Table of Contents
| ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) |
| OF THE SECURITIES EXCHANGE ACT OF 1934 |
| TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) |
| OF THE SECURITIES EXCHANGE ACT OF 1934 |
| (State or other jurisdiction of incorporation or organization) |
(I.R.S. Employer Identification No.) |
| Title of each class |
Trading Symbol(s) |
Name of each exchange on which registered | ||
| Securities registered pursuant to Section 12(g) of the Act: |
None |
| Accelerated filer ☐ | Non-accelerated filer ☐ |
Smaller reporting company | ||||
| Emerging growth company | ||||||
Table of Contents
WATERS CORPORATION AND SUBSIDIARIES
ANNUAL REPORT ON FORM 10-K
INDEX
Table of Contents
PART I
Item 1: Business
General
Waters Corporation (the “Company,” “Waters,” “we,” “our,” or “us”), a global leader in analytical instruments and software, has pioneered innovations in chromatography, mass spectrometry and thermal analysis serving life, materials and food sciences for more than 65 years. With approximately 7,900 employees worldwide, Waters operates directly in over 35 countries and has products available in more than 100 countries. The Company primarily designs, manufactures, sells and services high-performance liquid chromatography (“HPLC”), ultra-performance liquid chromatography (“UPLC” and together with HPLC, referred to as “LC”) and mass spectrometry (“MS”) technology systems and support products, including chromatography columns, other consumable products and comprehensive post-warranty service plans. These systems are complementary products that are frequently employed together (“LC-MS”) and sold as integrated instrument systems using common software platforms. In addition, the Company designs, manufactures, sells and services thermal analysis, rheometry and calorimetry instruments through its TA InstrumentsTM (“TA”) product line. The Company is also a developer and supplier of advanced software-based products that interface with the Company’s instruments, as well as other manufacturers’ instruments.
The Company’s products are used by pharmaceutical, clinical, biochemical, industrial, nutritional safety, environmental, academic and governmental customers working in research and development, quality assurance and other laboratory applications. LC is a standard technique and is utilized in a broad range of industries to detect, identify, monitor and measure the chemical, physical and biological composition of materials, and to purify a full range of compounds. MS technology, principally in conjunction with chromatography, is employed in drug discovery and development, including clinical trial testing, the analysis of proteins in disease processes (known as “proteomics”), nutritional safety analysis and environmental testing. LC-MS instruments combine a liquid phase sample introduction and separation system with mass spectrometric compound identification and quantification. The Company’s thermal analysis, rheometry and calorimetry instruments are used in predicting the suitability and stability of fine chemicals, pharmaceuticals, water, polymers, metals and viscous liquids for various industrial, consumer goods and healthcare products, as well as for life science research.
Waters Corporation, organized as a Delaware corporation in 1991, is a holding company that owns all of the outstanding common stock of Waters Technologies Corporation, its operating subsidiary. Waters Corporation became a publicly traded company with its initial public offering (“IPO”) in November 1995. Since the IPO, the Company has added three significant and complementary technologies to its range of products with the acquisitions of TA Instruments in May 1996, Micromass Limited in September 1997 and Wyatt Technology in May 2023. On May 16, 2023, the Company completed the acquisition of Wyatt Technology, LLC and its three operating subsidiaries, Wyatt Technology Europe GmbH, Wyatt Technology France and Wyatt Technology UK Ltd. (collectively, “Wyatt”), for a total purchase price of $1.3 billion in cash. Wyatt is a pioneer in innovative light scattering and field-flow fractionation instruments, software, accessories and services. The acquisition will expand WatersTM portfolio and increase exposure to large molecule applications.
Business Segments
The Company’s business activities, for which discrete financial information is available, are regularly reviewed and evaluated by the chief operating decision maker. As a result of this evaluation, the Company determined that it has two operating segments: Waters and TA. The Waters operating segment is primarily in the business of designing, manufacturing, selling and servicing LC and MS instrument systems, columns and other precision chemistry consumables that can be integrated and used along with other analytical instruments. The TA operating segment is primarily in the business of designing, manufacturing, selling and servicing thermal analysis, rheometry and calorimetry instruments. The Company’s two operating segments have similar economic characteristics; product processes; products and services; types and classes of customers; methods of distribution;
1
Table of Contents
and regulatory environments. Because of these similarities, the two segments have been aggregated into one reporting segment for financial statement purposes. Operations of the recently acquired Wyatt business are part of the Waters operating segment.
Information concerning revenues and long-lived assets attributable to each of the Company’s products, services and geographic areas is set forth in Note 18 in the Notes to the Consolidated Financial Statements, which is incorporated herein by reference.
Waters Products and Markets
High-Performance and Ultra-Performance Liquid Chromatography
HPLC is a standard technique used to identify and analyze the constituent components of a variety of chemicals and other materials. The Company believes that HPLC’s performance capabilities enable it to separate, identify and quantify a high proportion of all known chemicals. As a result, HPLC is used to analyze substances in a wide variety of industries for research and development purposes, quality control and process engineering applications.
The most significant end-use markets for HPLC are those served by the pharmaceutical and life science industries. In these markets, HPLC is used extensively to understand diseases, identify new drugs, develop manufacturing methods and assure the potency and purity of new pharmaceuticals. HPLC is also used in a variety of other applications, such as analyses of foods and beverages for nutritional labeling and compliance with safety regulations and the testing of water and air purity within the environmental testing industry, as well as applications in other industries, such as chemical and consumer products. Waters also has in vitro diagnostic labelled products that are used as general-purpose instruments for clinical diagnostic applications, such as newborn screening and therapeutic drug management, in countries where these products are registered. HPLC is also used by universities, research institutions and governmental agencies, such as the United States Food and Drug Administration (“FDA”) and the United States Environmental Protection Agency (“EPA”) and their foreign counterparts that mandate safety and efficacy testing.
In 2004, Waters introduced a novel technology that the Company describes as ultra-performance liquid chromatography that utilizes a packing material with small, uniform diameter particles and a specialized instrument, the ACQUITYTM UPLCTM System, to accommodate the increased pressure and narrower chromatographic bands that are generated by these small and tightly packed particles. By using the ACQUITY UPLC System, researchers and analysts are able to achieve more comprehensive chemical separations and faster analysis times in comparison with many analyses previously performed by HPLC. In addition, in using the ACQUITY UPLC System, researchers have the potential to extend the range of applications beyond that of HPLC, enabling them to uncover more levels of scientific information. While offering significant performance advantages, the ACQUITY UPLC System is also compatible with the Company’s software products and the general operating protocols of HPLC. For these reasons, the Company’s customers and field sales and support organizations are well positioned to utilize this innovative technology and instrument. In 2018, the Company introduced the ACQUITY ArcTM Bio System, a versatile, iron-free, bio-inert, quaternary liquid chromatograph specifically engineered to improve bioseparation analytical methods. The Company also introduced the ACQUITY UPLC PLUS System series in 2018, consisting of the ACQUITY UPLC H-Class PLUS System, ACQUITY UPLC H-Class PLUS Bio System and ACQUITY UPLC I-Class PLUS Systems, which incorporate foundational enhancements into the legacy systems.
Waters manufactures LC instruments that are offered in configurations that allow for varying degrees of automation, from component configured systems for academic teaching and research applications to fully automated systems for regulated and high sample throughput testing, and that have a variety of detection technologies, from optical-based ultra-violet absorbance, refractive index and fluorescence detectors to a suite of MS-based detectors, optimized for certain analyses.
2
Table of Contents
In 2019, the Company introduced the ACQUITY Advanced Polymer ChromatographyTM System, which is the first fully solvent-compatible UPLC System to perform size exclusion, gradient polymer elution and solvent compatible reversed-phase liquid chromatographic separations on a single platform. The all-in-one system gives research scientists greater analytical versatility and speed when conducting research on next-generation polymers. In 2020, the Company introduced the Waters Arc HPLC System, a new HPLC system for routine testing in the pharmaceutical, food, academic and materials markets. A key target application is quality control in laboratories performing batch release tests on small molecule pharmaceuticals. In 2021, the Company introduced the new ACQUITY Premier LC solution and the Arc Premier System both featuring Waters MaxPeakTM High Performance Surface (“HPS”) Technology. MaxPeak HPS Technology, which was first introduced with the Company’s introduction of ACQUITY Premier Columns in 2020, is a surface technology that forms a barrier between the sample and the metal surfaces of both the system and column, eliminating the need for system passivation, mitigating the loss of metal-sensitive analytes and yielding higher quality data in less time and effort.
During the second half of 2023, Waters introduced the DynaProTM ZetaStarTM instrument through its Wyatt TechnologyTM portfolio for nanoparticle analysis. The new instrument simultaneously enables dynamic and static light scattering and dynamic and electrophoretic light scattering measurements, all in one device. By combining multiple light scattering techniques and automatically assessing data quality and performing adaptive data capture, the ZetaStar instrument delivers both increased sensitivity and faster measurements to aid the precise development of complex biologics, using extremely low sample volumes.
The primary consumable products for LC instruments are chromatography columns. These columns are packed with separation media used in the LC testing process and are typically replaced at regular intervals. The chromatography column contains one of several types of packing material, typically stationary phase particles made from silica or polymeric resins. As a pressurized sample is introduced to the column inlet and permeates through the packed column, it is separated into its constituent components.
Waters HPLC columns can be used on Waters-branded and competitors’ LC systems. The Company believes that it is one of a few suppliers in the world that manufactures silica and polymeric resins, packs columns and distributes its own products. In doing so, the Company believes it can better ensure product consistency, a key attribute for its customers in quality control laboratories and can react quickly to new customer requirements. The Company believes that its ACQUITY UPLC Columns are used primarily on its ACQUITY UPLC Systems and, furthermore, that its ACQUITY UPLC Systems primarily use ACQUITY UPLC Columns. In 2019, the Company introduced the BioResolveTM SCX mAb Columns and VanGuardTM FIT Cartridge technologies. These new cation exchange column lines with specialized consumables are designed to simplify and improve the characterization and monitoring of monoclonal antibody (“mAb”) therapeutics, as well as enable mAb charge-variant analyses as required by the World Health Organization, the FDA and the International Conference on Harmonization for confirming the efficacy and safety of biologics and biosimilars with discovery, development and manufacturing applications. In 2020, Waters introduced ACQUITY Premier Columns, at the time a new family of premium sub-2-micron columns featuring MaxPeak HPS Technology. The columns are for use with any brand of UPLC System and can measurably improve data quality by mitigating the loss of sample analytes due to analyte-to-surface interactions.
The Company’s precision chemistry consumable products also include environmental and nutritional safety testing products, including Certified Reference Materials and Proficiency Testing products. Laboratories around the world and across multiple industries use these products for quality control and proficiency testing and also purchase product support services required to help with their federal and state mandated accreditation requirements or with quality control over critical pharmaceutical analysis.
In 2023, the Company introduced the first in a new line of size exclusion chromatography columns aimed at improving analysis while lowering the cost of gene therapies, specifically adeno-associated viral (“AAV”) vectors. The new Waters XBridgeTM Premier GTx BEHTM size exclusion chromatography columns double the
3
Table of Contents
speed of measuring the potency and safety of AAVs. Combining the columns with light scattering technologies from its Wyatt Technology portfolio deepens the level of information acquired from a single experiment and optimizes the manufacturing of these novel gene delivery vehicles.
In 2020, the Company acquired all of the outstanding stock of Andrew Alliance, S.A. (“Andrew Alliance”). Andrew Alliance offers lab workflow automation solutions with the combination of its software platform and smart, connected laboratory equipment and accessories. The addition of Andrew Alliance to our portfolio has allowed us to positively impact our customers’ workflows by improving the repeatability, performance and speed of laboratory operations and chemistry workflows.
In 2023, the Company introduced the AllianceTM iS HPLC System, the next-generation intelligent HPLC System, designed to reduce compliance risk by adding new levels of proactive error detection, troubleshooting and ease-of-use. When combined with Waters compliance-ready EmpowerTM Chromatography Software and eConnectTM HPLC Columns, the Alliance iS HPLC System streamlines the task of making accurate and precise measurements by detecting and eliminating common errors. In doing so, the Alliance iS HPLC System helps quality control laboratories to consistently meet quality, safety, compliance and on-time product delivery goals. This system also integrates with the cloud-native waters_connectTM System Monitoring Software enabling real-time monitoring of the Alliance iS HPLC System and any other chromatography instruments controlled by Empower Software. Laboratory managers can view the live status of their HPLC instrument fleet from anywhere and at any time to further improve equipment utilization and overall productivity.
Also in 2023, the Company introduced the new bioprocess walk-up solutions designed to further simplify biologic sample preparation and analysis. This solution eliminated the need to send bioreactor samples to a central laboratory for analysis making it even easier to accelerate upstream bioprocess development by up to six weeks over traditional methods.
Mass Spectrometry and Liquid Chromatography-Mass Spectrometry
MS is a powerful analytical technology that is used to identify unknown compounds, to quantify known materials and to elucidate the structural and chemical properties of molecules by measuring the masses of molecules that have been converted into ions.
The Company is a technology and market leader in the development, manufacture, sale and service of MS instruments and components. These instruments are typically integrated and used along with other complementary analytical instruments and systems, such as LC, chemical electrophoresis and gas chromatography. A wide variety of instrumental designs fall within the overall category of MS instrumentation, including devices that incorporate quadrupole, ion trap, time-of-flight (“Tof”), magnetic sector and ion mobility technologies. Furthermore, these technologies are often used in tandem to maximize the speed and/or efficacy of certain experiments.
Currently, the Company offers a wide range of MS instrument systems utilizing various combinations of quadrupole, Tof and ion mobility designs. These instrument systems are used in drug discovery and development, as well as for environmental, clinical and nutritional safety testing. The overwhelming majority of mass spectrometers sold by the Company are designed to utilize an LC system and a liquid compatible interface (such as an electrospray ionization source) as the sample introduction device. These products supply a diverse market with a strong emphasis on the pharmaceutical, biomedical, clinical, food and beverage and environmental market segments worldwide.
MS is an increasingly important detection technology for LC. The Company’s smaller-sized mass spectrometers, such as the single quadrupole detector and the tandem quadrupole detector (“TQD”), are often referred to as LC “detectors” and are typically sold as part of an LC system or as an LC system upgrade. Larger quadrupole systems, such as the XevoTM TQ MS System and Xevo TQ-S MS System, are used primarily for experiments performed for late-stage drug development, including clinical trial testing. Quadrupole time-of-flight
4
Table of Contents
(“Q-Tof”) instruments, such as the Company’s SYNAPTTM G2-S HDMS System, are often used to analyze the role of proteins in disease processes, an application sometimes referred to as “proteomics.”
LC and MS are typically embodied within an analytical system tailored for either a dedicated class of analyses or as a general-purpose analytical device. An increasing percentage of the Company’s customers are purchasing LC and MS components simultaneously and it has become common for LC and MS instrumentation to be used within the same laboratory and operated by the same user. The descriptions of LC and MS above reflect the historical segmentation of these analytical technologies and the historical categorization of their respective practitioners. Increasingly in today’s instrument market, this segmentation and categorization is becoming obsolete as a high percentage of instruments used in the laboratory embody both LC and MS technologies as part of a single device. In response to this development and to further promote the high utilization of these hybrid instruments, the Company has organized its Waters operating segment to develop, manufacture, sell and service integrated LC-MS systems.
In 2019, the Company introduced the BioAccordTM System, a liquid chromatography-mass spectrometry solution that expands access to high-resolution time-of-flight mass spectrometry capabilities. The system provides new levels of user experience with automated setup and self-diagnosis delivered through an intuitive user interface. Also in 2019, the Company introduced the SELECT SERIESTM CyclicTM IMS System, which seamlessly integrates cyclic ion mobility technology into a high-performance research-grade time-of-flight mass spectrometer. In addition, the Company introduced the SYNAPT XS System, a new highly flexible, high-resolution mass spectrometer for research and development labs focused on discovery applications. The Company also reinforced its tandem quadrupole mass spectrometry portfolio during the current year with upgrades to the Xevo TQ-S micro MS System and the introduction of the new Xevo TQ-S cronos MS System. The Xevo TQ-S micro System features new performance enhancements that bring the quantitation of highly polar, ionic compounds in food to a higher level. The Xevo TQ-S cronos System is a new, tandem quadrupole mass spectrometer purposely built for routine quantitation of large numbers of small-molecule organic compounds over a wide concentration range. The Xevo TQ-S micro System and the Xevo TQ-S cronos System are also well suited to meet regulatory requirements for pesticide residue analysis, the monitoring for contaminants in processed foods, identifying drugs of abuse, and performing impurity profiling of pharmaceuticals. In 2020, the Company introduced the RADIANTM ASAPTM System, a novel direct mass detector engineered for non-mass spectrometry experts to conduct fast and accurate analyses of solids and liquids with minimal sample prep. Also in 2020, the Company introduced enhancements for the Waters Xevo G2-XS QTof System, SYNAPT XS System and SELECT SERIES Cyclic IMS System, including a new fragmentation technique and imaging option.
In 2021, the Company introduced the SELECT SERIES MRT MS System, a high-resolution mass spectrometer that combines Multi-Reflecting Time-of-Flight (“MRT”) technology with enhanced desorption electrospray ionization and new matrix-assisted laser desorption ionization imaging sources. The platform will serve as the basis for Waters next generation Tof instruments with applications in pharmaceutical, biomedical, natural products, and materials research. Also in 2021, the Company released the ACQUITY RDaTM Detector featuring SmartMSTM Technology, the company’s newest Tof MS designed to improve the ease and reliability of small molecule analysis for pharmaceutical, academic, food, and forensic applications. The Company also introduced a new peptide multi-attribute method workflow for the BioAccord LC-MS System in 2021, which is an end-to-end workflow for analyzing monoclonal antibodies and other protein and peptide-based drugs.
