volatility |
71 | % | ||
| Estimated dividend yield |
None | |||
| Weighted-average grant-date fair value per share of stock appreciation rights granted |
$ | 29.81 |
The stock appreciation rights are currently liability-classified awards for which compensation expense and the liability are remeasured at each reporting date through the date of settlement based on the portion of the requisite service period rendered. At December 31, 2011, 262,800 stock appreciation rights were outstanding, none of which were exercisable.
Warrants
At December 31, 2011, warrants to purchase an aggregate of 22,904 shares of Medivation common stock at a weighted-average exercise price of $6.92 per share were outstanding. These outstanding warrants expire between 2014 and 2017.
Stock-Based Compensation
Stock-based compensation expense was as follows:
| Years Ended December 31, | ||||||||||||
| 2011 | 2010 | 2009 | ||||||||||
| Stock-based compensation expense recognized as: |
||||||||||||
| Research and development expense |
$ | 5,795 | $ | 7,629 | $ | 5,664 | ||||||
| Selling, general and administrative expense |
8,090 | 5,901 | 5,062 | |||||||||
|
|
|
|
|
|
|
|||||||
| Total |
$ | 13,885 | $ | 13,530 | $ | 10,726 | ||||||
|
|
|
|
|
|
|
|||||||
81
Table of Contents
The Company applies a forfeiture rate when determining non-cash stock-based compensation expense to account for an estimate of the granted awards not expected to vest. If actual forfeitures differ from the expected rate, the Company may be required to make additional adjustments to compensation expense in future periods. At December 31, 2011, the unrecognized stock-based compensation expense related to awards granted under the Medivation Equity Incentive Plan totaled $27.7 million, which is expected to be recognized as non-cash compensation expense over a weighted-average period of 2.68 years.
8. RETIREMENT PLAN
The Company has a defined contribution savings plan, which qualifies as a deferred salary arrangement under Section 401(k) of the Internal Revenue Code, or IRC. The 401(k) Plan is for the benefit of all employees and permits voluntary contributions by employees up to 100% of their annual pretax compensation limited by the Internal Revenue Service, or the IRS, imposed maximum contribution. The Company matches 100% of the first 3% of employee contributions and 50% of the next 2% of employee contributions. The Company’s contributions and the employee contributions are fully vested when contributed. The plan assets are held in trust and invested as directed by the plan participants. Employer matching contributions to the plan were $0.6 million, $0.5 million and $0.4 million for the years ended December 31, 2011, 2010 and 2009, respectively.
9. INCOME TAXES
The Company’s pre-tax loss for financial statement purposes was $43.2 million, $32.5 million and $46.5 million for the years ended December 31, 2011, 2010 and 2009, respectively. The income tax (benefit) expense consisted of the following:
| Years Ended December 31, | ||||||||||||
| 2011 | 2010 | 2009 | ||||||||||
| Current: |
||||||||||||
| Federal |
$ | (4,309 | ) | $ | 5,012 | $ | 441 | |||||
| State |
(22 | ) | (3,440 | ) | 7,831 | |||||||
|
|
|
|
|
|
|
|||||||
| Total current |
(4,331 | ) | 1,572 | 8,272 | ||||||||
| Deferred: |
||||||||||||
| Federal |
— | — | — | |||||||||
| State |
— | — | — | |||||||||
|
|
|
|
|
|
|
|||||||
| Total deferred |
— | — | — | |||||||||
|
|
|
|
|
|
|
|||||||
| Total income tax (benefit) expense |
$ | (4,331 | ) | $ | 1,572 | $ | 8,272 | |||||
|
|
|
|
|
|
|
|||||||
A reconciliation of the statutory Federal income tax rate of 35% to the Company’s effective income tax rates is as follows:
| Years Ended December 31, | ||||||||||||
| 2011 | 2010 | 2009 | ||||||||||
| Federal tax provision at statutory rate |
35.00 | % | 35.00 | % | 35.00 | % | ||||||
| State taxes (net of Federal benefit) |
2.27 | % | (4.17 | %) | 6.91 | % | ||||||
| Change in reserve |
(0.94 | %) | (1.84 | %) | — | |||||||
| Orphan drug credit |
(0.70 | %) | (1.06 | %) | — | |||||||
| Stock-based compensation |
0.06 | % | (2.03 | %) | (1.26 | %) | ||||||
| Change in valuation allowance |
(29.94 | %) | (39.65 | %) | (66.60 | %) | ||||||
| Research and development credits |
7.05 | % | 9.72 | % | 6.54 | % | ||||||
| Net proceeds from stockholder securities law settlement |
(2.43 | %) | — | — | ||||||||
| Other |
(0.33 | %) | (0.81 | %) | 1.52 | % | ||||||
|
|
|
|
|
|
|
|||||||
| Benefit (provision) for taxes |
10.04 | % | (4.84 | %) | (17.89 | %) | ||||||
|
|
|
|
|
|
|
|||||||
82
Table of Contents
Deferred income taxes reflect the net tax effects of temporary differences between the carrying amounts of assets and liabilities for financial reporting purposes and the tax basis of assets and liabilities. Significant components of the Company’s deferred tax assets for Federal and state income taxes are follows:
| December 31, | ||||||||
| 2011 | 2010 | |||||||
| Deferred tax assets: |
||||||||
| Deferred revenue |
$ | 54,116 | $ | 76,638 | ||||
| Net operating loss carry forward |
30,767 | 4,786 | ||||||
| Stock-based compensation |
9,872 | 8,243 | ||||||
| Research and development credits |
11,280 | 3,280 | ||||||
| Depreciation, amortization and other |
133 | 120 | ||||||
| Accruals and reserves |
552 | 729 | ||||||
|
|
|
|
|
|||||
| Total deferred tax assets |
106,720 | 93,796 | ||||||
| Less: valuation allowance |
(106,720 | ) | (93,796 | ) | ||||
|
|
|
|
|
|||||
| Net deferred tax assets |
$ | — | $ | — | ||||
|
|
|
|
|
|||||
The income tax benefit for 2011 was approximately $4.3 million, which represents an effective tax rate of 10.04%. The income tax benefit mainly consists of a benefit due to the ability to carry back the current year Federal net operating loss to the prior two tax years. The difference in the effective tax rate for 2011 as compared to 2010 is primarily attributable to the benefit in 2011 due to the ability to carry back the current year Federal net operating loss to the prior two tax years.
The income tax provision for 2010 was approximately $1.6 million, which mainly consists of the Federal and state income tax and represents an effective tax rate of (4.84)%. The decrease in the effective tax rate in 2010 as compared to 2009 was primarily attributable to the state tax benefit recognized in 2010 from the 2009 California income tax refund.
During the year ended December 31, 2010, the Company accelerated the recognition of revenue related to the Astellas non-refundable, up-front payment received in 2009 for income tax purposes, while such revenue was deferred for financial statement purposes. The Company also accelerated the recognition of the milestone payment of $10.0 million received in October 2010 from Astellas for income tax purposes, while such revenue was deferred for financial statement purposes. Due to the suspension of California Net Operating Loss utilization in 2010, the Company was not able to utilize the net operating loss carryforwards to offset the taxable income in 2010.
Due to the Company’s lack of earnings history, the net deferred tax assets have been fully offset by a valuation allowance. The valuation allowance increased by $12.9 million, $12.9 million and $30.8 million during the years ended December 31, 2011, 2010 and 2009, respectively.