In 2022, the Company introduced the Xevo TQ Absolute System, the most sensitive and compact benchtop tandem mass spec in its class. The Company introduced the new Xevo G3 Q-Tof Mass Spectrometer with CONFIRM Sequence, a new oligonucleotide sequencing confirmation app for the waters_connect Software platform and an electrospray ionization source for the high-resolution SELECT SERIES MRT Mass Spectrometer.
5
Table of Contents
Based upon 2023 reports from independent marketing research firms and publicly disclosed sales figures from competitors, the Company believes that it is one of the world’s largest manufacturers and distributors of LC and LC-MS instrument systems, chromatography columns and other consumables and related services.
The Company has been a developer and supplier of software-based products that interface with both the Company’s and other suppliers’ instruments. The Company’s newest software technology for mass spectrometry is the waters_connect Software platform. In 2019, the Company introduced the first of a series of applications on this platform supporting the BioAccord System and the Xevo G2 XS Mass Spectrometer. These applications support biopharmaceutical workflows, simplifying the collection of often complex LCMS data for use in biopharmaceutical development and into QC where it is used to assure the quality of existing medicines and new drug formulations. The platform design of waters_connect Software has enabled rapid delivery of several major updates including new biopharma application workflows designed in close collaboration with biopharmaceutical innovators to solve specific challenges they face with existing solutions. The platform also provides the foundation for the connected lab of the future where data is no longer siloed but can be securely shared among a community of connected scientists. The waters_connect Software joins the existing suite of informatics products: Empower Chromatography Data Software, MassLynxTM Mass Spectrometry Software and NuGenesisTM Scientific Data Management System, each of which is used to support innovations within world-leading institutions. In 2020, Waters announced the availability of Empower BC LAC/ETM Solution with SecureSync, an enhanced solution to preserve the ability for laboratories to work locally when organizations with distributed laboratory environments experience an enterprise interruption.
On December 15, 2020, the Company acquired all of the outstanding stock of Integrated Software Solutions Pty Limited and its two operating subsidiaries Integrated Software Solutions Limited and Integrated Software Solutions USA, LLC (collectively, “ISS”). ISS offers clinical laboratory software systems that support and further expand product offerings within our clinical business. The net assets acquired primarily relate to ISS’ laboratory information system, OMNI-Lab.
In 2022, the Company introduced a new Per-and Polyfluoroalkyl Substances (“PFAS”) quantitation workflow enabled by enhancements to its waters_connect Software for quantitation software and the Company introduced Extraction+TM Connected Device, a new software-controlled product for the Waters Andrew+TM Pipetting Robot that automates the preparation of biological, food, forensics and environmental samples by solid phase extraction.
In 2023, the Company introduced the next generation Xevo TQ Absolute IVD Mass Spectrometer, expanding its family of MassTrakTM IVD LC-MS/MS Systems for clinical diagnostic applications. The powerful analytical performance of the Xevo TQ Absolute IVD Mass Spectrometer is up to five times more sensitive for quantifying clinical analytes. This sensitivity enables clinical laboratories to detect and measure trace level analytes within a sample at lower detection levels than previously possible. It can also extend the testing capabilities of the clinical laboratory to include lower volume samples obtained in less-invasive assays such as saliva, breath, dried blood spots and multiplex panels and large molecules. The new MassTrak LC-MS/MS IVD System includes the ACQUITY UPLC I-Class PLUS System with the Xevo TQ Absolute IVD Mass Spectrometer. The ACQUITY UPLC I-Class PLUS System is designed to deliver rapid and accurate sample analysis to enhance the sensitivity of any mass spectrometer and simplify the characterization of the most complex sample. The Xevo TQ Absolute IVD System provides more consistent instrument-to-instrument performance, with a user-friendly design that maximizes service uptime. Its innovative design is also 45% smaller and uses 50% less nitrogen gas and electricity than comparable tandem quadrupole-mass spectrometry systems, making it ideal for hospital labs and independent commercial labs with both sustainability and business growth goals to meet.
In addition, in 2023, the Company introduced the industry’s first targeted imaging mass spectrometer based on its Xevo TQ Absolute Tandem Quadrupole Mass Spectrometer which is the most sensitive and compact mass spectrometer in its class. The new instrument combines the Waters DESI XS source with the Xevo TQ Absolute
6
Table of Contents
System and is five times more sensitive and five times faster than discovery-based imaging systems at precisely determining whether a particular small molecule drug product, and how much of it, reaches its intended target, such as a brain, liver or lung, in a test subject.
Also in 2023, the Company announced new updates to its SELECT SERIES MRT System that increases its specificity and utility for UPLC-MS/MS metabolomics and drug discovery applications and for mass spectrometry imaging experiments. The MRT System now offers 50% higher resolution, making it capable of 300,000 FWHM resolution, a 3X faster scan rate and parts-per-billion mass accuracy. These MRT System enhancements are designed to help research scientists unambiguously identify analytes of interest in samples of blood, urine and tissue, contributing to a greater understanding of molecules and their mechanisms of action in numerous scientific fields. It is compatible with numerous MS imaging sources including DESI and MALDI, and generates crystal-clear, high-resolution images without compromising mass spectral resolution or accuracy.
In addition, in 2023, the Company combined its BioAccord LC-MS System and the Waters Andrew+ Pipetting Robot, connecting via new protocols in OneLabTM Software to create fully integrated and easy-to-use bioprocess walk-up solutions. It is designed to enable less experienced LC-MS users to acquire critical quality attribute data for analysis of drug product and cell culture media. Capturing data directly at the bioproduction laboratory can help bioprocess engineers improve process understanding, leading to more robust manufacturing processes and accelerated development timelines.
Waters Service
Services provided by Waters enable customers to maximize technology productivity, support customer compliance activities and provide transparency into enterprise resource management efficiencies. The customer benefits from improved budget control, data-driven technology adoption and accelerated workflow at a site or on a global perspective. The Company considers its service offerings to be highly differentiated from its competition, as evidenced by a consistent increase in annual service revenues. The Company’s principal competitors in the service market include Revvity, Inc., Agilent Technologies, Inc. and Thermo Fisher Scientific Inc. These competitors can provide certain services on Waters instruments to varying degrees and always present competitive risk.
The servicing and support of instruments, software and accessories is an important source of revenue and represented over 35% of sales for Waters in 2023. These revenues are derived primarily through the sale of support plans, demand services, spare parts, customer performance validation services and customer training. Support plans typically involve scheduled instrument maintenance and an agreement to promptly repair a non-functioning instrument in return for a fee described in a contract that is priced according to the configuration of the instrument.
The waters_connect System Monitoring is a new Software-as-a-Service application that enables laboratory managers and analysts to monitor the real-time status of any chromatography instruments, regardless of the manufacturer, running on Waters Empower Software. The waters_connect System Monitoring application was developed especially for high-volume quality assurance/quality control laboratories. It can reduce the turnaround time of product release samples and facilitate the planning and progress of critical analyses via live, at-a-glance dashboard views of the operational status of chromatography instruments. The cloud-native application also helps lab managers utilize capital resources better by providing an understanding of instrument history and usage levels and improve the productivity of their teams.
TA Products and Markets
Thermal Analysis, Rheometry and Calorimetry
Thermal analysis measures the physical or thermodynamic characteristics of materials as a function of temperature. Changes in temperature affect several characteristics of materials, such as their heat flow
7
Table of Contents
characteristics, physical state, weight, dimension and mechanical and electrical properties, which may be measured by one or more thermal analysis techniques, including calorimetry. Consequently, thermal analysis techniques are widely used in the development, production and characterization of materials in various industries, such as plastics, chemicals, automobiles, pharmaceuticals and electronics.
Rheometry instruments often complement thermal analyzers in characterizing materials. Rheometry characterizes the flow properties of materials and measures their viscosity, elasticity and deformation under different types of “loading” or other conditions. The information obtained under such conditions provides insight into a material’s behavior during processing, packaging, transport, usage and storage.
Thermal analysis, rheometry and calorimetry instruments are heavily used in material testing laboratories and, in many cases, provide information useful in predicting the suitability and stability of industrial polymers, fine chemicals, pharmaceuticals, water, metals and viscous liquids in various industrial, consumer goods and healthcare products, as well as for life science research. As with systems offered by Waters, a range of instrument configurations is available with increasing levels of sample handling and information processing automation. In addition, systems and accompanying software packages can be tailored for specific applications.
In 2020, TA introduced the new DiscoveryTM X3TM Differential Scanning Calorimeter (“X3 DSC”), Discovery Hybrid Rheometers and TAM IV Micro XL isothermal microcalorimeter. The X3 DSC accelerates productivity in customers’ laboratories by enabling three samples to be measured in a single experiment, compared to the single-sample series operation of the other available DSC offerings in the market. This particularly addresses a need in high-throughput laboratories in industries such as composites, electronics and polymer manufacturing. The new line of Discovery Hybrid Rheometers provides increased sensitivity and versatility of rheometry measurements, supporting the development of next-generation, high-performance materials by improving the productivity and efficiency of materials science research. The TAM IV Micro XL isothermal calorimeter supports the development of new battery chemistries by measuring self-discharge and unwanted reactions that reduce battery life and efficiency.
In 2021, TA introduced the TMA 450 RH Instrument and the Discovery SA Instrument. The TMA 450 RH Instrument provides measurements of dimensional compatibility of materials under controlled temperature and humidity that are important for the development of new electronic devices. The Discovery SA Instrument is used in pharmaceutical development to assess the impact of moisture in drug product processing and storage on crystalline structure, which is related to drug product efficacy.
In 2021, TA introduced the TRIOSTM AutoPilot Software for its thermal analyzer product line. This software helps laboratory staff using TA’s thermal analyzers create routine and streamlined standard operating procedures improving the speed and productivity of thermal analysis measurements.
In 2022, TA introduced the Powder Rheology Accessory, which enables our Discovery Hybrid Rheometers to characterize the behavior of powders during storage, dispensing, processing and end-use. The Powder Rheology Accessory provides relevant property and processing measurements for battery electrode coatings to prevent defects that cause cell failure and pharmaceutical tablets to prevent instabilities of API blends.
In 2022, TA introduced Polymer Workflow Guided Methods, which provides walk up and use functionality by codifying polymer workflows. Guided Methods leverages the power of TRIOS AutoPilot Software and enables novice users to quickly learn and use the instrument to set up test methods, run tests, and execute analyses across our Thermal Analysis and Rheology product lines.
In 2023, TA introduced a new Battery Cycler Microcalorimeter Solution for high-resolution characterization of battery cells. The instrument and software combination enables non-destructive testing under real-world operating conditions and significantly reduces experiment time from months to weeks, while providing decisive insights for greater battery efficiency, safety and stability.
8
Table of Contents
TA Service
Similar to Waters, the servicing and support of TA’s instruments is an important source of revenue and represented more than 25% of sales for TA in 2023. TA operates independently from the Waters operating segment, though many of its overseas offices are jointly occupied with Waters to achieve operational efficiencies. TA has dedicated field sales and service operations. Service sales are primarily derived from the sale of support plans, replacement parts and billed labor fees associated with the repair, maintenance and upgrade of installed systems.
Global Customers
The Company typically has a broad and diversified customer base that includes pharmaceutical accounts, other industrial accounts, universities and governmental agencies. Purchase of the Company’s instrument systems is often dependent on its customers’ capital spending, or funding as in the cases of academic, governmental and research institutions, which often fluctuate from year to year. The pharmaceutical segment represents the Company’s largest sector and includes multinational pharmaceutical companies, generic drug manufacturers, contract research organizations (“CROs”) and biotechnology companies. The Company’s other industrial customers include chemical manufacturers, polymer manufacturers, food and beverage companies and environmental testing laboratories. The Company also sells to universities and governmental agencies worldwide. The Company’s technical sales and support staff members work closely with its customers in developing and implementing applications that meet their full range of analytical requirements. During 2023, 57% of the Company’s net sales were to pharmaceutical accounts, 31% to other industrial accounts and 12% to academic institutions and governmental agencies. Although the Company transacts business with various government agencies, no government contract is of such magnitude that a renegotiation of profits or termination of the contract at the election of the government agency would have a material adverse effect on the Company’s financial results.
The Company typically experiences seasonality in its orders that is reflected as an increase in sales in the fourth quarter, as a result of purchasing habits for capital goods of many customers who tend to exhaust their spending budgets by calendar year-end. The Company does not rely on any single customer for a material portion of its sales. During fiscal years 2023, 2022 and 2021, no single customer accounted for more than 2% of the Company’s net sales.
Sales and Service
The Company has one of the largest direct sales and service organizations focused exclusively on the analytical workflows offered by the Company. Across these product technologies, using respective specialized sales and service workforces, the Company serves its customer base with 85 sales offices throughout the world as of December 31, 2023 and approximately 4,300, 4,500 and 4,300 field representatives in 2023, 2022 and 2021, respectively. This investment in sales and service personnel serves to maintain and expand the Company’s installed base of instruments. The Company’s sales representatives have direct responsibility for account relationships, while service representatives work in the field to install instruments, train customers and minimize instrument downtime. In-house and field-based technical support representatives work directly with customers, providing them assistance with applications and procedures on Company products. The Company provides customers with comprehensive information through various corporate and geographic-specific internet websites and product literature, and also makes consumable products available through electronic ordering facilities and a dedicated catalog.
Manufacturing and Distribution
The Company provides high product quality by overseeing each stage of the production of its instruments, columns and chemical reagents.
9
Table of Contents
The Company currently assembles a portion of its LC instruments at its facility in Milford, Massachusetts, where it performs machining, assembly and testing. The Milford facility maintains quality management and environmental management systems in accordance with the requirements of ISO 9001:2015, ISO 13485:2016, ISO 45001:2018 and ISO 14001:2015, and adheres to applicable regulatory requirements (including the FDA Quality System Regulation and the European In-Vitro Diagnostic Directive). The Company outsources manufacturing of certain electronic components, such as computers, monitors and circuit boards, to outside vendors that meet the Company’s quality requirements. In addition, the Company outsources the manufacturing of certain LC instrument systems and components to well-established contract manufacturing firms in Singapore. The Company’s Singapore entity is ISO 9001:2015 certified and manages all Asian outsourced manufacturing as well as the distribution of all products from Asia. The Company may pursue outsourcing opportunities as they arise but believes it maintains adequate supply chain and manufacturing capabilities in the event of disruption or natural disasters.
The Company primarily manufactures and distributes its LC columns at its facilities in Taunton, Massachusetts and Wexford, Ireland. In February 2018, the Company’s Board of Directors approved expanding its Taunton location. The Company has incurred costs of approximately $248 million on a new state-of-the-art facility, which is substantially complete as of December 31, 2023. The Taunton facility processes, sizes and treats silica and polymeric media that are packed into columns, solid phase extraction cartridges and bulk shipping containers in both Taunton and Wexford. The Wexford facility also manufactures and distributes certain data, instruments and software components for the Company’s LC, MS and TA product lines. The Company’s Taunton facility is certified to ISO 9001:2015 and ISO 14001:2015. The Wexford facility is certified to ISO 9001:2015, ISO 13485:2016/EN ISO 13485:2016 and ISO 14001:2015. VICAM manufactures antibody-linked resins and magnetic beads that are packed into columns and kits in Milford, Massachusetts and Nixa, Missouri. The Company manufactures and distributes its Analytical Standards and Reagents and Environmental Resource Associates (“ERA”) product lines at its facility in Golden, Colorado, which is certified to ISO 9001:2015 and accredited to ISO/IEC 17025:2017, ISO/IEC 17034:16, ISO/IEC 17043:2010 and TNI Standard Vol. 3:2016. Some ERA products are also manufactured in the Wexford, Ireland facility, which is also accredited to ISO/IEC 17025:2017 and ISO/IEC 17034:2016.
The Company manufactures and distributes its MS products at its facilities in Wilmslow, England and Wexford, Ireland. Certain components or modules of the Company’s MS instruments are manufactured at its facility in Solihull, England and by long-standing outside contractors. Each stage of this supply chain is closely monitored by the Company to maintain high quality and performance standards. The instruments, components or modules are then returned to the Company’s facilities, where its engineers perform final assembly, calibrations to customer specifications and quality control procedures. The Company’s MS facilities are certified to ISO 9001:2015, ISO 13485:2016/EN ISO 13485:2016 and ISO 14001:2015 (Wexford only) and adhere to applicable regulatory requirements (including the FDA Quality System Regulation and the European In-Vitro Diagnostic Directive).
TA’s thermal analysis, rheometry and calorimetry products are manufactured and distributed at the Company’s New Castle, Delaware, Eden Prairie, Minnesota, Lindon, Utah and Hüllhorst, Germany facilities. Similar to MS, elements of TA’s products are manufactured by outside contractors and are then returned to the Company’s facilities for final assembly, calibration and quality control. The Company’s New Castle facility is certified to the ISO 9001:2015 standard and the Eden Prairie facility is certified to both ISO 9001:2015 and ISO/IEC 17025:2017 standards, and the Lindon facility is certified to ISO 9001:2015.
All instrument manufacturing for Wyatt products takes place at its facilities in Santa Barbara, California. The Company’s Wyatt facility in Santa Barbara, California is certified to ISO 9001:2015
Raw Materials
The Company purchases a variety of raw materials, primarily consisting of high temperature alloy sheet metal and castings, forgings, pre-plated metals and electrical components from various vendors. The materials used by
10
Table of Contents
the Company’s operations are generally available from a number of sources and in sufficient quantities to meet current requirements subject to normal lead times.