The following table summarizes activity related to the Company’s gross unrecognized tax positions:
| December 31, | ||||||||||||
| 2011 | 2010 | 2009 | ||||||||||
| Balance as of beginning of year |
$ | 4,128 | $ | 889 | $ | 358 | ||||||
| Additions based on tax positions related to the current year |
500 | 393 | 429 | |||||||||
| Additions based on tax position related to prior year |
109 | 2,846 | 102 | |||||||||
| Decreases based on tax positions related to prior year |
(801 | ) | — | — | ||||||||
|
|
|
|
|
|
|
|||||||
| Balance as of end of year |
$ | 3,936 | $ | 4,128 | $ | 889 | ||||||
|
|
|
|
|
|
|
|||||||
83
Table of Contents
Approximately $0.6 million and $1.2 million of the total gross unrecognized tax benefits at December 31, 2011 and 2010, respectively, if recognized, would affect the effective tax rate. The Company does not anticipate a material change in unrecognized tax benefits during the next 12 months.
As a result of the Company’s net operating loss carryforwards, all of its tax years are subject to Federal and state examination. At December 31, 2011, there are no Federal or state tax audits in process. The Company was under audit by the Internal Revenue Service, or IRS, for the tax year of 2008. The audit was closed in 2011 with no adjustment.
Federal and state tax laws impose substantial restrictions on the utilization of net operating loss and credit carryforwards in the event of an “ownership change” for tax purposes, as defined in Internal Revenue Code Section 382. The Company completed Section 382 studies through December 31, 2010, and concluded that ownership changes occurred in 2004, 2007 and 2010. The ownership changes did not result in a reduction of its net operating loss or in its research and development credits expiring unused. If additional ownership change occurs, the utilization of net operating loss and credit carryforwards could be significantly reduced.
As of December 31, 2011, the Company has Federal net operating loss carryforwards of approximately $77.3 million, which will expire in 2031, if not utilized. Also, as of December 31, 2011, the Company has state net operating loss carryforwards of approximately $154.1 million, which will expire at various dates between the years 2015 and 2031, if not utilized.
The Company has Federal research and development credit and Orphan Drug credit carryforwards of approximately $13.3 million as of December 31, 2011. The Federal tax credit carryforwards expire in the year 2024 through 2031, if not used. In addition, the Company has California research and development credit carryforwards of approximately $1.4 million as of December 31, 2011. The California research credits do not expire.
10. FAIR VALUE MEASUREMENTS
The Company follows ASC 820-10, “Fair Value Measurements and Disclosures,” which among other things, defines fair value, establishes a consistent framework for measuring fair value and expands disclosure for each major asset and liability category measured at fair value on either a recurring or nonrecurring basis. Fair value is an exit price, representing the amount that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market participants. As such, fair value is a market-based measurement that should be determined based on assumptions that market participants would use in pricing an asset or liability. As a basis for considering such assumptions, a three-tier fair value hierarchy has been established, which prioritizes the inputs used in measuring fair value as follows:
| • | Level 1—Inputs are unadjusted, quoted prices in active markets for identical assets or liabilities at the measurement date. |
| • | Level 2—Inputs (other than quoted prices included in Level 1) are either directly or indirectly observable for the asset or liability through correlation with market data at the measurement date and for the duration of the instrument’s anticipated life. |
| • | Level 3—Inputs reflect management’s best estimate of what market participants would use in pricing the asset or liability at the measurement date. Consideration is given to the risk inherent in the valuation technique and the risk inherent in the inputs to the model. |
84
Table of Contents
At December 31, 2011 and 2010, the Company did not have any assets or liabilities classified as Level 2 or Level 3. Assets classified as Level 1 are included in the table below:
| Fair value measurements using: | ||||||||||||||||
| Fair Value | Level 1 | Level 2 | Level 3 | |||||||||||||
| December 31, 2011: |
||||||||||||||||
| Cash and cash equivalents: |
||||||||||||||||
| Money market funds |
$ | 44,044 | $ | 44,044 | — | — | ||||||||||
| Short-term investments: |
||||||||||||||||
| U.S. Treasury bills |
$ | 74,996 | $ | 74,996 | — | — | ||||||||||
| December 31, 2010: |
||||||||||||||||
| Cash and cash equivalents: |
||||||||||||||||
| Money market funds |
$ | 53,657 | $ | 53,657 | — | — | ||||||||||
| Short-term investments: |
||||||||||||||||
| U.S. Treasury note |
$ | 99,964 | $ | 99,964 | — | — | ||||||||||
11. COMMITMENTS AND CONTINGENCIES
(a) Operating Leases
The majority of the Company’s operating lease payments relate to its corporate headquarters. The Company’s corporate headquarters are located at 201 Spear Street, San Francisco, California, where it leases approximately 34,000 square feet of office space pursuant to lease agreements that expire in July 2012 and May 2013.