The Company is subject to rules of the Securities and Exchange Commission (“SEC”) under the Dodd-Frank Wall Street Reform and Consumer Protection Act, which require disclosure as to whether certain materials (tantalum, tin, gold and tungsten), known as conflict minerals, which may be contained in the Company’s products, are mined from the Democratic Republic of the Congo and adjoining countries. In 2022, the Company was not able to determine with certainty the country of origin of some of the conflict minerals in its manufactured products. However, the Company does not have knowledge that any of its conflict minerals originated from the Democratic Republic of the Congo or adjoining countries. The Company is in the process of evaluating its 2023 supply chain, and the Company plans to file its 2023 Form SD with the SEC in May 2024. The results of this and future evaluations may impose additional costs and may introduce new risks related to the Company’s ability to verify the origin of any conflict minerals contained in its products.
In addition, the Company continues to monitor environmental, health and safety regulations in countries in which it operates throughout the world, in particular, European Union and China Restrictions on the use of certain Hazardous Substances in electrical and electronic equipment and European Union Waste Electrical and Electronic Equipment directives. Further information regarding these regulations is available on the Company’s website, www.waters.com, under the caption “About Waters / Corporate Governance”.
Research and Development
The Company maintains an active research and development program focused on the development and commercialization of products that extend, complement and update its existing product offering. The Company’s research and development expenditures for 2023, 2022 and 2021 were $175 million, $176 million and $168 million, respectively.
Nearly all of the Company’s LC products have been developed at the Company’s main research and development center located in Milford, Massachusetts, with input and feedback from the Company’s extensive field organizations and customers. The majority of the Company’s MS products are developed at facilities in England and most of the Company’s current materials characterization products are developed at the Company’s research and development center in New Castle, Delaware. At December 31, 2023, 2022 and 2021, there were approximately 1,200 employees involved in the Company’s research and development efforts. The Company has increased research and development expenses from its continued commitment to invest significantly in new product development and existing product enhancements, and as a result of acquisitions. Despite the Company’s active research and development programs, there can be no assurance that the Company’s product development and commercialization efforts will be successful or that the products developed by the Company will be accepted by the marketplace.
In 2020, the Company opened a new research laboratory in Cambridge, MA, which serves as a strategic, collaborative space in the community, where Waters can partner with academia, research and industry to accelerate the next generation of scientific advancements.
Human Capital
We believe that our people differentiate our business and are vital to our continued success. As a result, we have made important investments in our workforce through initiatives and programs that support talent development and inclusion and enhance our Total Rewards programs.
Employees
The Company employed approximately 7,900, 8,200 and 7,800 employees at December 31, 2023, 2022 and 2021, respectively, with approximately 38% of the Company’s employees located in the United States. The
11
Table of Contents
Company believes its employee relations are generally good. The Company’s employees are not unionized or affiliated with any internal or external labor organizations.
In July 2023, the Company made organizational changes to better align its resources with its growth and innovation strategies, resulting in a worldwide workforce reduction that impacted approximately 5% of the Company’s employees.
Talent Development
We believe that our future success depends in a significant part on our continued ability to attract and retain highly skilled employees and then contribute to the growth and development of these employees.
We further the growth and development of our employees by investing in various programs, digital platforms and workshops that build professional and technical skills. In addition, management periodically assesses succession planning for certain key positions and reviews our workforce to identify high potential employees for future growth and development.
Inclusion & Diversity
We believe inclusion is a core tenet of organizational success and that fostering a sense of inclusivity allows our employees to maximize their performance contribution to our business. In 2021, we hired our first Director of Diversity, Equity and Inclusion to help strategize and focus our inclusivity efforts. As part of our company-led initiatives to drive an inclusive workplace, we have created Employee Circles and Employee Hubs, which are voluntary, employee-driven employee resource groups worldwide to foster a diverse and inclusive culture through awareness, education and employee connections. Our five Employee Circles are people of color, LGBTQ+ pride, veterans, women and disability. Our global Employee Circles provide opportunities for Waters employees to connect with teammates throughout the world, while Employee Hubs encourage engagement at the local level. Our Employee Circles and Hubs led various initiatives in 2023 to build a strong culture of inclusion and awareness. We have also rolled out training to all employees to support an inclusive culture that values diverse perspectives.
We believe that part of fostering a diverse, inclusive and equitable organization and workforce means understanding the makeup of our current employees. As of December 31, 2023, our workforce identifies as:
| • | 32% female, with women occupying 34% of company leadership roles (defined as Senior Director or above) compared with 22% in 2017, an increase of 12 percentage points; and |
| • | 23% racially and/or ethnically diverse, with 11% of our workforce identifying as Asian, 3.5% as identifying as Black or African American, 7% identifying as Hispanic/Latinx and 1% identifying as two or more races. |
Recruitment
Waters has focused on expanding diversity in our recruitment processes, including developing partnerships with organizations that support diversity in hiring and employee engagement. Current partnerships include: the National Organization of Black Chemists and Chemical Engineers, Out in Tech, Vercida, Home Base and Fairygodboss to expand the pipeline of strong candidates.
Health and Safety
The health and safety of our employees is our highest priority. Through online and in-person training programs, we believe that we foster a safe workplace and ensure that all employees are empowered to prevent accidents and injuries.
12
Table of Contents
We manufacture products deemed essential to critical infrastructure, including health and safety, food and agriculture, and energy, and as a result, the majority of our production sites continued operating during the COVID-19 pandemic.
Competition
The analytical instrument systems, supplies and services market is highly competitive. The Company encounters competition from several worldwide suppliers and other companies in both domestic and foreign markets for each of its three primary technologies. The Company competes in its markets primarily on the basis of product performance, reliability, service and, to a lesser extent, price. Competitors continuously introduce new products and have instrument businesses that are generally more diversified than the Company’s business. Some competitors have greater financial resources and broader distribution than the Company’s.
In the markets served by Waters, the Company’s principal competitors include: Agilent Technologies, Inc., Shimadzu Corporation, Bruker Corporation, Danaher Corporation and Thermo Fisher Scientific Inc. In the markets served by TA, the Company’s principal competitors include: Perkin Elmer, Inc., NETZSCH-Geraetebau GmbH, Thermo Fisher Scientific Inc., Malvern PANalytical Ltd., a subsidiary of Spectris plc, Anton-Paar GmbH and others not identified here.
The market for consumable LC products, including separation columns, is highly competitive and generally more fragmented than the analytical instruments market. The Company encounters competition in the consumable columns market from chemical companies that produce column sorbents and small specialized companies that primarily pack purchased sorbents into columns and subsequently package and distribute columns. The Company believes that it is one of the few suppliers that processes silica and polymeric resins, packs columns and distributes its own products. The Company competes in this market on the basis of performance, reproducibility, reputation and, to a lesser extent, price. In recent years, the Company’s principal competitors for consumable products have included: Danaher Corporation; Merck KGaA; Agilent Technologies, Inc.; General Electric Company and Thermo Fisher Scientific Inc. The ACQUITY UPLC Instrument is designed to offer a predictable level of performance when used with ACQUITY UPLC Columns and the Company believes that the expansion of the ACQUITY UPLC Instrument base will enhance its chromatographic column business because of the high level of synergy between ACQUITY UPLC Columns and the ACQUITY UPLC Instruments.
Patents, Trademarks and Licenses
The Company owns a number of United States and foreign patents and has patent applications pending in the United States and abroad. Certain technology and software has been acquired or is licensed from third parties. The Company also owns a number of trademarks. The Company’s patents, trademarks and licenses are viewed as valuable assets to its operations. However, the Company believes that no one patent or group of patents, trademark or license is, in and of itself, essential to the Company such that its loss would materially affect the Company’s business as a whole.
Environmental Matters and Climate Change
The Company is subject to foreign and U.S. federal, state and local laws, regulations and ordinances that (i) govern activities or operations that may have adverse environmental effects, such as discharges to air and water as well as handling and disposal practices for solid and hazardous wastes, and (ii) impose liability for the costs of cleaning up and certain damages resulting from sites of past spills, disposals or other releases of hazardous substances. The Company believes that it currently conducts its operations and has operated its business in the past in substantial compliance with applicable environmental laws. From time to time, Company operations have resulted or may result in noncompliance with environmental laws or liability for cleanup pursuant to environmental laws. The Company does not currently anticipate any material adverse effect on its
13
Table of Contents
operations, financial condition or competitive position as a result of its efforts to comply with environmental laws.
The Company is sensitive to the growing global debate with respect to climate change. An internal sustainability working group develops increasingly robust data with respect to the Company’s utilization of carbon producing substances in an effort to continuously reduce the Company’s carbon footprint. In 2019, the Company published a sustainability report identifying the various actions and behaviors the Company adopted in 2018 concerning its commitment to both the environment and the broader topic of social responsibility. The Company has continued to publish a sustainability report (which was renamed the ESG Report in 2022) on an annual basis. In November 2023, the Company published its 2023 ESG Report, detailing the Company’s efforts to address its environmental impact and uphold its social responsibilities in 2023. See Item 1A, Risk Factors – The effects of climate change could harm the Company’s business, for more information on the potential significance of climate change legislation. See also Note 18 in the Notes to the Consolidated Financial Statements for financial information about geographic areas.
Available Information
The Company files or furnishes all required reports with the SEC. The Company is an electronic filer and the SEC maintains a website that contains reports, proxy and information statements and other information regarding issuers that file electronically with the SEC. The address of the SEC electronic filing website is http://www.sec.gov. The Company also makes available, free of charge on its website, its annual report on Form 10-K, quarterly reports on Form 10-Q, current reports on Form 8-K and any amendments to those reports as soon as reasonably practicable after such material is electronically filed with or furnished to the SEC. The website address for Waters Corporation is http://www.waters.com and SEC filings can be found under the caption “Investors”. The Company is providing its website address solely for the information of investors. The Company does not intend the address to be an active link or to otherwise incorporate the contents of the website, including any reports that are noted in this annual report on Form 10-K (this “Annual Report”) as being posted on the website, into this Annual Report. Investors and others should note that we may announce material information to our investors using our investor relations website (ir.waters.com), SEC filings, press releases, public conference calls and webcasts. We use these channels, as well as social media, to communicate with our investors and the public about our Company, our business and other issues. It is possible that the information that we post on these channels could be deemed to be material information. We therefore encourage investors to visit these websites from time to time.
Forward-Looking Statements
This Annual Report, including the information incorporated by reference herein, contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended (the “Securities Act”), and Section 21E of the Securities Exchange Act of 1934, as amended (the “Exchange Act”). Statements that are not statements of historical fact may be deemed forward-looking statements. You can identify these forward-looking statements by the use of the words “feels”, “believes”, “anticipates”, “plans”, “expects”, “may”, “will”, “would”, “intends”, “suggests”, “appears”, “estimates”, “projects”, “should” and similar expressions, whether in the negative or affirmative. These forward-looking statements are subject to various risks and uncertainties, many of which are outside the control of the Company, including, and without limitation:
| • | foreign currency exchange rate fluctuations potentially affecting translation of the Company’s future non-U.S. operating results, particularly when a foreign currency weakens against the U.S. dollar; |
| • | current global economic, sovereign and political conditions and uncertainties, including the effect of new or proposed tariff or trade regulations, changes in inflation and interest rates, the impacts and costs of war, in particular as a result of the ongoing conflicts between Russia and Ukraine and in the Middle East, and the possibility of further escalation resulting in new geopolitical and regulatory instability and the Chinese government’s ongoing tightening of restrictions on procurement by government-funded customers; |
14
Table of Contents
| • | the Company’s ability to access capital, maintain liquidity and service the Company’s debt in volatile market conditions; |
| • | risks related to the effects of any pandemic on our business, financial condition, results of operations and prospects; |
| • | changes in timing and demand for the Company’s products among the Company’s customers and various market sectors, particularly as a result of fluctuations in their expenditures or ability to obtain funding; |
| • | the ability to realize the expected benefits related to the Company’s various cost-saving initiatives, including workforce reductions and organizational restructurings; |
| • | the introduction of competing products by other companies and loss of market share, as well as pressures on prices from competitors and/or customers; |
| • | changes in the competitive landscape as a result of changes in ownership, mergers and continued consolidation among the Company’s competitors; |
| • | regulatory, economic and competitive obstacles to new product introductions, lack of acceptance of new products and inability to grow organically through innovation; |
| • | rapidly changing technology and product obsolescence; |
| • | risks associated with previous or future acquisitions, strategic investments, joint ventures and divestitures, including risks associated with achieving the anticipated financial results and operational synergies; contingent purchase price payments; and expansion of our business into new or developing markets; |
| • | risks associated with unexpected disruptions in operations; |
| • | failure to adequately protect the Company’s intellectual property, infringement of intellectual property rights of third parties and inability to obtain licenses on commercially reasonable terms; |
| • | the Company’s ability to acquire adequate sources of supply and its reliance on outside contractors for certain components and modules, as well as disruptions to its supply chain; |
| • | risks associated with third-party sales intermediaries and resellers; |
| • | the impact and costs of changes in statutory or contractual tax rates in jurisdictions in which the Company operates as well as shifts in taxable income among jurisdictions with different effective tax rates, the outcome of ongoing and future tax examinations and changes in legislation affecting the Company’s effective tax rate; |
| • | the Company’s ability to attract and retain qualified employees and management personnel; |
| • | risks associated with cybersecurity and technology, including attempts by third parties to defeat the security measures of the Company and its third-party partners; |
| • | increased regulatory burdens as the Company’s business evolves, especially with respect to the U.S. Food and Drug Administration and U.S. Environmental Protection Agency, among others, and in connection with government contracts; |
| • | regulatory, environmental and logistical obstacles affecting the distribution of the Company’s products, completion of purchase order documentation and the ability of customers to obtain letters of credit or other financing alternatives; |
| • | risks associated with litigation and other legal and regulatory proceedings; and |
| • | the impact and costs incurred from changes in accounting principles and practices. |
Certain of these and other factors are further described below in Item 1A, Risk Factors, of this Annual Report. Actual results or events could differ materially from the plans, intentions and expectations disclosed in
15
Table of Contents
the forward-looking statements, whether because of these factors or for other reasons. All forward-looking statements speak only as of the date of this Annual Report, and forward-looking statements in documents that are incorporated by reference hereto speak only as of the date of those documents. Such forward-looking statements are expressly qualified in their entirety by the cautionary statements included in this report. Except as required by law, the Company does not assume any obligation to update any forward-looking statements.
Item 1A: Risk Factors
The Company is subject to risks and uncertainties, including, but not limited to, the following:
RISKS RELATED TO MACROECONOMIC CONDITIONS
The Company’s international operations may be negatively affected by political events, wars or terrorism, economic conditions and regulatory changes, related to either a specific country or a larger region. These potential political, currency and economic disruptions, as well as foreign currency exchange rate fluctuations, could have a material adverse effect on the Company’s results of operations or financial condition.
Approximately 69% and 70% of the Company’s net sales in 2023 and 2022, respectively, were outside of the United States and were primarily denominated in foreign currencies. In addition, the Company has considerable manufacturing operations in Ireland and the U.K., as well as significant subcontractors located in Singapore. As a result, a significant portion of the Company’s sales and operations are subject to certain risks, including adverse developments in the political, regulatory and economic environment, in particular, uncertainty regarding possible changes to foreign and domestic trade policy; the effect of the U.K.’s exit from the European Union as well as the financial difficulties and debt burden experienced by a number of European countries; impact and costs of terrorism or war, in particular as a result of the ongoing conflict between Russia and Ukraine and in the Middle East, and the possibility of further escalation resulting in new geopolitical and regulatory instability; the instability and possible dissolution of the euro as a single currency; sudden movements in a country’s foreign exchange rates due to a change in a country’s sovereign risk profile or foreign exchange regulatory practices; trade protection measures including embargoes, sanctions and tariffs; differing tax laws and changes in those laws; restrictions on investments and/or limitations regarding foreign ownership; nationalization of private enterprises which may result in the confiscation of assets; credit risk and uncertainties regarding the collectability of accounts receivable; the impact of global health pandemics and epidemics, such as COVID-19; changes in inflation and interest rates; instability in the global banking industry; rising energy prices and potential energy shortages; difficulties in protecting intellectual property; difficulties in staffing and managing foreign operations; and associated adverse operational, contractual and tax consequences.
In 2023, the Company generated $441 million of total net sales from China, down from $565 million in 2022. This significant 22% reduction in sales from China resulted from lower customer demand for our products across all customer classes, driven by various factors. Such factors include a decline in the economic conditions in China, trade tensions and tariffs between the U.S. and China and their impact on our customers’ purchasing decisions, increased competition from local and international competitors in China, the Chinese government’s ongoing tightening of restrictions on procurement by government-funded customers and other regulatory and compliance challenges and uncertainties in the Chinese market, all of which had, and may continue to have, an adverse effect on our business and operations in China.
In particular, China’s government continues to play a significant role in regulating industry development by imposing sector-specific policies, and it maintains control over China’s economic growth through setting monetary policy and determining treatment of particular industries or companies. The U.S. government has called for substantial changes to foreign trade policy with China and has recently raised, and has proposed to further raise in the future, tariffs on several Chinese goods. China has retaliated with increased tariffs on U.S. goods, which may increase our cost of doing business in China. Any further changes in U.S. trade policy could trigger retaliatory actions by affected countries, including China, resulting in trade wars and increased costs for
16
Table of Contents
goods imported into the U.S. and impacting our ability to sell our products in China and other affected countries. Accordingly, our financial position or results of operations can be adversely influenced by political, economic, legal, compliance, social and business conditions in China generally.
Additionally, the U.S. dollar value of the Company’s net sales, cost of sales, operating expenses, interest, taxes and net income varies with foreign currency exchange rate fluctuations. Significant increases or decreases in the value of the U.S. dollar relative to certain foreign currencies, particularly the euro, Japanese yen, British pound and Chinese renminbi, could have a material adverse effect or benefit on the Company’s results of operations or financial condition.
From time to time, the Company enters into certain foreign currency exchange contracts that are intended to offset some of the market risk associated with sales denominated in foreign currencies. We cannot predict the effectiveness of these transactions or their impact upon our future operating results, and from time to time they may negatively affect our quarterly earnings.
Global economic conditions may have an adverse effect on the demand for, and supply of, the Company’s products and harm the Company’s financial results.
The Company is a global business that may be adversely affected by changes in global economic conditions such as changes in the rate of inflation (including the cost of raw materials, commodities and supplies) and interest rates. Both our domestic and international markets experience varying degrees of inflationary and interest rate pressures. These changes in global economic conditions may affect the demand for, and supply of, the Company’s products and services. This may result in a decline in sales in the future, increased rate of order cancellations or delays, increased risk of excess or obsolete inventories, longer sales cycles and potential difficulty in collecting sales proceeds. There can be no assurance regarding demand for the Company’s products and services in the future.
Disruption in worldwide financial markets could adversely impact the Company’s access to capital and financial condition.
Financial markets in the U.S., Europe and Asia have experienced times of extreme disruption, including, among other things, sharp increases in the cost of new capital, credit rating downgrades and bailouts, severely diminished capital availability and severely reduced liquidity in money markets. Financial and banking institutions have also experienced disruptions, resulting in large asset write-downs, higher costs of capital, rating downgrades and reduced desire to lend money. There can be no assurance that there will not be future deterioration or prolonged disruption in financial markets or financial institutions. Any future deterioration or prolonged disruption in financial markets or financial institutions in which the Company participates may impair the Company’s ability to access its existing cash, utilize its existing syndicated bank credit facility funded by such financial institutions or access sources of new capital, which it may need to meet its capital needs. The cost to the Company of any new capital raised and interest expense would increase if this were to occur.
Public health crises, epidemics or pandemics, such as the COVID-19 pandemic have had, and could in the future have, a negative impact on the Company’s business and operations.
Public health crises, epidemics or pandemics have had, and could in the future have, a negative impact on our business and operations, including Company sales and cash flow. Such public health crises, epidemics and pandemics have the potential to create significant volatility, uncertainty and worldwide economic disruption, resulting in an economic slowdown of potentially extended duration, as seen with the COVID-19 pandemic from 2020 to 2022. The Company’s global operations expose it to risks associated with such public health crises, epidemics and pandemics, which could have an adverse effect on its business, results of operations and financial condition. The degree to which such public health crisis, epidemics or pandemics ultimately affects the Company’s business, results of operations and financial condition is highly uncertain and cannot be predicted.
17
Table of Contents
RISKS RELATED TO OUR BUSINESS
The Company’s financial results are subject to changes in customer demand, which may decrease for a number of reasons, many beyond the Company’s control.
The demand for the Company’s products is dependent upon the size of the markets for its LC, LC-MS, light scattering, thermal analysis, rheometry and calorimetry products; the timing and level of capital spending and expenditures of the Company’s customers; changes in governmental regulations, particularly those affecting drug, food and drinking water testing; funding available to academic, governmental and research institutions; general economic conditions and the rate of economic growth in the Company’s major markets; and competitive considerations. The Company typically experiences seasonality in its orders that is reflected as an increase in sales in its fourth quarter as a result of purchasing habits for capital goods by customers that tend to exhaust their spending budgets by calendar year-end. However, there can be no assurance that the Company will effectively forecast customer demand and appropriately allocate research and development expenditures to products with high growth and high margin prospects. Additionally, there can be no assurance that the Company’s results of operations or financial condition will not be adversely impacted by a change in any of the factors listed above or the continuation of uncertain global economic conditions.
The analytical instrument market may also, from time to time, experience low sales growth. Approximately 57% and 59% of the Company’s net sales in 2023 and 2022, respectively, were to worldwide pharmaceutical accounts, which are periodically subject to unfavorable market conditions and consolidations. Unfavorable industry conditions could have a material adverse effect on the Company’s results of operations or financial condition.
Competitors may introduce more effective or less expensive products than the Company’s, which could result in decreased sales. The competitive landscape may transform as a result of potential changes in ownership, mergers and continued consolidations among the Company’s competitors, which could harm the Company’s business.
The analytical instrument market, and, in particular, the portion related to the Company’s HPLC, UPLC, LC-MS, light scattering, thermal analysis, rheometry and calorimetry product lines, is highly competitive. The Company encounters competition from several international instrument suppliers and other companies in both domestic and foreign markets. Some competitors have instrument businesses that are generally more diversified than the Company’s business, but are typically less focused on the Company’s chosen markets. Over the years, some competitors have merged with other competitors for various reasons, including increasing product line offerings, improving market share and reducing costs. There can be no assurance that the Company’s competitors will not introduce new, disruptive technologies that displace the Company’s existing technologies or more effective and less costly products than those of the Company or that the Company will be able to increase its sales and profitability from new product introductions. There can be no assurance that the Company’s sales and marketing forces will compete successfully against the Company’s competitors in the future.
Strategies for organic growth require developing new technologies and bringing these new technologies to market, which could negatively impact the Company’s financial results.
The Company’s corporate strategy is fundamentally based on winning through organic innovation and deep application expertise. The Company is in the process of developing new products with recently acquired technologies. The future development of these new products will require a significant amount of spending over the next few years before any significant, robust sales will be realized. Furthermore, these new products will be sold into both the non-clinical and clinical markets, and any new products requiring FDA clearance may take longer to bring to market. There can be no assurance given as to the timing of these new product launches and the ultimate realization of sales and profitability in the future.
In addition, the Company’s products are subject to rapid changes in technology. Rapidly changing technology could make some or all of our product lines obsolete unless the Company is able to continually improve our existing products and develop new products. If the Company fails to develop and introduce products
18
Table of Contents
in a timely manner in response to changing technology, market demands or the requirements of our customers, the Company’s product sales may decline, and we could experience an adverse effect on our results of operations or financial condition.
The Company may face risks associated with previous or future acquisitions, strategic investments, joint ventures and divestitures.
In the normal course of business, the Company may engage in discussions with third parties relating to possible acquisitions, strategic investments, joint ventures and divestitures. The Company may pursue transactions that complement or augment its existing products and services, such as the Wyatt acquisition that was completed in May 2023. Such transactions involve numerous risks, including difficulties in integrating the acquired operations, technologies and products; diversion of management’s attention from other business concerns; inability to predict financial results; potential departures of key employees of the acquired company; and difficulties in effectively transferring divested businesses and liabilities. If the Company successfully identifies acquisitions in the future, completing such acquisitions may result in new issuances of the Company’s stock that may be dilutive to current owners; increases in the Company’s debt and contingent liabilities; and additional amortization expense related to intangible assets. For example, the Company financed the Wyatt acquisition, in part, through borrowings under its revolving credit facility, resulting in a significant increase in the Company’s outstanding debt. Acquired businesses may also expose the Company to new risks and new markets, and the Company may have difficulty addressing these risks in a cost-effective and timely manner. Any of these transaction-related risks could have a material adverse effect on the Company’s profitability. In addition, the Company may not be able to identify, successfully complete, or integrate potential acquisitions in the future. Even if the Company can do so, it cannot be sure that these acquisitions will have a positive impact on the Company’s business or operating results.
The Company’s software or hardware may contain coding or manufacturing errors that could impact their function, performance and security, and result in other negative consequences.
Despite testing prior to the release and throughout the lifecycle of a product or service, the detection and correction of any errors in released software or hardware can be time consuming and costly. This could delay the development or release of new products or services, or new versions of products or services, create security vulnerabilities in the Company’s products or services, and adversely affect market acceptance of products or services. If the Company experiences errors or delays in releasing its software or hardware, or new versions thereof, its sales could be affected and revenues could decline. Errors in software or hardware could expose the Company to product liability, performance and warranty claims as well as harm to brand and reputation, which could impact future sales.
A successful product liability claim brought against the Company in excess of, or outside the coverage of, the Company’s insurance coverage could have a material adverse effect on our business, financial condition and results of operations. The Company may not be able to maintain product liability insurance on acceptable terms, if at all, and insurance may not provide adequate coverage against potential liabilities.
Disruption of operations at the Company’s manufacturing facilities could harm the Company’s financial condition.
The Company manufactures LC instruments at facilities in Milford, Massachusetts and through a subcontractor in Singapore; precision chemistry separation columns at its facilities in Taunton, Massachusetts and Wexford, Ireland; MS products at its facilities in Wilmslow, England, Solihull, England and Wexford, Ireland; thermal analysis and rheometry products at its facilities in New Castle, Delaware; and other instruments and consumables at various other locations as a result of the Company’s acquisitions. Any prolonged disruption to the operations at any of these facilities, whether due to labor difficulties, destruction of or damage to any facility, power interruptions, cybersecurity incidents, weather events or natural disasters (including the potential impacts of climate change) or other reasons, could harm our customer relationships, impede our ability to generate sales and have a material adverse effect on the Company’s results of operations or financial condition.
19
Table of Contents
Failure to adequately protect intellectual property could have materially adverse effects on the Company’s results of operations or financial condition.
Our success depends on our ability to obtain, maintain, and enforce patents on our technology, maintain our trademarks, and protect our trade secrets. There can be no assurance that any patents held by the Company will not be challenged, invalidated or circumvented or that the rights granted thereunder will provide competitive advantages to the Company. Additionally, there could be successful claims against the Company by third-party patent holders with respect to certain Company products that may infringe the intellectual property rights of such third parties. In the event that a claim relating to intellectual property is asserted against the Company, or third parties hold pending or issued patents that relate to the Company’s products or technology, the Company may seek licenses to such intellectual property or challenge those patents. However, the Company may be unable to obtain these licenses on commercially reasonable terms, if at all, and the challenge of the patents may be unsuccessful. The Company’s failure to obtain the necessary licenses or other rights could impact the sale, manufacture, or distribution of its products and, therefore, could have a material adverse effect on its results of operations and financial condition. The Company’s patents, including those licensed from others, expire on various dates.
The Company also depends in part on its trademarks and the strength of its proprietary brands, which the Company considers important to its business. The Company’s inability to protect or preserve the value of its intellectual property rights for any reason, including the Company’s inability to successfully defend against counterfeit, knock-offs, grey-market, infringing or otherwise unauthorized products, could damage the Company’s brand and reputation and harm its business.
The Company also relies on trade secrets and proprietary know-how with which it seeks to protect its products, in part, by confidentiality agreements with its collaborators, employees and consultants. These agreements may not adequately protect the Company’s trade secrets and other proprietary rights. These agreements may be breached, and the Company may not have adequate remedies for any breach. In addition, the Company’s trade secrets may otherwise become known or be independently developed by its competitors. If the Company is unable to protect its intellectual property rights, it could have an adverse and material effect on the Company’s results of operations or financial condition.
The Company’s business would suffer if the Company were unable to acquire adequate sources of supply.
Most of the raw materials, components and supplies purchased by the Company are available from a number of different suppliers; however, a number of items are purchased from limited or single sources of supply. Consolidation among such suppliers could also result in other limited or sole-source suppliers for the Company in the future. Disruption of these sources could have, at a minimum, a temporary adverse effect on shipments and the financial results of the Company. In addition, price increases from these suppliers could have an adverse effect on the Company’s margins. A prolonged inability to obtain certain materials or components could have an adverse effect on the Company’s financial condition or results of operations and could result in damage to its relationships with its customers and, accordingly, adversely affect the Company’s business.
The Company’s sales would deteriorate if the Company’s outside contractors fail to provide necessary components or modules.
Certain components or modules of the Company’s LC and MS instruments are manufactured by outside contractors, including the manufacturing of LC instrument systems and related components by contract manufacturing firms in Singapore. The ability of these contractors to perform their obligations is largely outside of the Company’s control. Failure by these outside contractors to perform their obligations in a timely manner or at satisfactory quality levels could have an adverse effect on the supply chain and the financial results of the Company. In addition, if one or more of such contractors experience significant disruption in services or institute a significant price increase, the Company may have to seek alternative providers, its costs could increase and the delivery of its products could be prevented or delayed. A prolonged inability to obtain these components or modules could have an adverse effect on the Company’s financial condition or results of operations.
20
Table of Contents
The Company’s business could be harmed by actions of third-party sales intermediaries and other third parties that sell our products.
The Company sells some products through third parties, including third-party sales intermediaries and value-added resellers. This exposes us to various risks, including competitive pressure, concentration of sales volumes, credit risks and compliance risks. We may rely on one or a few key third-party sales intermediaries for a product or market and the loss of these third-party sales intermediaries could reduce our revenue or net earnings. Third-party sales intermediaries may also face financial difficulties, including bankruptcy, which could harm our collection of accounts receivable. Moreover, violations of the U.S. Foreign Corrupt Practices Act (“FCPA”), the U.K. Bribery Act or similar anti-bribery laws by distributors or other third-party intermediaries could materially and adversely impact our business, reputation and results of operations. Risks related to our use of third-party sales intermediaries and other third parties may reduce sales, increase expenses and weaken our competitive position.
The Company is subject to laws and regulations governing government contracts, and failure to address these laws and regulations or comply with government contracts could harm its business by leading to a reduction in revenues associated with these customers.
The Company derives a portion of its revenue from direct and indirect sales to U.S. federal, state and local as well as foreign governments and their respective agencies, and, as a result, it is subject to various statutes and regulations that apply to companies doing business with the government. The laws governing government contracts differ from the laws governing private contracts and government contracts may contain pricing terms and conditions that are not applicable to private contracts. The Company is also subject to investigation for compliance with the regulations governing government contracts. A failure to comply with these regulations could result in suspension of these contracts, criminal, civil and administrative penalties or debarment, which could negatively impact the Company’s business and operations. If the Company’s government contracts are terminated, if it is suspended from government work or if its ability to compete for new contracts is adversely affected, the Company’s business could be negatively impacted.
The Company’s financial results are subject to unexpected shifts in pre-tax income between tax jurisdictions, changing application of tax law and tax audit examinations.
The Company is subject to rates of income tax that range from 0% up to 34% in various jurisdictions in which it conducts business. In addition, the Company typically generates a substantial portion of its income in the fourth quarter of each fiscal year. Geographical shifts in income from previous quarters’ projections caused by factors including, but not limited to, changes in volume and product mix and fluctuations in foreign currency translation rates, could therefore have potentially significant favorable or unfavorable effects on the Company’s income tax expense, effective tax rate and results of operations.
Governments in the jurisdictions in which the Company operates implement changes to tax laws and regulations from time to time. From 2024, various foreign jurisdictions are beginning to implement aspects of the guidance issued by the Organization for Economic Co-operation and Development related to the new Pillar Two system of global minimum tax rules. These new tax laws and regulations, and any changes in corporate income tax rates or regulations regarding transfer pricing or repatriation of dividends or capital, as well as changes in the interpretation of existing tax laws and regulations, could adversely affect the Company’s cash flow and lead to increases in its overall tax burden, which would negatively affect the Company’s profitability. The Company continues to monitor the adoption of the Pillar Two rules in additional jurisdictions.
The Company entered into a new Development and Expansion Incentive in Singapore that provides a concessionary income tax rate of 5% on certain types of income for the period April 1, 2021 through March 31, 2026. Prior to April 1, 2021, the Company had a tax exemption in Singapore on certain types of income, based upon the achievement and continued satisfaction of certain operational and financial milestones, which the Company met as of December 31, 2020 and maintained through March 2021. The Company had determined that it was more likely than not to realize the tax exemption in Singapore and, accordingly, did not recognize any reserves for unrecognized tax benefits on its balance sheet related to this tax exemption. If any of the milestone targets were not met, the Company would not have been entitled to the tax exemption on income earned in Singapore dating back to the start date of the agreement (April 1, 2016), and all the tax benefits previously
21
Table of Contents
recognized would be reversed, resulting in the recognition of income tax expense equal to the statutory tax of 17% on income earned during that period.
As a global business, the Company is subject to tax audit examinations in various jurisdictions throughout the world. The Company must manage the cost and disruption of responding to governmental audits, investigations and proceedings. In addition, the impact of the settlement of pending or future tax audit examination could have an unfavorable effect on the Company’s income tax expense, effective tax rate and results of operations.
The Company may be required to recognize impairment charges for our goodwill and other intangible assets.
As of December 31, 2023, the net carrying value of the Company’s goodwill and other intangible assets totaled approximately $1.9 billion. The Wyatt acquisition significantly increased the carrying value of the Company’s goodwill and other intangible assets, which could lead to potential impairments if Wyatt’s financial results are significantly less than anticipated in the future. In accordance with generally accepted accounting principles, the Company periodically assesses these assets to determine if they are impaired. Significant negative industry or economic trends, disruptions to the Company’s business, inability to effectively integrate acquired businesses, unexpected significant changes or planned changes in use of our assets, changes in the structure of our business, divestitures, market capitalization declines or increases in associated discount rates can impair the Company’s goodwill and other intangible assets. Any charges relating to such impairments adversely affect the Company’s financial statements in the periods recognized.
RISKS RELATED TO HUMAN CAPITAL MANAGEMENT
We may not be able to attract and retain qualified employees.