In December 2011, the Company entered into a lease agreement for approximately 57,000 square feet of office space located at 525 Market Street, San Francisco, California, which is intended to serve as its future corporate headquarters following construction and build-out. The term of the lease is for seven years, with an anticipated commencement date of June 2012. The Company has an option to extend the term of the lease for an additional five years.
The future minimum rentals under the Company’s non-cancelable operating leases having initial or remaining terms in excess of one year at December 31, 2011 were as follows:
| 2012 |
$ | 3,599 | ||
| 2013 |
4,814 | |||
| 2014 |
4,563 | |||
| 2015 |
4,620 | |||
| 2016 and after |
11,206 | |||
|
|
|
|||
| Total |
$ | 28,802 | ||
|
|
|
Rent expense, net of sublease income, for the years ended December 31, 2011, 2010 and 2009 was $2.8 million, $2.2 million and $2.8 million, respectively. Sublease income was not significant for the periods presented. In addition to the future minimum rental payments included in the table above, certain lease agreements also require the Company to make additional payments during the lease term for taxes, insurance, and other operating expenses.
In March 2010, the Company terminated a lease for approximately 64,000 square feet of office space located at 345 Spear Street, San Francisco, California, that it entered into in November 2009, which was intended to serve as the Company’s corporate headquarters. The lease, which was terminated as a result of the negative CONNECTION trial results, resulted in a termination fee of $1.5 million, of which half was recorded as expense in the fourth quarter of 2009 and the remaining half was recorded as expense in the first quarter of 2010.
85
Table of Contents
(b) Restricted Cash
The Company has outstanding letters of credit collateralized by restricted cash totaling $6.0 million and $0.8 million at December 31, 2011 and 2010, respectively, to secure various operating leases. At December 31, 2011, $0.5 million and $5.5 million of restricted cash associated with these letters of credit were classified as current and long-term assets, respectively, on the consolidated balance sheets, and at December 31, 2010, $0.8 million of restricted cash was classified as long-term assets.
(c) Litigation
In March 2010, the first of several putative securities class action lawsuits was commenced in the U.S. District Court for the Northern District of California, naming as defendants the Company and certain of its officers. The lawsuits are largely identical and allege violations of the Securities Exchange Act of 1934, as amended. The plaintiffs allege, among other things, that the defendants disseminated false and misleading statements about the effectiveness of dimebon for the treatment of Alzheimer’s disease. The plaintiffs purport to seek damages, an award of their costs and injunctive relief on behalf of a class of stockholders who purchased or otherwise acquired common stock in the Company between September 21, 2006 and March 2, 2010. The actions were consolidated in September 2010 and in April 2011 the court entered an order appointing Catoosa Fund L.P. and its attorneys as lead plaintiff and lead counsel. Thereafter, the lead plaintiff filed a consolidated, amended complaint, which was dismissed without prejudice as to all defendants in August 2011. The lead plaintiff filed a second amended complaint in November 2011. In January 2012, the Company filed a motion to dismiss the second amended complaint, which is scheduled to be heard by the Court on March 16, 2012. This lawsuit is subject to inherent uncertainties, and the actual cost will depend upon many unknown factors. The outcome of the litigation is necessarily uncertain, and the Company could be forced to expend significant resources in the defense of the suit and it may not prevail. The Company has not established any reserve for any potential liability relating to this lawsuit. The Company’s management believes that the Company has meritorious defenses and intends to defend the lawsuit vigorously. The Company believes it is entitled to coverage under its relevant insurance policies, subject to a $350,000 retention, but coverage could be denied or prove to be insufficient.
12. RESTRUCTURING
In response to the negative CONNECTION trial data, the Company implemented a restructuring in March 2010 in which it eliminated 23 full-time positions and vacated approximately 3,700 square feet of office space. Terminated individuals were eligible for a package consisting of a severance payment, continuing medical coverage and outplacement services. The Company recorded restructuring charges totaling $0.9 million during the first quarter of 2010, of which $0.4 million was classified as selling, general and administrative expense and $0.5 million was classified as research and development expense. No amounts related to the restructuring were recorded in 2011.
86
Table of Contents
13. SELECTED QUARTERLY FINANCIAL DATA (UNAUDITED)
The following table presents the unaudited quarterly results of operations of the Company for the years ended December 31, 2011 and 2010, respectively. The unaudited information is prepared on the same basis as the audited consolidated financial statements. The Company’s operating results for any quarter are not necessarily indicative of results for any future quarters or for a full year.
| Quarters Ended | ||||||||||||||||
| March 31, | June 30, | September 30, | December 31, | |||||||||||||
| 2011: |
||||||||||||||||
| Collaboration revenue |
$ | 14,709 | $ | 15,799 | $ | 14,940 | $ | 14,941 | ||||||||
| Operating expenses |
$ | (23,774 | ) | $ | (26,175 | ) | $ | (26,430 | ) | $ | (26,940 | ) | ||||
| Loss from operations |
$ | (9,065 | ) | $ | (10,376 | ) | $ | (11,490 | ) | $ | (11,999 | ) | ||||
| Net loss |
$ | (8,452 | ) | $ | (9,474 | ) | $ | (10,044 | ) | $ | (10,871 | ) | ||||
| Basic and diluted net loss per common share |
$ | (0.24 | ) | $ | (0.27 | ) | $ | (0.29 | ) | $ | (0.31 | ) | ||||
| Weighted-average common shares used in the calculation of basic and diluted net loss per common share |
34,663 | 34,888 | 34,909 | 35,375 | ||||||||||||
| 2010: |
||||||||||||||||
| Collaboration revenue |
$ | 15,734 | $ | 15,792 | $ | 14,350 | $ | 16,632 | ||||||||
| Operating expenses |
$ | (33,421 | ) | $ | (23,229 | ) | $ | (21,087 | ) | $ | (17,496 | ) | ||||
| Loss from operations |
$ | (17,687 | ) | $ | (7,437 | ) | $ | (6,737 | ) | $ | (864 | ) | ||||
| Net loss |
$ | (17,465 | ) | $ | (7,240 | ) | $ | (5,443 | ) | $ | (3,889 | ) | ||||
| Basic and diluted net loss per common share |
$ | (0.51 | ) | $ | (0.21 | ) | $ | (0.16 | ) | $ | (0.11 | ) | ||||
| Weighted-average common shares used in the calculation of basic and diluted net loss per common share |
33,953 | 34,053 | 34,570 | 34,573 | ||||||||||||
14. SUBSEQUENT EVENTS
(a) Termination of Pfizer Collaboration Agreement and Dimebon Program
In January 2012, the Company reported negative top line results from its Phase 3 CONCERT trial of dimebon in patients with mild-to-moderate Alzheimer’s disease. The Company previously had reported negative top line results from its Phase 3 CONNECTION trial of dimebon in patients with mild-to-moderate Alzheimer’s disease and its Phase 3 HORIZON trial of dimebon in patients with Huntington disease. In January 2012, Pfizer exercised its right to terminate the collaboration agreement and the Company and Pfizer discontinued development of dimebon for all indications. During the ensuing 180 days, the Company and Pfizer will work together to wind down their remaining collaboration activities.