Our future success depends upon the continued service of our executive officers and other key management and technical personnel, and on our ability to continue to identify, attract, retain and motivate them. Implementing our business strategy requires specialized engineering and other talent, as our revenues are highly dependent on technological and product innovations. The market for employees in our industry is extremely competitive, and competitors for talent, particularly engineering talent, increasingly attempt to hire, and to varying degrees have been successful in hiring, our employees. A number of such competitors for talent are significantly larger than us and are able to offer compensation in excess of what we are able to offer. Additionally, macroeconomic conditions, including wage inflation, could have a material impact on our ability to attract and retain talent, our turnover rate and the cost of operating our business. In July 2023, the Company made organizational changes to better align its resources with its growth and innovation strategies, resulting in a worldwide workforce reduction that impacted approximately 5% of the Company’s employees. These workforce reductions may not have the desired impact on our cost-saving initiatives, as they could adversely affect our productivity, morale, customer relationships, product quality, innovation capabilities and ability to execute our strategic plans. Moreover, these workforce reductions could expose us to potential litigation, severance costs, reputational damage and loss of key personnel. If we are unable to manage the effects of these workforce reductions or achieve the expected benefits from them, our business, financial condition and results of operations could be materially and adversely affected. Further, existing immigration laws make it more difficult for us to recruit and retain highly skilled foreign national graduates of universities in the United States, making the pool of available talent even smaller. If we are unable to attract and retain qualified employees, our business may be harmed.
The loss of key members of management and the risks inherent in succession planning could adversely affect the Company’s results of operations or financial condition.
The operation of the Company requires managerial and operational expertise. None of the Company’s key management employees, with the exception of the Chief Executive Officer and Chief Financial Officer, have an employment contract with the Company and there can be no assurance that such individuals will remain with the Company. If, for any reason, other key personnel do not continue to be active in management, the Company’s results of operations or financial condition could be adversely affected. The Company’s success also depends on its ability to execute leadership succession plans. The inability to successfully transition key management roles could have a material adverse effect on the Company’s operating results.
22
Table of Contents
RISKS RELATED TO CYBERSECURITY
Disruption, cyber-attack or unforeseen problems with the security, maintenance or upgrade of the Company’s information and web-based systems could have an adverse effect on the Company’s business strategy, results of operations and financial condition.
The Company relies on its technology infrastructure and that of its third-party partners, including its software and banking partners, among other functions, to interact with suppliers, sell products and services, fulfill contract obligations, ship products, collect and make electronic wire and check based payments and otherwise conduct business. The Company’s technology infrastructure and that of its third-party partners has been, and may in the future be, vulnerable to damage or interruption from, but not limited to, natural disasters, power loss, telecommunication failures, terrorist attacks, computer viruses, ransomware, unauthorized access to customer or employee data, unauthorized access to and funds transfers from Company bank accounts and other attempts to harm the Company’s systems. In the event of such an incident, the Company has in the past, and may in the future, suffer interruptions in service, loss of assets or data or reduced functionality. The Company attempts to mitigate cybersecurity risks by employing a number of proactive measures, including mandatory ongoing employee training and awareness, technical security controls, enhanced data protection and maintenance of backup and protective systems. Despite these mitigation measures, the Company’s systems and those of its partners remain potentially vulnerable to cybersecurity threats, any of which could have a material adverse effect on the Company’s business. To date, cybersecurity incidents have not resulted in a material adverse impact to the Company’s business strategy, results of operations, or financial condition, but future incidents could have such an impact. Additionally, the Company must maintain and periodically upgrade its information and web-based systems, which has caused and will in the future cause temporary interruptions to its technology infrastructure. Any prolonged disruption to the Company’s technology infrastructure, at any of its facilities, could have a material adverse effect on the Company’s business strategy, results of operations or financial condition. While the Company maintains cyber insurance, this insurance may not, however, be sufficient to cover the financial, legal, business or reputational losses that may result from an interruption or breach of its systems.
If the Company’s security measures are compromised or fail to adequately protect its technology infrastructure, research and development efforts or manufacturing operations, the Company’s products and services may be perceived as vulnerable or unreliable, the information protected by the Company’s controls and processes may be subject to unauthorized access, acquisition or modification, the Company’s brand and reputation could be damaged, the services that the Company provides to its customers could be disrupted, and customers may stop using the Company’s products and services, all of which could reduce the Company’s revenue and earnings, increase its expenses and expose it to legal claims and regulatory actions.
The Company is in the business of designing, manufacturing, selling and servicing analytical instruments to life science, pharmaceutical, biochemical, industrial, nutritional safety and environmental, academic and governmental customers working in research and development, quality assurance and other laboratory applications, and the Company is also a developer and supplier of software and software-based products that support instrument systems. Many of the Company’s customers are in highly regulated industries. While the Company has invested time and resources implementing measures designed to protect the integrity and security of its technology infrastructure, research and development processes, manufacturing operations, products and services, and the internal and external data managed by the Company, there is a risk these measures will be defeated or compromised or that they are otherwise insufficient to protect against existing or emerging threats. The Company also has acquired companies, products, services and technologies over time and may face inherent risk when integrating these acquisitions into the Company. In addition, at times, the Company faces attempts by third parties to defeat its security measures or exploit vulnerabilities in its systems. These risks will increase as the Company continues to grow and expand geographically, and its systems, products and services become increasingly digital and sensor-and web-based.
The Company could suffer significant damage to its brand and reputation if a security incident resulted in unauthorized access to, acquisition of, or modification to the Company’s technology infrastructure, research and development processes, manufacturing operations, its products and services as well as the internal and external
23
Table of Contents
data managed by the Company. Such an incident could disrupt the Company’s operations and customers could lose confidence in the Company’s ability to deliver quality and reliable products or services. This could negatively impact sales and could increase costs related to fixing and addressing these incidents and any vulnerabilities exposed by them, as well as to lawsuits, regulatory investigations, claims or legal liability including contractual liability, costs and expenses owed to customers and business partners.
RISKS RELATED TO COMPLIANCE, REGULATORY OR LEGAL MATTERS
Changes in governmental regulations and compliance failures could harm the Company’s business.
The Company is subject to regulation by various federal, state and foreign governments and agencies in areas including, among others, health and safety, import/export, privacy and data protection, FCPA and environmental laws and regulations. A portion of the Company’s operations are subject to regulation by the FDA and similar foreign regulatory agencies. These regulations are complex, can change frequently and govern an array of product activities, including design, development, labeling, manufacturing, promotion, sales and distribution. Any failure by the Company to comply with applicable governmental regulations could result in product recalls, the imposition of fines, restrictions on the Company’s ability to conduct or expand its operations or the cessation of all or a portion of its operations. Additionally, the Company develops, configures and markets its products and services to meet customer needs created by these regulations, and any significant change in regulations could reduce demand for its products, increase its expenses or otherwise materially impact its financial position and results of operations.
Regulators globally are increasingly imposing greater fines and penalties for privacy and data protection violations, and the European Union, as an example, has enacted a broad data protection regulation with fines based on a percentage of global revenues. Changes in laws or regulations associated with enhanced protection of certain sensitive types of personal information, such as information related to health, could greatly increase the cost of compliance and the cost of providing the Company’s products or services. Any failure, or perceived failure, by the Company to comply with laws and regulations on privacy, data security or consumer protection, or other policies, public perception, standards, self-regulatory requirements or legal obligations, could result in lost or restricted business, proceedings, actions or fines brought against the Company or levied by governmental entities or others, or could otherwise adversely affect the business and harm the Company’s reputation.
Some of the Company’s operations are subject to domestic and international laws and regulations with respect to the manufacturing, handling, use or sale of toxic or hazardous substances. This requires the Company to devote substantial resources to maintain compliance with those applicable laws and regulations. If the Company fails to comply with such requirements in the manufacturing or distribution of its products, it could face civil and/or criminal penalties and potentially be prohibited from distributing or selling such products until they are compliant.
Some of the Company’s products are also subject to the rules of certain industrial standards bodies, such as the International Standards Organization. The Company must comply with these rules, as well as those of other agencies, such as the United States Occupational Safety and Health Administration. Failure to comply with such rules could result in the loss of certification and/or the imposition of fines and penalties, which could have a material adverse effect on the Company’s operations.
As a publicly-traded company, the Company is subject to the rules of the SEC and the New York Stock Exchange. In addition, the Company must comply with the Sarbanes-Oxley regulations, which require the Company to establish and maintain adequate internal control over financial reporting. The Company’s efforts to comply with such laws and regulations are time consuming and costly. While we continue to enhance our controls, we cannot be certain that we will be able to prevent future significant deficiencies or material weaknesses. Failure to comply with such regulations or having inadequate internal controls could have a material adverse effect on the Company’s financial condition and operations, which could cause investors to lose confidence in our reported financial information and could have a negative effect on the trading price of our stock and our access to capital.
24
Table of Contents
The Company is subject to the rules of the SEC under the Dodd-Frank Wall Street Reform and Consumer Protection Act, which require disclosure as to whether certain materials (tantalum, tin, gold and tungsten), known as conflict minerals, which may be contained in the Company’s products, are mined from the Democratic Republic of the Congo and adjoining countries. In 2022, the Company was not able to determine with certainty the country of origin of some of the conflict minerals in its manufactured products. However, the Company does not have knowledge that any of its conflict minerals originated from the Democratic Republic of the Congo or adjoining countries. The Company is in the process of evaluating its 2023 supply chain, and the Company plans to file its 2023 Form SD with the SEC in May 2024. The results of this and future evaluations may impose additional costs and may introduce new risks related to the Company’s ability to verify the origin of any conflict minerals contained in its products.
The Company may be harmed by improper conduct of any of our employees, agents or business partners.
We cannot provide assurance that our internal controls and compliance systems will always protect the Company from acts committed by employees, agents or business partners that would violate domestic and international laws, including laws governing payments to government officials, bribery, fraud, kickbacks and false claims, pricing, sales and marketing practices, conflicts of interest, competition, export and import compliance, money laundering and data privacy. In particular, the FCPA, the U.K. Bribery Act and similar anti-bribery laws generally prohibit companies and their intermediaries from making improper payments to government officials for the purpose of obtaining or retaining business, and we operate in many parts of the world that have experienced governmental corruption to some degree. Any such improper actions or allegations of such acts could damage our reputation and subject us to civil or criminal investigations in the U.S. and in other jurisdictions and related shareholder lawsuits, could lead to substantial civil and criminal, monetary and non-monetary penalties and could cause us to incur significant legal and investigatory fees. In addition, the government may seek to hold us liable as a successor for violations committed by companies in which we invest or that we acquire. We also rely on our suppliers to adhere to our supplier standards of conduct and material violations of such standards of conduct could occur that could have a material effect on our business, reputation and financial statements. In addition, any allegations of issues resulting from the misuse of our products could, even if untrue, adversely affect our reputation and our customers’ willingness to purchase products from us. Any such allegations could cause us to lose customers and divert our resources from other tasks, which could materially and adversely affect our business and operating results.
Environmental, social and corporate governance (“ESG”) issues, including those related to climate change and sustainability, may have an adverse effect on our business, financial condition and results of operations and damage our reputation.
There is an increasing focus from certain investors, customers, consumers, employees and other stakeholders concerning ESG matters. Additionally, public interest and legislative pressure related to public companies’ ESG practices continue to grow. If our ESG practices fail to meet regulatory requirements or investor, customer, consumer, employee or other stakeholders’ evolving expectations and standards for responsible corporate citizenship in areas including environmental stewardship and sustainability, support for local communities, director and employee diversity, human capital management, employee health and safety practices, product quality, supply chain management, corporate governance and transparency, our reputation, brand and employee retention may be negatively impacted, and our customers and suppliers may be unwilling to continue to conduct business with us.
Customers, consumers, investors and other stakeholders are increasingly focusing on environmental issues, including climate change, energy and water use, plastic waste and other sustainability concerns. Concern over climate change or plastics and packaging materials, in particular, may result in new or increased legal and regulatory requirements to reduce or mitigate impacts to the environment. Changing customer and consumer preferences or increased regulatory requirements may result in increased demands or requirements regarding plastics and packaging materials, including single-use and non-recyclable plastic products and packaging, other components of our products and their environmental impact on sustainability, or increased customer and
25
Table of Contents
consumer concerns or perceptions (whether accurate or inaccurate) regarding the effects of substances present in certain of our products. Complying with these demands or requirements could cause us and companies in our supply chain to incur additional manufacturing, operating or product development costs.
If we do not adapt to or comply with new regulations, or fail to meet evolving investor, industry or stakeholder expectations and concerns regarding ESG issues, investors may reconsider their capital investment in our Company, and customers and consumers may choose to stop purchasing our products, which could have a material adverse effect on our reputation, business or financial condition.
The Company is subject to or otherwise responsible for a variety of litigation and other legal and regulatory proceedings in the ordinary course of business that can adversely affect our business, results of operations and financial condition.
From time to time, the Company and its subsidiaries are subject to or otherwise responsible for a variety of litigation and other legal and regulatory proceedings in the ordinary course of business, as well as regulatory subpoenas, requests for information, investigations and enforcement. Defending or otherwise responding to these matters can divert the Company’s management’s attention and may cause it to incur significant expenses. The Company believes it has meritorious arguments in its current litigation matters and believes any outcome, either individually or in the aggregate, will not be material to the Company’s financial position or results of operations. However, each of these matters is subject to uncertainties, and it is possible that some of these matters may be resolved unfavorably to the Company.
GENERAL RISK FACTORS
The effects of climate change could harm the Company’s business.
The Company’s manufacturing processes for certain of its products involve the use of chemicals and other substances that are regulated under various international, federal, state and local laws governing the environment. In the event that any future climate change legislation would require that stricter standards be imposed by domestic or international environmental regulatory authorities with respect to the use and/or levels of possible emissions from such chemicals and/or other substances, the Company may be required to make certain changes and adaptations to its manufacturing processes. Any such changes could have a material adverse effect on the financial statements of the Company.
Another potential effect of climate change is an increase in the severity of global weather conditions. The Company’s manufacturing facilities are located in the U.S., U.K., Ireland and Germany. In addition, the Company manufactures a growing percentage of its HPLC, UPLC and MS products in both Singapore and Ireland. Severe weather and geological conditions or events, including earthquakes, hurricanes and/or tsunamis, could potentially cause significant damage to the Company’s manufacturing facilities in each of these countries. The effects of such damage and the resulting disruption of manufacturing operations and the impact of lost sales could have a material adverse impact on the financial results of the Company.
Estimates and assumptions made in accounting for the Company’s results from operations are dependent on future results, which involve significant judgments and may be imprecise and may differ materially from actual results.
The preparation of consolidated financial statements in conformity with generally accepted accounting principles requires the Company to make estimates and judgments that affect the reported amounts of assets, liabilities, revenues and expenses, and related disclosure of contingent liabilities at the dates of the financial statements. These estimates and assumptions must be made due to certain information used in preparation of our financial statements which is dependent on future events, cannot be calculated with a high degree of precision from data available or is not capable of being readily calculated based on generally accepted methodologies. The Company believes that the accounting related to revenue recognition, goodwill and intangible assets, income taxes, uncertain tax positions, litigation, business combinations and asset acquisitions and inventory valuation involves
26
Table of Contents
significant judgments and estimates. Actual results for all estimates could differ materially from the estimates and assumptions used, which could have a material adverse effect on our financial condition and results of operations.
The Company’s financial condition and results of operations could be adversely affected by changes to the Company’s retirement plans or retirement plan assets.
The Company sponsors various retirement plans, both inside and outside the United States. Any changes in regulations made by governments in countries in which the Company sponsors retirement plans could adversely impact the Company’s cash flows or results of operations. In connection with these retirement plans, the Company is exposed to market risks associated with changes in the various capital markets. For example, changes in long-term interest rates affect the discount rate that is used to measure the Company’s retirement plan obligations and related expense. In addition, changes in the market value of investments held by the retirement plans could materially impact the funded status of the retirement plans, and affect the related pension expense and level and timing of contributions required under applicable laws.
The Company’s financial condition and results of operations could be adversely affected if the Company is unable to maintain a sufficient level of cash flow.
The Company had $2.4 billion in debt and $396 million in cash, cash equivalents and investments as of December 31, 2023. As of December 31, 2023, the Company also had the ability to borrow an additional $0.9 billion from its existing, committed credit facility. All but a small portion of the Company’s debt was in the U.S. There is a substantial cash requirement in the United States to fund operations and capital expenditures, service debt interest obligations, finance potential United States acquisitions and continue authorized stock repurchase programs. As such, the Company’s financial condition and results of operations could be adversely impacted if the Company is unable to generate and maintain a sufficient level of cash flow to address these requirements through (1) cash from operations, (2) the Company’s ability to access its existing cash and revolving credit facility, (3) the ability to expand the Company’s borrowing capacity and (4) other sources of capital obtained at an acceptable cost.
Debt covenants, and the Company’s failure to comply with them, could negatively impact the Company’s capital and financial results.
The Company’s existing debt is, and future debt may be, subject to restrictive debt covenants that limit the Company’s ability to engage in certain activities that could otherwise benefit the Company. These debt covenants include restrictions on the Company’s ability to enter into certain contracts or agreements, which may limit the Company’s ability to make dividend or other payments, secure other indebtedness, enter into transactions with affiliates and consolidate, merge or transfer all or substantially all of the Company’s assets. The Company is also required to meet specified financial ratios under the terms of the Company’s debt agreements. The Company’s ability to comply with these financial restrictions and all other covenants is dependent on the Company’s future performance, which is subject to, but not limited to, prevailing economic conditions and other factors, including factors that are beyond the Company’s control, such as foreign exchange rates, interest rates, changes in technology and changes in the level of competition. Failure to comply with restrictive debt covenants that are not waived or cured could result in an event of default under the applicable debt instrument, which could permit acceleration of the applicable debt and require the Company to prepay the debt before its scheduled due date.