The Company estimates that it and Pfizer will complete their collaboration wind down activities in the first half of 2012. Thus, during the first quarter of 2012 the Company revised its estimate of the remaining performance period under its former collaboration agreement with Pfizer. The Company expects the performance period to conclude in the first half of 2012. The remaining deferred revenue balance relating to the former collaboration agreement with Pfizer, which totaled $72.0 million at December 31, 2011, will be recognized as revenue over the remaining performance period.
87
Table of Contents
(b) Oakbrook Terrace Lease
In February 2012, the Company entered into a lease agreement for 15,336 square feet of office space located in Oakbrook Terrace, Illinois, which is intended to serve as its commercial headquarters following construction and build-out. The term of the lease is seven years and seven months, with an anticipated commencement date of May 2012. The Company selected this location for its commercial headquarters based on its proximity to the Company’s collaboration partner, Astellas. The annual non-cancelable rent payments are approximately $0.3 million per year. In connection with the execution of the lease, the Company delivered to the lessor a letter of credit collateralized by restricted cash totaling $0.8 million.
88
Table of Contents
Exhibit Index
| Incorporated By Reference | ||||||||||||||||||||
| Exhibit |
Exhibit Description |
Form | File No. | Exhibit | Filing Date | Filed Herewith | ||||||||||||||
| 3.1 | Amended and Restated Certificate of Incorporation. | 10-QSB | 000-20837 | 3.1 | (a) | 8/15/2005 | ||||||||||||||
| 3.2 | Certificate of Amendment of Amended and Restated Certificate of Incorporation. | 10-QSB | 000-20837 | 3.1 | (b) | 8/15/2005 | ||||||||||||||
| 3.3 | Certificate of Amendment to the Amended and Restated Certificate of Incorporation. | 10-QSB | 000-20837 | 3.1 | (c) | 8/15/2005 | ||||||||||||||
| 3.4 | Certificate of Designations of the Series C Junior Participating Preferred Stock of Medivation, Inc. | 10-KSB | 001-32836 | 3.1 | (d) | 2/19/2008 | ||||||||||||||
| 3.5 | Amended and Restated Bylaws of Medivation, Inc. | 10-K | 001-32836 | 3.2 | 3/16/2009 | |||||||||||||||
| 4.1 | Common Stock Certificate. | SB-2/A | 333-03252 | 4.1 | 6/14/1996 | |||||||||||||||
| 4.2 | Rights Agreement, dated as of December 4, 2006, between Medivation, Inc. and American Stock Transfer & Trust Company, as Rights Agent, which includes the form of Certificate of Designations of the Series C Junior Participating Preferred Stock of Medivation, Inc. as Exhibit A, the form of Right Certificate as Exhibit B and the Summary of Rights to Purchase Preferred Shares as Exhibit C. | 8-K | 001-32836 | 4.1 | 12/4/2006 | |||||||||||||||
| 10.1 | Warrant to purchase Common Stock of Medivation Neurology, Inc. assumed by Orion Acquisition Corp. II issued to Joseph J. Grano, Jr., dated as of June 8, 2004. | SB-2 | 333-122431 | 10.5 | (a) | 1/31/2005 | ||||||||||||||
| 10.2* | Warrant to purchase Common Stock of Medivation Neurology, Inc. assumed by Orion Acquisition Corp. II issued to David T. Hung, M.D., dated as of November 16, 2004. | SB-2 | 333-122431 | 10.6 | 1/31/2005 | |||||||||||||||
| 10.3* | Amended and Restated 2004 Equity Incentive Award Plan. | 10-KSB | 001-32836 | 10.4 | (a) | 2/19/2008 | ||||||||||||||
| 10.4* | Form of Stock Option Agreement under the 2004 Equity Incentive Award Plan. | 10-KSB | 000-20837 | 10.7 | (b) | 2/11/2005 | ||||||||||||||
| 10.5* | Form of Stock Option Agreement for Early Exercisable Options under the 2004 Equity Incentive Award Plan. | 10-KSB | 000-20837 | 10.7 | (c) | 2/11/2005 | ||||||||||||||
Table of Contents
| Incorporated By Reference | ||||||||||||||||||||||
| Exhibit |
Exhibit Description |
Form | File No. | Exhibit | Filing Date | Filed Herewith |
||||||||||||||||
| 10.6** | Amended and Restated Collaboration Agreement, dated as of October 20, 2008, between Medivation, Inc. and Pfizer Inc. | 10-Q | 001-32836 | 10.8 | 11/10/2008 | |||||||||||||||||
| 10.7* | Change of Control Severance Benefits Agreement, dated as of February 2, 2009, between Medivation, Inc. and David Hung, M.D. | 10-K | 001-32836 | 10.11 | 3/16/2009 | |||||||||||||||||
| 10.8* | Form of Medivation, Inc. Change of Control Severance Benefits Agreement. | 10-K | 001-32836 | 10.13 | 3/16/2009 | |||||||||||||||||
| 10.9** | Collaboration Agreement, dated as of October 26, 2009, by and between Medivation, Inc. and Astellas US LLC. | 10-K | 001-32836 | 10.15 | 3/15/2010 | |||||||||||||||||
| 10.10** | Amendment No. 1 to Collaboration Agreement, dated January 1, 2010, by and among Medivation, Inc., Astellas Pharma Inc. and Astellas US LLC | 10-Q | 001-32836 | 10.1 | 8/9/2011 | |||||||||||||||||
| 10.11** | Amendment No. 2 to Collaboration Agreement, dated May 13, 2011, by and among Medivation, Inc., Astellas Pharma Inc. and Astellas US LLC | 10-Q | 001-32836 | 10.2 | 8/9/2011 | |||||||||||||||||
| 10.12 | Office Lease Agreement, dated as of November 2, 2009, by and between Medivation, Inc. and PPF OFF 345 Spear Street, LP. | 10-K | 001-32836 | 10.16 | 3/15/2010 | |||||||||||||||||
| 10.13* | Compensation Information for Non-Employee Directors. | X | ||||||||||||||||||||
| 10.14** | Exclusive License Agreement, dated as of August 12, 2005, as amended through October 21, 2009, by and between Medivation, Inc. and The Regents of the University of California. | 10-Q/A | 001-32836 | 10.18 | 8/20/2010 | |||||||||||||||||
| 10.15 | Office Lease, dated April 18, 2007, by and between CREA Spear Street Terrace LLC and Medivation, Inc. | 10-K | 001-32836 | 10.16 | 3/16/2011 | |||||||||||||||||
| 10.16 | Sublease, dated November 10, 2008, by and between MacFarlane Partners Investment Management, LLC and Medivation, Inc. | 10-K | 001-32836 | 10.17 | 3/16/2011 | |||||||||||||||||
| 10.17 | First Amendment to Lease, dated September 16, 2009, by and between CREA Spear Street Terrace LLC and Medivation, Inc. | 10-K | 001-32836 | 10.18 | 3/16/2011 | |||||||||||||||||
Table of Contents
| Incorporated By Reference | ||||||||||||||||||||||
| Exhibit |
Exhibit Description |
Form | File No. | Exhibit | Filing Date | Filed Herewith |
||||||||||||||||
| 10.18 | Second Amendment to Lease, dated November 30, 2010, by and between CREA Spear Street Terrace LLC and Medivation, Inc. | 10-K | 001-32836 | 10.19 | 3/16/2011 | |||||||||||||||||
| 10.19* | Bonuses for Fiscal Year 2010 and Base Salaries for Fiscal Year 2011 for Certain Executive Officers. | 10-K | 001-32836 | 10.20 | 3/16/2011 | |||||||||||||||||
| 10.20* | Medivation, Inc. 2011 Bonus Plan Summary. | 10-K | 001-32836 | 10.21 | 3/16/2011 | |||||||||||||||||
| 10.21* | Form of Restricted Stock Unit Grant Notice and Agreement under the 2004 Equity Incentive Award Plan. | 10-K | 001-32836 | 10.22 | 3/16/2011 | |||||||||||||||||
| 10.22* | Separation Agreement, dated as of September 21, 2011, between Medivation, Inc. and Rohan Palekar. | 8-K | 001-32836 | 10.1 | 9/27/2011 | |||||||||||||||||