Item 1B: Unresolved Staff Comments
None.
Item 1C: Cybersecurity
We maintain a robust system of data protection and cybersecurity resources, technology and processes. We regularly evaluate new and emerging risks and ever-changing legal and compliance requirements. We make
27
Table of Contents
strategic investments to address these risks and legal and compliance requirements to keep Company, customer and employee data secure. We monitor risks of sensitive information compromise at our business partners where relevant and reevaluate these risks on a periodic basis. We also perform annual and ongoing cybersecurity training and awareness for our employees.
We have a longstanding information security risk management framework structured according to the National Institute of Standards and Technology Cybersecurity Framework, industry best practices, privacy legislation, and other global and local standards and regulations. This risk management framework is under the specific oversight of the Company’s Vice President and Chief Information Officer (the “CIO”) and includes a defense-in-depth approach with multiple layers of security controls, including network segmentation, security monitoring, endpoint protection, and identity and access management, as well as data protection best practices and data loss prevention controls. Our Audit and Finance Committee is updated on the overall performance of our information security risk management framework on an annual basis by the CIO.
Our cybersecurity awareness program includes regular phishing simulations, annual general cybersecurity awareness, and data protection modules, as well as more contextual and personalized modules for targeted users and roles. We also perform simulations and drills at both a technical and leadership level at least annually. We incorporate external expertise and guidance in all aspects of our cybersecurity program. We complete annual internal security audits and vulnerability assessments of the Company’s information systems and related controls, including systems affecting personal data. In addition, we leverage cybersecurity specialists to complete annual external audits and objective assessments of our cybersecurity program and practices, including our data protection practices, as well as to conduct targeted attack simulations. We continually enhance our information security capabilities in order to protect against emerging threats, while also increasing our ability to detect and respond to cyber incidents and maximize our resilience to recover from potential cyber-attacks. We have a robust incident response plan in place that provides a documented playbook for responding to cybersecurity incidents and facilitates coordination across multiple parts of our Company. Additionally, we have purchased network security and cyber liability insurance in order to provide a level of financial protection, should a data breach occur.
Despite the existence of mitigation measures, the Company’s systems and those of its partners remain potentially vulnerable to cybersecurity threats, any of which could have a material adverse effect on the Company’s business. To date, cybersecurity incidents have not resulted in a material adverse impact to the Company’s business strategy, results of operations and financial condition, but future incidents could have such an impact. See Item 1A, Risk Factors - Risks Related to Cybersecurity.
The Board of Directors oversees the Company’s information security risk management framework that seeks to identify new risks, develop and implement risk mitigation plans, and monitor the results affecting the Company’s business and operations on an ongoing basis. The CIO manages this framework, in collaboration with the Company’s businesses and functions. The CIO presents updates to the Audit and Finance Committee at least annually and, as necessary, to the full Board of Directors. These reports include detailed updates on the Company’s performance preparing for, preventing, detecting, responding to and recovering from cyber incidents. The CIO also promptly informs and updates the Board of Directors about any information security incidents that may pose significant risk to the Company. The Company’s program is periodically evaluated by external experts, and the results of those reviews are reported to the Audit and Finance Committee and the Board of Directors. Together with management, the Audit and Finance Committee reviews the Company’s risk assessment and risk management practices and discusses major cybersecurity risk exposures as well as steps taken by management to monitor and control such exposures.
The Company’s Vice President and Chief Information Officer has over 24 years of business experience managing risks from cybersecurity threats/developing and implementing cybersecurity policies and procedures, as well as several relevant certifications.
28
Table of Contents
Item 2: Properties
Waters Corporation operates 21 United States facilities and 71 international facilities, including field offices. The Company believes its facilities are suitable and adequate for its current production level and for reasonable growth over the next several years. The Company’s primary facilities are summarized in the table below.
Primary Facility Locations (1)
| Location |
Function (2) | Owned/Leased | ||
| Golden, CO |
M, R, S, D, A | Leased | ||
| New Castle, DE |
M, R, S, D, A | Owned | ||
| Franklin, MA |
D | Leased | ||
| Milford, MA |
M, R, S, A | Owned | ||
| Taunton, MA |
M, R | Owned | ||
| Cambridge, MA |
R, S | Leased | ||
| Eden Prairie, MN |
M, R, S, D, A | Leased | ||
| Nixa, MO |
M, S, D, A | Leased | ||
| Lindon, UT |
M, R, S, D, A | Leased | ||
| Santa Barbara, CA |
M, R, S, D, A | Leased | ||
| Beijing, China |
S, A | Leased | ||
| Shanghai, China |
R, S, A | Leased | ||
| Solihull, England |
M, A | Owned | ||
| Wilmslow, England |
M, R, S, D, A | Owned | ||
| St. Quentin, France |
S, A | Leased | ||
| Hüllhorst, Germany |
M, R, S, D, A | Owned | ||
| Wexford, Ireland |
M, R, D, A | Owned | ||
| Bangalore, India |
M, R, S, D, A | Owned/Leased | ||
| Etten-Leur, Netherlands |
S, D, A | Owned | ||
| Brasov, Romania |
R, A | Leased | ||
| Singapore |
R, S, D, A | Leased |
| (1) | The Company operates more than one primary facility within certain states and foreign countries. |
| (2) | M = Manufacturing; R = Research; S = Sales and Service; D = Distribution; A = Administration |
The Company operates and maintains 10 field offices in the United States and 58 field offices abroad in addition to sales offices in the primary facilities listed above. The Company’s field office locations are listed below.
Field Office Locations (3)
| United States |
International | |||||
| Costa Mesa, CA |
Australia | Hong Kong | People’s Republic of China | |||
| Pleasanton, CA |
Austria | India | Portugal | |||
| Wood Dale, IL |
Belgium | Ireland | Poland | |||
| Carmel, IN |
Brazil | Israel | Puerto Rico | |||
| Woburn, MA |
Canada | Italy | Spain | |||
| Columbia, MD |
Czech Republic | Japan | Sweden | |||
| Morrisville, NC |
Denmark | Korea | Switzerland | |||
| Parsippany, NJ |
Finland | Malaysia | Taiwan | |||
| Plymouth Meeting, PA |
France | Mexico | United Arab Emirates | |||
| Bellaire, TX |
Germany | Netherlands | United Kingdom | |||
| Hungary | Norway | |||||
| (3) | The Company operates more than one field office within certain states and foreign countries. |
29
Table of Contents
Item 3: Legal Proceedings
From time to time, the Company and its subsidiaries are involved in various lawsuits, claims, investigations and proceedings covering a wide range of matters that arise in the ordinary course of business. The Company believes it has meritorious arguments in its current litigation matters and believes any outcome, either individually or in the aggregate, will not be material to the Company’s financial position or results of operations. However, each of these matters is subject to uncertainties, and it is possible that some of these matters may be resolved unfavorably to the Company.
Item 4: Mine Safety Disclosures
Not applicable.
30
Table of Contents
PART II
| Item 5: | Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities |
The Company’s common stock is registered under the Exchange Act and is listed on the New York Stock Exchange under the symbol “WAT”. As of February 23, 2024, the Company had 69 common stockholders of record. The Company has not declared or paid any dividends on its common stock in its past three fiscal years and does not intend to pay cash dividends in the foreseeable future. Any future determination to pay cash dividends will be made at the discretion of the Board of Directors and will depend on restrictions and other factors the Board of Directors may deem relevant. The Company has not made any sales of unregistered equity securities in the years ended December 31, 2023, 2022 and 2021.
Securities Authorized for Issuance under Equity Compensation Plans
Equity compensation plan information is incorporated by reference from Part III, Item 12, Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters, of this document and should be considered an integral part of this Item 5.
31
Table of Contents
Stock Price Performance Graph
The following performance graph and related information shall not be deemed to be “soliciting material” or to be “filed” with the SEC, nor shall such information be incorporated by reference into any future filing under the Securities Act or the Exchange Act, except to the extent that the Company specifically incorporates it by reference into such filing.
The following graph compares the cumulative total return on $100 invested as of December 31, 2018 (the last day of public trading of the Company’s common stock in fiscal year 2018) through December 31, 2023 (the last day of public trading of the common stock in fiscal year 2023) in the Company’s common stock, the NYSE Market Index, the SIC Code 3826 Index and the S&P 500 Index. The return of the indices is calculated assuming reinvestment of dividends during the period presented. The Company has not paid any dividends since its IPO. The stock price performance shown on the graph below is not necessarily indicative of future price performance.
COMPARISON OF CUMULATIVE TOTAL RETURN SINCE DECEMBER 31, 2018
AMONG WATERS CORPORATION, NYSE MARKET INDEX, SIC CODE 3826 INDEX – LABORATORY ANALYTICAL INSTRUMENTS AND S&P 500 INDEX
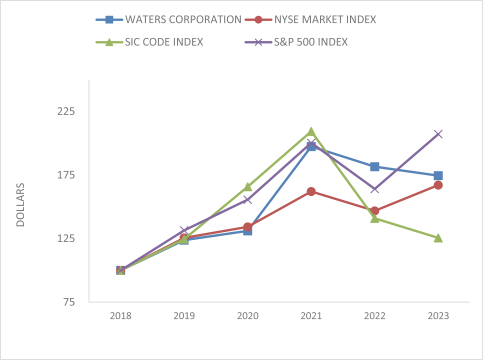
| 2018 | 2019 | 2020 | 2021 | 2022 | 2023 | |||||||||||||||||||
| WATERS CORPORATION |
100.00 | 123.85 | 131.15 | 197.51 | 181.60 | 174.52 | ||||||||||||||||||
| NYSE MARKET INDEX |
100.00 | 125.51 | 134.28 | 162.04 | 146.89 | 167.12 | ||||||||||||||||||
| SIC CODE INDEX |
100.00 | 124.58 | 165.82 | 209.38 | 140.93 | 125.60 | ||||||||||||||||||
| S&P 500 INDEX |
100.00 | 131.49 | 155.68 | 200.37 | 164.08 | 207.21 | ||||||||||||||||||
32
Table of Contents
Purchases of Equity Securities by the Issuer
The following table provides information about purchases by the Company during the three months ended December 31, 2023 of equity securities registered by the Company under the Exchange Act (in thousands, except per share data):
| Period |
Total Number of Shares Purchased (1) |
Average Price Paid per Share |
Total Number of Shares Purchased as Part of Publicly Announced Programs |
Maximum Dollar Value of Shares That May Yet Be Purchased Under the Programs (2) |
||||||||||||
| October 1, 2023 to October 28, 2023 |
— | $ | — | — | $ | 961,207 | ||||||||||
| October 29, 2023 to November 25, 2023 |
— | $ | — | — | $ | 961,207 | ||||||||||
| November 26, 2023 to December 31, 2023 |
2 | $ | 317.00 | — | $ | 961,207 | ||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total |
2 | $ | 317.00 | — | $ | 961,207 | ||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| (1) | The Company repurchased approximately one thousand shares of common stock at a cost of less than $1 million related to the vesting of restricted stock during the three months ended December 31, 2023. |
| (2) | In January 2019, the Company’s Board of Directors authorized the Company to repurchase up to $4 billion of its outstanding common stock in open market or private transactions over a two-year period. This program replaced the remaining amounts available under the pre-existing authorization. In December 2020, the Company’s Board of Directors authorized the extension of the share repurchase program through January 21, 2023. In December 2022, the Company’s Board of Directors amended and extended this repurchase program’s term by one year such that it shall now expire on January 21, 2024 and increased the total authorization to $4.8 billion, an increase of $750 million. In December 2023, the Company’s Board of Directors authorized the extension of the share repurchase program through January 21, 2025. The Company’s remaining authorization is $1.0 billion. The size and timing of these purchases, if any, will depend on our stock price and market and business conditions, as well as other factors. |
33
Table of Contents
Item 6: Reserved
Item 7: Management’s Discussion and Analysis of Financial Condition and Results of Operations
Business Overview
The Company has two operating segments: Waters and TA. Waters products and services primarily consist of high-performance liquid chromatography (“HPLC”), ultra-performance liquid chromatography (“UPLC” and, together with HPLC, referred to as “LC”), mass spectrometry (“MS”) and precision chemistry consumable products and related services. TA products and services primarily consist of thermal analysis, rheometry and calorimetry instrument systems and service sales. The Company’s products are used by pharmaceutical, biochemical, industrial, nutritional safety, environmental, academic and government customers. These customers use the Company’s products to detect, identify, monitor and measure the chemical, physical and biological composition of materials and to predict the suitability and stability of fine chemicals, pharmaceuticals, water, polymers, metals and viscous liquids in various industrial, consumer goods and healthcare products. Operations of the recently acquired Wyatt business are part of the Waters operating segment.
Wyatt Acquisition
On May 16, 2023, the Company completed the acquisition of Wyatt Technology, LLC and its three operating subsidiaries, Wyatt Technology Europe GmbH, Wyatt Technology France and Wyatt Technology UK Ltd. (collectively, “Wyatt”), for a total purchase price of $1.3 billion in cash. Wyatt is a pioneer in innovative light scattering and field-flow fractionation instruments, software, accessories, and services. The acquisition will expand Waters portfolio and increase exposure to large molecule applications. The Company financed this transaction with a combination of cash on its balance sheet and borrowings under its revolving credit facility. The Company’s financial results for the year ended December 31, 2023 include the financial results of the Wyatt acquisition from the acquisition date.
Financial Overview
The Company’s operating results are as follows for the years ended December 31, 2023, 2022 and 2021 (dollars in thousands, except per share data):
| Year Ended December 31, | % change | |||||||||||||||||||
| 2023 | 2022 | 2021 | 2023 vs 2022 |
2022 vs 2021 |
||||||||||||||||
| Revenues: |
||||||||||||||||||||
| Product sales |
$ | 1,903,050 | $ | 1,988,169 | $ | 1,822,070 | (4 | %) | 9 | % | ||||||||||
| Service sales |
1,053,366 | 983,787 | 963,804 | 7 | % | 2 | % | |||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Total net sales |
2,956,416 | 2,971,956 | 2,785,874 | (1 | %) | 7 | % | |||||||||||||
| Costs and operating expenses: |
||||||||||||||||||||
| Cost of sales |
1,195,223 | 1,248,182 | 1,156,533 | (4 | %) | 8 | % | |||||||||||||
| Selling and administrative expenses |
736,014 | 658,026 | 626,968 | 12 | % | 5 | % | |||||||||||||
| Research and development expenses |
174,945 | 176,190 | 168,358 | (1 | %) | 5 | % | |||||||||||||
| Purchased intangibles amortization |
32,558 | 6,366 | 7,143 | 411 | % | (11 | %) | |||||||||||||
| Acquired in-process research and development |
— | 9,797 | — | * | * | * | * | |||||||||||||
| Litigation provision |
— | — | 5,165 | — | * | * | ||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Operating income |
817,676 | 873,395 | 821,707 | (6 | %) | 6 | % | |||||||||||||
| Operating income as a % of sales |
27.7 | % | 29.4 | % | 29.5 | % | ||||||||||||||
| Other income, net |
807 | 2,228 | 17,203 | (64 | %) | (87 | %) | |||||||||||||
| Interest expense, net |
(82,240 | ) | (37,777 | ) | (32,717 | ) | 118 | % | 15 | % | ||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Income before income taxes |
736,243 | 837,846 | 806,193 | (12 | %) | 4 | % | |||||||||||||
| Provision for income taxes |
94,009 | 130,091 | 113,350 | (28 | %) | 15 | % | |||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Net income |
$ | 642,234 | $ | 707,755 | $ | 692,843 | (9 | %) | 2 | % | ||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Net income per diluted common share |
$ | 10.84 | $ | 11.73 | $ | 11.17 | (8 | %) | 5 | % | ||||||||||
| ** | Percentage not meaningful |
34
Table of Contents
The Company’s net sales decreased 1% in 2023 as compared to 2022 and increased 7% in 2022 as compared to 2021. The Company’s sales in 2023 were negatively impacted by a 22% reduction of sales in China due to lower customer demand for our products. The sales growth in 2022 was driven by strong customer demand across most major geographies, end markets and product categories. Excluding China, the Company’s sales growth increased 5% and 6% in 2023 and 2022, respectively. Foreign currency translation decreased sales growth by 1% and 5% in 2023 and 2022, respectively. The Wyatt acquisition increased sales growth by 3% in 2023.
Instrument system sales decreased 7% in 2023 as compared to 2022 and increased 11% in 2022 as compared to 2021. In 2023, the decrease in instrument system sales resulted from weaker customer demand in China, which was partially offset by sales growth in the U.S. and Europe. Excluding China, the Company’s instrument system sales grew 1%. In addition, Wyatt’s instrument system sales added 4% to the Company’s instrument system sales growth. In 2022, the increase was driven by the broad-based increase in customer demand across all existing and newly introduced LC, LC-MS, and Thermal Analysis instrument system sales. Foreign currency translation decreased instrument system sales growth by 1% and 5% in 2023 and 2022, respectively.
Recurring revenues (combined sales of precision chemistry consumables and services) increased 6% and 3% in 2023 and 2022, respectively. Recurring revenues were negatively impacted by foreign currency translation in 2023 and 2022, which decreased sales by 1% and 6%, respectively.
Operating income was $818 million in 2023, a decrease of 6% as compared to 2022. This decrease in operating income was primarily due to higher salary expenses related to merit compensation, $26 million in severance-related costs associated with a workforce reduction and costs related to the Wyatt acquisition, including $13 million in due diligence costs, $27 million of intangible asset amortization and $19 million of costs associated with retention agreements. The negative effect of foreign currency translation lowered operating income by approximately $23 million during 2023.