| 10.23* | Bonuses for Fiscal Year 2011 and Base Salaries for Fiscal Year 2012 for Certain Executive Officers | 8-K | 001-32836 | 10.1 | 12/15/2011 | |||||||||||||||||
| 10.24* | Medivation, Inc. 2012 Bonus Plan Summary | 8-K | 001-32836 | 10.2 | 12/15/2011 | |||||||||||||||||
| 10.25* | Form of Indemnification Agreement for directors and officers | X | ||||||||||||||||||||
| 10.26* | Offer Letter, dated August 31, 2011, by and between Medivation, Inc. and Cheryl Cohen. |
X | ||||||||||||||||||||
| 10.27 | Office Lease, dated as of December 28, 2011, by and between Knickerbocker Properties, Inc. XXXIII and Medivation, Inc. | X | ||||||||||||||||||||
| 10.28* | Form of Stock Appreciation Right Grant Notice and Agreement under the 2004 Equity Incentive Award Plan | X | ||||||||||||||||||||
| 10.29* | Form of Performance Share Award Grant Notice and Agreement under the 2004 Equity Incentive Award Plan | X | ||||||||||||||||||||
| 21.1 | Subsidiaries of Medivation, Inc. | X | ||||||||||||||||||||
| 23.1 | Consent of Independent Registered Public Accounting Firm. | X | ||||||||||||||||||||
| 24.1 | Power of Attorney (contained on signature page). | X | ||||||||||||||||||||
| 31.1 | Certification pursuant to Rule 13a-14(a)/15d-14(a). | X | ||||||||||||||||||||
| 31.2 | Certification pursuant to Rule 13a-14(a)/15d-14(a). | X | ||||||||||||||||||||
Table of Contents
| Incorporated By Reference | ||||||||||||||
| Exhibit |
Exhibit Description |
Form | File No. | Exhibit | Filing Date | Filed Herewith |
||||||||
| 32.1† | Certifications of Chief Executive Officer and Chief Financial Officer. | X | ||||||||||||
| 101.INS# | XBRL Instance Document. | X | ||||||||||||
| 101.SCH# | XBRL Taxonomy Extension Schema Document. | X | ||||||||||||
| 101.CAL# | XBRL Taxonomy Extension Calculation Linkbase Document | X | ||||||||||||
| 101.DEF# | XBRL Taxonomy Extension Definition Linkbase Document | X | ||||||||||||
| 101.LAB# | XBRL Taxonomy Extension Labels Linkbase Document | X | ||||||||||||
| 101.PRE# | XBRL Taxonomy Extension Presentation Linkbase Document | X | ||||||||||||
| * | Indicates management contract or compensatory plan or arrangement. |
| ** | Confidential treatment has been granted with respect to certain portions of this exhibit. |
| † | The certifications attached as Exhibit 32.1 accompany this Annual Report on Form 10-K, are not deemed filed with the Securities and Exchange Commission and are not to be incorporated by reference into any filing of Medivation, Inc., under the Securities Act of 1933, as amended, or the Securities Exchange Act of 1934, as amended, whether made before or after the date of this Annual Report on Form 10-K, irrespective of any general incorporation language contained in such filing. |
| # | Pursuant to applicable securities laws and regulations, the Registrant is deemed to have complied with the reporting obligation related to the submission of interactive data files in such exhibits and is not subject to liability under any anti-fraud provisions of the federal securities laws as long as the Registrant has made a good faith attempt to comply with the submission requirements and promptly amends the interactive data files after becoming aware that the interactive data files fails to comply with the submission requirements. These interactive data files are deemed not filed or part of a registration statement or prospectus for purposes of sections 11 or 12 of the Securities Act, are deemed not filed for the purposes of section 18 of the Exchange Act and otherwise are not subject to liability under those sections. |