In July 2023, the Company made organizational changes to better align its resources with its growth and innovation strategies, resulting in a worldwide workforce reduction that impacted approximately 5% of the Company’s employees. The Company incurred approximately $26 million of severance-related costs and paid approximately $19 million of severance-related costs in 2023, with the remaining costs to be paid in the first half of 2024. The Company estimates that the savings from this reduction in workforce will be approximately $48 million on an annual basis.
Operating income was $873 million in 2022, an increase of 6% as compared to 2021. This increase was primarily a result of the increase in sales volume and pricing increases, partially offset by higher electronic component and freight inflationary costs and the negative effect of foreign currency translation. The effect of foreign currency translation lowered operating income by approximately $71 million during 2022.
Operating income as a percentage of sales was 27.7%, 29.4% and 29.5% in 2023, 2022 and 2021, respectively.
The Company’s effective tax rates were 12.8%, 15.5% and 14.1% for 2023, 2022 and 2021, respectively. Net income per diluted share was $10.84, $11.73 and $11.17 in 2023, 2022 and 2021, respectively.
The Company generated $603 million, $612 million and $747 million of net cash flows provided by operating activities in 2023, 2022 and 2021, respectively. The decrease in 2023 operating cash flow was primarily a result of lower sales volumes, higher income tax payments and higher incentive compensation payments in 2023 as compared to 2022.
Net cash used in investing activities included $1.3 billion for the Wyatt acquisition in 2023 and capital expenditures related to property, plant, equipment and software capitalization of $161 million, $176 million and
35
Table of Contents
$161 million in 2023, 2022 and 2021, respectively. The cash flows used in investing activities in 2023, 2022 and 2021 included $16 million, $32 million, and $49 million, respectively, of capital expenditures related to the major expansion of the Company’s precision chemistry consumable operations in the United States.
During 2023, the Company funded the Wyatt acquisition with a combination of cash on hand and borrowings under its revolving credit facility. The Company’s outstanding debt on December 31, 2023 was $2.4 billion, a change of $0.8 billion from December 31, 2022, which resulted in the Company’s interest expense in 2023 increasing by $50 million to $99 million.
On March 3, 2023, the Company entered into an agreement to amend the credit agreement governing its revolving credit facility (the “2023 Amendment”). The 2023 Amendment increases the borrowing capacity by $200 million to an aggregate total borrowing capacity of $2.0 billion.
In December 2023, the Company’s Board of Directors authorized the extension of the existing share repurchase program through January 21, 2025. The Company’s remaining authorization is $1.0 billion. During the years ended December 31, 2023, 2022 and 2021, the Company repurchased $58 million, $616 million and $640 million of the Company’s outstanding common stock, respectively, under the share repurchase programs. While the Company believes that it has the financial flexibility to fund these share repurchases, as well as to invest in research, technology and business acquisitions, given current cash levels and debt borrowing capacity, it has temporarily suspended its share repurchases due to its acquisition of Wyatt in the second quarter of 2023.
Results of Operations
Sales by Geography
Geographic sales information is presented below for the years ended December 31, 2023, 2022 and 2021 (dollars in thousands):
| Year Ended December 31, | % change | |||||||||||||||||||
| 2023 | 2022 | 2021 | 2023 vs. 2022 |
2022 vs. 2021 |
||||||||||||||||
| Net Sales: |
||||||||||||||||||||
| Asia: |
||||||||||||||||||||
| China |
$ | 440,707 | $ | 565,143 | $ | 521,128 | (22 | %) | 8 | % | ||||||||||
| Japan |
167,202 | 167,220 | 182,597 | — | (8 | %) | ||||||||||||||
| Asia Other |
399,916 | 399,380 | 372,040 | — | 7 | % | ||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Total Asia |
1,007,825 | 1,131,743 | 1,075,765 | (11 | %) | 5 | % | |||||||||||||
| Americas: |
||||||||||||||||||||
| United States |
927,982 | 886,140 | 774,014 | 5 | % | 14 | % | |||||||||||||
| Americas Other |
180,591 | 169,495 | 151,206 | 7 | % | 12 | % | |||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Total Americas |
1,108,573 | 1,055,635 | 925,220 | 5 | % | 14 | % | |||||||||||||
| Europe |
840,018 | 784,578 | 784,889 | 7 | % | — | ||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Total net sales |
$ | 2,956,416 | $ | 2,971,956 | $ | 2,785,874 | (1 | %) | 7 | % | ||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
In 2023, sales decreased 1% as compared to 2022, primarily as a result of a 22% decrease in China sales during 2023, which was partially offset by broad-based sales growth across most other major regions. The decline in China sales was primarily driven by lower demand for our instrument systems and chemistry products resulting from increased government regulations and lower spending by our customers due to weak economic conditions in China. Excluding China, the Company’s sales increased 5% and 6% in 2023 and 2022, respectively. Foreign currency translation decreased sales growth by 1% and 5% in 2023 and 2022, respectively.
In 2023, sales increased 5% in the U.S. and 7% in Europe, while decreasing 11% in Asia, with the effect of foreign currency translation increasing sales growth in Europe by 2% and decreasing sales growth in Asia by 4%,
36
Table of Contents
which includes a 9% decrease in sales in Japan resulting from foreign currency translation. Wyatt’s sales contributed 5% and 3% of sales growth to the U.S. and Europe in 2023, respectively.
The sales growth in 2022 was broad-based across most major regions. Foreign currency translation decreased total sales growth by 5% in 2022 as the U.S. dollar strengthened significantly against all other major currencies. The geographies that were the most negatively impacted by the strengthening of the U.S. dollar in 2022 were Europe and Japan, as the weakening of the euro and Japanese yen lowered sales growth in Europe and Japan by 10% and 17%, respectively. China sales increased 8% in 2022, with foreign currency translation decreasing China sales growth by 2% in 2022. This increase in China sales was driven by strong customer demand for our products and services despite the negative impact that the COVID-19 pandemic had on our business in China in 2022.
Sales by Trade Class
Net sales by customer class are presented below for the years ended December 31, 2023, 2022 and 2021 (dollars in thousands):
| Year Ended December 31, | % change | |||||||||||||||||||
| 2023 | 2022 | 2021 | 2023 vs. 2022 |
2022 vs. 2021 |
||||||||||||||||
| Pharmaceutical |
$ | 1,696,875 | $ | 1,751,665 | $ | 1,667,061 | (3 | %) | 5 | % | ||||||||||
| Industrial |
909,003 | 909,805 | 829,204 | — | 10 | % | ||||||||||||||
| Academic and government |
350,538 | 310,486 | 289,609 | 13 | % | 7 | % | |||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Total net sales |
$ | 2,956,416 | $ | 2,971,956 | $ | 2,785,874 | (1 | %) | 7 | % | ||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
In 2023, sales to pharmaceutical customers decreased 3%, primarily driven by weakness in customer demand in China, with foreign currency translation decreasing pharmaceutical sales growth by 1% and Wyatt contributing 3% to the Company’s pharmaceutical sales growth. Combined sales to industrial customers, which include material characterization, food, environmental and fine chemical markets, were flat in 2023, with foreign currency translation decreasing industrial sales growth by 1% and Wyatt contributing 1% to industrial sales growth. Combined sales to academic and government customers increased 13% in 2023, with foreign currency translation decreasing academic and government sales growth by 1% and Wyatt contributing 4% to academic and government sales growth. Sales to our academic and government customers are highly dependent on when institutions receive funding to purchase our instrument systems and, as such, sales can vary significantly from period to period.
In 2022, sales to pharmaceutical customers increased 5%, driven by strong growth in most major regions, partially offset by the negative impact from foreign currency translation which decreased pharmaceutical sales by 5%. Combined sales to industrial customers increased 10%, with foreign currency translation decreasing sales growth by 5%. Combined sales to academic and government customers increased 7%, with foreign currency translation decreasing academic and government sales growth by 6%.
37
Table of Contents
Waters Products and Services Net Sales
Net sales for Waters products and services were as follows for the years ended December 31, 2023, 2022 and 2021 (dollars in thousands):
| Year Ended December 31, | % change | |||||||||||||||||||||||||||||||
| 2023 | % of Total |
2022 | % of Total |
2021 | % of Total |
2023 vs. 2022 |
2022 vs. 2021 |
|||||||||||||||||||||||||
| Waters instrument systems |
$ | 1,108,702 | 43 | % | $ | 1,210,456 | 46 | % | $ | 1,089,248 | 44 | % | (8 | %) | 11 | % | ||||||||||||||||
| Chemistry consumables |
541,469 | 20 | % | 525,399 | 20 | % | 507,209 | 21 | % | 3 | % | 4 | % | |||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||
| Total Waters product sales |
1,650,171 | 63 | % | 1,735,855 | 66 | % | 1,596,457 | 65 | % | (5 | %) | 9 | % | |||||||||||||||||||
| Waters service |
951,419 | 37 | % | 890,607 | 34 | % | 876,626 | 35 | % | 7 | % | 2 | % | |||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||
| Total Waters net sales |
$ | 2,601,590 | 100 | % | $ | 2,626,462 | 100 | % | $ | 2,473,083 | 100 | % | (1 | %) | 6 | % | ||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||
Waters products and service sales decreased 1% and 6% in 2023 and 2022, respectively, with the effect of foreign currency translation decreasing Waters sales growth by 1% and 6% in 2023 and 2022, respectively. The Wyatt acquisition increased Waters products and service sales by approximately 3% in 2023. Waters instrument system sales (LC and MS technology-based) decreased 8% in 2023, primarily driven by weaker customer demand in China. Excluding China, the Company’s instrument system sales were flat as compared to 2022. In addition, Wyatt’s instrument system sales contributed 5% to Waters instrument system sales growth in 2023. Waters chemistry consumables sales were significantly impacted by the lower customer demand in China for our products. Excluding China, the Company’s chemistry sales grew 7% in 2023. This sales growth was primarily due to the continued strong demand in most major geographies, driven by the uptake in columns and application-specific testing kits to pharmaceutical customers, partially offset by the negative impact from foreign currency translation, which decreased chemistry sales growth by 1% in 2023. Waters service sales increased 7% in 2023 due to higher service demand billing, partially offset by the negative impact from foreign currency translation, which decreased service sales growth by 1% in 2023. Wyatt service revenues added 2% to Waters service revenue growth in 2023.
In 2022, Waters products and service sales increased 6%, with foreign currency translation decreasing Waters sales growth by 6%. Waters instrument system sales grew 11%, with foreign currency translation lowering sales growth by 5%. The increase in the Waters instrument system sales can be attributed to strong customer demand for our existing products as well as growing contributions made by recent product introductions. The increase in Waters chemistry consumables sales was primarily due to the strong demand in most major geographies, partially offset by the negative impact from foreign currency translation which decreased sales by 5%. Waters service sales increased due to higher service demand billing, particularly in China and the United States, partially offset by the negative impact from foreign currency translation which decreased by 6%.
TA Product and Services Net Sales
Net sales for TA products and services were as follows for the years ended December 31, 2023, 2022 and 2021 (dollars in thousands):
| Year Ended December 31, | % change | |||||||||||||||||||||||||||||||
| 2023 | % of Total |
2022 | % of Total |
2021 | % of Total |
2023 vs. 2022 |
2022 vs. 2021 |
|||||||||||||||||||||||||
| TA instrument systems |
$ | 252,879 | 71 | % | $ | 252,314 | 73 | % | $ | 225,613 | 72 | % | — | 12 | % | |||||||||||||||||
| TA service |
101,947 | 29 | % | 93,180 | 27 | % | 87,178 | 28 | % | 9 | % | 7 | % | |||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||
| Total TA net sales |
354,826 | 100 | % | 345,494 | 100 | % | 312,791 | 100 | % | 3 | % | 10 | % | |||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||
TA instrument system and service sales increased 3% and 10% in 2023 and 2022, respectively. Foreign currency translation had minimal impact on sales growth in 2023 and decreased sales growth by 6% in 2022. In
38
Table of Contents
2023, sales growth was broad-based across most major geographies, partially offset by weakness in China and the rest of Asia. These increases were primarily driven by strong customer demand for our thermal analysis instruments and services.
Cost of Sales
Cost of sales decreased 4% in 2023 as compared to 2022, primarily due to the change in sales mix and the lower material and freight costs. In 2022, cost of sales increased 8% as compared to 2021, primarily due to the increase in sales volumes during the year as well as an increase in electronic component and freight inflationary costs.
Cost of sales is affected by many factors, including, but not limited to, foreign currency translation, product mix, product costs of instrument systems and amortization of software platforms. At current foreign currency exchange rates, the Company expects foreign currency translation to be neutral to gross profit during 2024.
Selling and Administrative Expenses
Selling and administrative expenses increased 12% and 5% in 2023 and 2022, respectively. The increase in 2023 is primarily driven by severance-related costs in connection with a reduction in workforce, which increased expenses by 4%; the Wyatt acquisition due diligence and integration costs, which increased expenses by 2%; and the Wyatt acquisition-related retention expense, which increased expenses by 3%. These increases were partially offset by lower incentive compensation costs. The increase in selling and administrative expenses in 2022 as compared to 2021 can be attributed to higher salary merit and variable incentive compensation costs due to an increase in the number of employees. The effect of foreign currency translation had minimal impact on selling and administrative expenses in 2023 and decreased expenses by 4% in 2022.
As a percentage of net sales, selling and administrative expenses were 24.9%, 22.1% and 22.5% for 2023, 2022, and 2021, respectively.
Research and Development Expenses
Research and development expenses decreased 1% and increased 5% in 2023 and 2022, respectively. The decrease in research and development expenses in 2023 can be attributed to increases from merit compensation and costs associated with new products and the development of new technology initiatives, being offset by lower incentive compensation costs. The impact of foreign currency exchange decreased expenses by 1% and 3% in 2023 and 2022, respectively.
Purchased Intangibles Amortization
The increase in purchased intangible amortization of $26 million in 2023 can be attributed to the Wyatt acquisition intangible assets.
Acquired In-Process Research & Development
In 2022, the Company completed an asset acquisition in which the CDMS technology assets of Megadalton were acquired for approximately $10 million in total purchase price, of which $5 million was paid at closing and the remaining $4 million will be paid in the future at various dates through 2029.
Other (Expense) Income, net
In 2022, the Company sold equity an equity investment for $10 million in cash and recorded a gain on the sale of approximately $7 million in other income, net on the statement of operations. The Company also incurred $6 million in losses on an equity investment within other income, net on the statement of operations.
In 2021, the Company executed a settlement agreement to resolve patent infringement litigation with Bruker Corporation and Bruker Daltronik GmbH regarding their timsTOF product line. In connection with the
39
Table of Contents
settlement, the Company is entitled to receive $10 million in guaranteed payments, including minimum royalty payments. In 2021, the Company recorded an unrealized gain of $10 million due to an observable change in the fair value of an existing investment that the Company does not have the ability to exercise significant influence over.
Interest Expense, net
Net interest expense in 2023 increased $44 million as compared to 2022 due to the additional borrowings by the Company to fund the Wyatt acquisition in 2023. Net interest expense in 2022 increased $5 million as compared to 2021 due to the lower interest income benefit from the lower notional amount of interest rate cross currency swap agreements.
Provision for Income Taxes
The four principal jurisdictions in which the Company manufactures are the U.S., Ireland, the U.K. and Singapore, where the statutory tax rates were 21%, 12.5%, 25% and 17%, respectively, as of December 31, 2023. The Company has a new Development and Expansion Incentive in Singapore that provides a concessionary income tax rate of 5% on certain types of income for the period April 1, 2021 through March 31, 2026. Prior to April 1, 2021, the Company had a tax exemption on income arising from qualifying activities in Singapore based upon the achievement of certain contractual milestones, which the Company met as of December 31, 2020 and maintained through March 2021. The effect of applying the concessionary income tax rates rather than the statutory tax rate to income arising from qualifying activities in Singapore increased the Company’s net income by $16 million, $20 million and $20 million, and increased the Company’s net income per diluted share by $0.27, $0.33 and $0.32 for the years ended December 31, 2023, 2022 and 2021, respectively.
The Company’s effective tax rate for the years ended December 31, 2023, 2022 and 2021 was 12.8%, 15.5% and 14.1%, respectively.
The 2023 effective tax rate differed from the 21% U.S. statutory tax rate primarily due to the jurisdictional mix of earnings, an $18 million recognition of a previously unrecognized tax benefit as a result of the completion of a tax examination, a $15 million provision related to the Global Intangible Low-Taxed Income (“GILTI”) tax and a tax benefit of $3 million on stock-based compensation.
The 2022 effective tax rate differed from the 21% U.S. statutory tax rate primarily due to the jurisdictional mix of earnings, an $18 million provision related to the GILTI tax and a tax benefit of $7 million on stock-based compensation.
The 2021 effective tax rate differed from the U.S. federal statutory tax rate primarily due to the jurisdictional mix of earnings, a $10 million provision related to the GILTI tax and a tax benefit of $7 million on stock-based compensation.
Effective starting in 2024, various foreign jurisdictions are beginning to implement aspects of the guidance issued by the Organization for Economic Co-operation and Development related to the new Pillar Two system of global minimum tax rules. The Company does not believe these changes in tax law will have a material impact on the Company’s financial position, results of operations and cash flows in 2024. The Company continues to monitor the adoption of the Pillar Two rules in additional jurisdictions.
40
Table of Contents
Liquidity and Capital Resources
Condensed Consolidated Statements of Cash Flows (in thousands):
| Year Ended December 31, | ||||||||||||
| 2023 | 2022 | 2021 | ||||||||||
| Net income |
$ | 642,234 | $ | 707,755 | $ | 692,843 | ||||||
| Depreciation and amortization |
165,905 | 130,423 | 131,680 | |||||||||
| Stock-based compensation |
36,868 | 42,564 | 29,918 | |||||||||
| Deferred income taxes |
(1,197 | ) | (31,988 | ) | 16,633 | |||||||
| Observable unrealized gain on investment |
— | — | (9,707 | ) | ||||||||
| Acquired in-process research and development and other non-cash items |
— | 10,003 | — | |||||||||
| Change in accounts receivable |
49,179 | (137,874 | ) | (62,448 | ) | |||||||
| Change in inventories |
(45,443 | ) | (101,902 | ) | (67,250 | ) | ||||||
| Change in accounts payable and other current liabilities |
(79,524 | ) | 60,984 | 46,110 | ||||||||
| Change in deferred revenue and customer advances |
10,433 | 12,862 | 37,845 | |||||||||
| Other changes |
(175,646 | ) | (81,166 | ) | (68,350 | ) | ||||||
|
|
|
|
|
|
|
|||||||
| Net cash provided by operating activities |
602,809 | 611,661 | 747,274 | |||||||||
| Net cash used in investing activities |
(1,442,265 | ) | (107,967 | ) | (231,630 | ) | ||||||
| Net cash used in financing activities |
754,951 | (509,633 | ) | (438,275 | ) | |||||||
| Effect of exchange rate changes on cash and cash equivalents |
(948 | ) | (14,766 | ) | (12,830 | ) | ||||||
|
|
|
|
|
|
|
|||||||
| (Decrease) increase in cash and cash equivalents |
$ | (85,453 | ) | $ | (20,705 | ) | $ | 64,539 | ||||
|
|
|
|
|
|
|
|||||||
Cash Flow from Operating Activities
Net cash provided by operating activities was $603 million, $612 million and $747 million in 2023, 2022 and 2021, respectively. The decrease in 2023 operating cash flow was primarily a result of lower net income, higher inventory levels, higher income tax payments and higher incentive compensation payments in 2023 as compared to 2022. The changes within net cash provided by operating activities include the following significant changes in the sources and uses of net cash provided by operating activities, in addition to the changes in net income:
| • | The changes in accounts receivable were primarily attributable to timing of payments made by customers and timing of sales. Days sales outstanding was 78 days at December 31, 2023, 77 days at December 31, 2022 and 66 days at December 31, 2021. |
| • | The increase in inventory can primarily be attributed to higher material costs as well as an increase in safety stock levels to help mitigate any future supply chain issues. |
| • | The changes in accounts payable and other current liabilities were a result of the timing of payments to vendors, as well as the annual payment of management incentive compensation. |
| • | An increase in income tax payments of $83 million as compared to the prior year and the payment of $26 million in Wyatt acquired liabilities. |
| • | Net cash provided from deferred revenue and customer advances results from annual increases in new service contracts as a higher installed base of customers renew annual service contracts. |
| • | Other changes were attributable to variation in the timing of various provisions, expenditures, prepaid income taxes and accruals in other current assets, other assets and other liabilities. |
Cash Flow from Investing Activities
Net cash used in investing activities totaled $1.4 billion, $108 million and $232 million in 2023, 2022 and 2021, respectively. Additions to fixed assets and capitalized software were $161 million, $176 million and $161 million
41
Table of Contents
in 2023, 2022 and 2021, respectively. The cash flows from investing activities in 2023, 2022 and 2021 include $16 million, $32 million and $49 million, respectively, of capital expenditures related to the major expansion of the Company’s precision chemistry consumable operations in the United States. The Company has incurred costs of $248 million on this new state-of-the-art facility, which is substantially complete as of December 31, 2023.
During 2023, 2022 and 2021, the Company purchased $2 million, $11 million and $280 million of investments, respectively, while $2 million, $78 million and $218 million of investments matured, respectively, and were used for financing activities described below.
In 2023, the Company completed the acquisition of Wyatt for a total purchase price of $1.3 billion in cash. Wyatt is a pioneer in innovative light scattering and field-flow fractionation instruments, software, accessories, and services. The acquisition will expand Waters’ portfolio and increase exposure to large molecule applications.
In 2022, the Company paid $5 million for the CDMS technology and intellectual property right asset from Megadalton, and the Company is required to make an additional $4 million of guaranteed payments at various dates in the future through 2029. The total purchase price of approximately $10 million was accounted for as Acquired In-Process Research and Development and expensed as part of costs and operating expenses in the statement of operations in 2023.
There were no business acquisitions in 2022 and 2021.
In 2022, the Company received $10 million in proceeds and made $1 million of investments in certain equity investments. In 2021, the Company made $2 million of investments in certain equity investments.
Cash Flow from Financing Activities
The Company entered into a credit agreement in September 2021 governing the Company’s five-year, $1.8 billion revolving facility that matures in September 2026. On March 3, 2023, in anticipation of closing of the Wyatt acquisition, the Company entered into an agreement to amend the credit agreement governing its revolving credit facility (the “2023 Amendment”). The 2023 Amendment increases the borrowing capacity by $200 million to an aggregate total borrowing capacity of $2.0 billion. As of December 31, 2023, the Company had a total of $2.4 billion in outstanding debt, which consisted of $1.3 billion in outstanding senior unsecured notes and $1.1 billion in borrowings under its credit agreement. The Company’s net debt borrowings as of December 31, 2023, 2022 and 2021 were $780 million, $60 million and $160 million higher than as of December 31, 2022, 2021 and 2020, respectively.
As of December 31, 2023, the Company has entered into three-year interest rate cross-currency swap derivative agreements with a notional value $625 million to hedge the variability in the movement of foreign currency exchange rates on a portion of its euro-denominated and yen-denominated net asset investments. As a result of entering into these agreements, the Company lowered net interest expense by approximately $11 million, $9 million and $15 million in 2023, 2022 and 2021, respectively. The Company anticipates that these swap agreements will lower net interest expense by approximately $7 million in 2024.
In December 2023, the Company’s Board of Directors authorized the extension of the existing share repurchase program through January 21, 2025. The Company’s remaining authorization is $1.0 billion. During 2023, 2022 and 2021, the Company repurchased $58 million, $616 million and $640 million, respectively, of the Company’s outstanding common stock under authorized share repurchase programs. In addition, the Company repurchased $12 million, $11 million and $9 million of common stock related to the vesting of restricted stock units during 2023, 2022 and 2021, respectively.
The Company received $30 million, $43 million and $56 million of proceeds from the exercise of stock options and the purchase of shares pursuant to the Company’s employee stock purchase plan during 2023, 2022 and 2021, respectively.
42
Table of Contents
The Company had cash, cash equivalents and investments of $396 million as of December 31, 2023. The majority of the Company’s cash and cash equivalents are generated from foreign operations, with $321 million held by foreign subsidiaries at December 31, 2023, of which $233 million was held in currencies other than U.S. dollars.
As of December 31, 2023, the Company’s material cash requirements include the following contractual and other obligations:
Long-term debt. As of December 31, 2023, the Company had $2.4 billion of cash requirements for the principal on long-term debt that will mature and be paid as follows: $50 million in 2024; $1.5 billion in 2026; $50 million in 2028; $300 million in 2029; $50 million in 2030 and $400 million in 2031.
Interest on Senior Unsecured Notes. As of December 31, 2023, the Company had $189 million of cash requirements for the interest on senior unsecured notes that is to be paid as follows: $39 million in 2024; $38 million in 2025; $32 million in 2026; $25 million in 2027; $23 million in 2028; $20 million in 2029; $10 million in 2030; and $2 million in 2031. See also Note 9 in the Notes to the Consolidated Financial Statements for financial information about interest payable.
2017 Tax Act liabilities. As a result of the 2017 Tax Act, the Company incurred a Transition Toll Tax, that would be paid over an eight-year period, starting in 2018, and will not accrue interest. As of December 31, 2023, the Company had a remaining cash requirement of $216 million of which $96 million and $120 million will be paid in 2024 and 2025, respectively. See also Note 10 in the Notes to the Consolidated Financial Statements for financial information about tax liabilities.
Operating Leases. The Company’s cash requirements for future lease payments were approximately $93 million as of December 31, 2023. See also Note 12 in the Notes to the Consolidated Financial Statements for financial information about lease liabilities.
Long-term Software Contract Commitments. For contracts the Company is committed to that are not cancelable without penalties. The Company’s contractual obligation with these vendors was approximately $22 million as of December 31, 2023.
Wyatt Retention Agreements. In conjunction with the Wyatt acquisition, the Company entered into retention agreements with certain employees, in which the Company agreed to pay a total of $40 million by the end of the second anniversary of the acquisition date provided the employees remain employed over that period of time.
Management believes, as of the date of this report, that the Company’s financial position, along with expected future cash flows from earnings based on historical trends and the ability to raise funds from external sources and the borrowing capacity from existing, committed credit facilities, will be sufficient to service debt and fund working capital and capital spending requirements, authorized share repurchase amounts and potential acquisitions for at least the next twelve months.
Critical Accounting Policies and Estimates
Summary
The preparation of consolidated financial statements requires the Company to make estimates and judgments that affect the reported amounts of assets, liabilities, revenues and expenses, and related disclosure of contingent liabilities. Critical accounting policies are those that are central to the presentation of the Company’s financial condition and results of operations that require management to make estimates about matters that are highly uncertain and that would have a material impact on the Company’s results of operations given changes in the estimate that are reasonably likely to occur from period to period or use of different estimates that reasonably could have been used in the current period. On an ongoing basis, the Company evaluates its policies and
43
Table of Contents
estimates. The Company bases its estimates on historical experience and on various other assumptions that are believed to be reasonable under the circumstances, the results of which form the basis for making judgments about the carrying values of assets and liabilities that are not readily apparent from other sources. Actual amounts may differ from these estimates under different assumptions or conditions. There are other items within the Company’s consolidated financial statements that require estimation, but are not deemed critical as defined above. Changes in estimates used in these and other items could potentially have a material impact on the Company’s consolidated financial statements.
Revenue Recognition
The Company recognizes revenue upon transfer of control of promised products and services to customers in an amount that reflects the consideration the Company expects to receive in exchange for those products or services. The Company generally enters into contracts that include a combination of products and services. Revenue is allocated to distinct performance obligations and is recognized net of allowances for returns and discounts.
The Company recognizes revenue on product sales at the time control of the product transfers to the customer. In substantially all of the Company’s arrangements, title of the product transfers at shipping point and, as a result, the Company determined control transfers at the point of shipment. In more limited cases, there are destination-based shipping terms and, thus, control is deemed to transfer when the products arrive at the customer site.
Generally, the Company’s contracts for products include a performance obligation related to installation. The Company has determined that the installation represents a distinct performance obligation and revenue is recognized separately upon the completion of installation. The Company determines the amount of the transaction price to allocate to the installation service based on the standalone selling price of the product and the service, which requires judgment. The Company determines the relative standalone selling price of installation based upon a number of factors, including hourly service billing rates and estimated installation hours. In developing these estimates, the Company considers past history, competition, billing rates of current services and other factors.
The Company has sales from standalone software, which are included in product revenue. These arrangements typically include software licenses and maintenance contracts, both of which the Company has determined are distinct performance obligations. The Company determines the amount of the transaction price to allocate to the license and maintenance contract based on the relative standalone selling price of each performance obligation. Software license revenue is recognized at the point in time when control has been transferred to the customer. The revenue allocated to the software maintenance contract is recognized on a straight-line basis over the maintenance period, which is the contractual term of the contract, as a time-based measure of progress best reflects the Company’s performance in satisfying this obligation. Unspecified rights to software upgrades are typically sold as part of the maintenance contract on a when-and-if-available basis.
Service revenue includes (i) service and software maintenance contracts and (ii) service calls (time and materials). Instrument service contracts and software maintenance contracts are typically annual contracts, which are billed at the beginning of the contract or maintenance period. The amount of the service and software maintenance contract is recognized on a straight-line basis to revenue over the maintenance service period, which is the contractual term of the contract, as a time-based measure of progress best reflects the Company’s performance in satisfying this obligation. There are no deferred costs associated with the service contract, as the cost of the service is recorded when the service is performed. Service calls are recognized to revenue at the time a service is performed.
The Company’s deferred revenue liabilities at December 31, 2023 of $324 million on the consolidated balance sheets consist of instrument service contract obligations and customer payments received in advance, prior to transfer of control of the instrument. The Company records deferred revenue primarily related to its service contracts, where consideration is billable at the beginning of the service period.
44
Table of Contents
Loss Provision on Inventory
The Company values all of its inventories at the lower of cost or net realizable value on a first-in, first-out basis (“FIFO”). The Company estimates revisions to its inventory valuations based on technical obsolescence, historical demand, projections of future demand, including in the Company’s current backlog of orders, and industry and market conditions. If actual future demand or market conditions are less favorable than those projected by management, additional write-downs may be required. The Company’s inventory balance at December 31, 2023 was recorded at its net realizable value of $516 million, which is net of write-downs of $41 million.
Long-Lived Assets, Intangible Assets and Goodwill
Goodwill and indefinite-lived intangible assets are not amortized, but are evaluated for impairment on an annual basis, or on an interim basis when events or changes in circumstances indicate that the carrying value may not be recoverable. In assessing the recoverability of goodwill and indefinite-lived intangible assets, we must make assumptions regarding the estimated future cash flows, including forecasted revenue growth and the discount rate to determine the fair value of these assets. If these estimates or their related assumptions change in the future, we may be required to record impairment charges against these assets in the reporting period in which the impairment is determined.
We test goodwill for impairment at the reporting unit level, which is the operating segment or one level below an operating segment. We have the option of performing a qualitative assessment to determine whether further impairment testing is necessary before performing the quantitative assessment. If as a result of the qualitative assessment, it is more-likely-than-not that the fair value of a reporting unit is less than its carrying amount, a quantitative impairment test will be required. Otherwise, no further testing will be required. If a quantitative impairment test is performed, we compare the fair values of the applicable reporting units with their aggregate carrying values, including goodwill. Estimating the fair value of the reporting units requires significant judgment by management. If the carrying amount of a reporting unit exceeds the fair value of the reporting unit, an impairment charge is recognized for the amount by which the carrying value amount exceeds the reporting unit’s fair value up to the total amount of goodwill allocated to the reporting unit. The Company performs an annual goodwill impairment assessment for its reporting units as of December 31 each year. The Company has two reporting units: Waters and TA. Goodwill is allocated to the reporting units at the time of acquisition.
The Company’s intangible assets include purchased technology; capitalized software; costs associated with acquiring Company patents, trademarks and intellectual properties, such as licenses; and acquired IPR&D. Purchased intangibles are recorded at their fair market values as of the acquisition date and amortized over their estimated useful lives, ranging from one to fifteen years. Other intangibles are amortized over a period ranging from one to ten years. Acquired IPR&D is amortized from the date of completion of the acquired program over its estimated useful life.
Goodwill totaled $1.3 billion and $430 million as of December 31, 2023 and 2022, respectively. Net intangible assets and long-lived assets amounted to $629 million and $639 million, as of December 31, 2023, respectively, and $227 million and $582 million as of December 31, 2022, respectively.
Income Taxes
As part of the process of preparing the consolidated financial statements, the Company is required to estimate its income taxes in each of the jurisdictions in which it operates. This process involves the Company estimating its income taxes, taking into account the amount, timing and character of taxable income, tax deductions and credits and assessing changes in tax laws, regulations, agreements and treaties. Differing treatment of items for tax and accounting purposes, such as depreciation, amortization and inventory reserves, result in deferred tax assets and liabilities, which are included within the consolidated balance sheets. In the event that actual results differ from these estimates, or the Company adjusts these estimates in future periods, such changes could materially impact the Company’s financial position and results of operations.
45
Table of Contents
The Company continually evaluates the necessity of establishing or changing a valuation allowance for deferred tax assets depending on whether it is more likely than not that the actual benefit of those assets will be realized in future periods.
Uncertain Tax Positions
The Company accounts for its uncertain tax return positions in accordance with the accounting standards for income taxes, which require financial statement reporting of the expected future tax consequences of uncertain tax positions on the presumption that all concerned tax authorities possess full knowledge of those tax positions, as well as all of the pertinent facts and circumstances, but prohibit any discounting of unrecognized tax benefits associated with those positions for the time value of money. The Company classified interest and penalties related to unrecognized tax benefits as a component of the provision for income taxes. At December 31, 2023, the Company had unrecognized tax benefits, excluding interest and penalties, of $14 million, which represents a decrease of $15 million resulting, primarily, from the completion of a tax examination in 2023. This decrease reduced the income tax expense in the statement of operations and did not impact cash flows.
The Company has a new Development and Expansion Incentive in Singapore that provides a concessionary income tax rate of 5% on certain types of income for the period April 1, 2021 through March 31, 2026. This new incentive has similar requirements for business spending targets, attaining and sustaining employment targets and performance of certain research and manufacturing activities as previous agreements. Prior to April 1, 2021, the Company had a tax exemption on income arising from qualifying activities in Singapore, based upon the achievement of certain contractual milestones, which the Company met as of December 31, 2020 and maintained through March 2021. These milestones include the following types of objectives: reaching and maintaining annual revenue and business spending targets; meeting capital expenditures targets; attaining and sustaining employment targets; and establishing a local research and development and service center. The Company determined that it was more likely than not to realize the tax exemption in Singapore and, accordingly, did not recognize any reserves for unrecognized tax benefits on its balance sheet related to this exemption. In the event that any of the milestone targets were not met, the Company would not be entitled to the tax exemption on income earned in Singapore and all the tax benefits previously recognized would be reversed, resulting in the recognition of income tax expense equal to the statutory tax of 17% on income earned during that period.
The effect of applying these concessionary income tax rates rather than the statutory tax rate to income arising from qualifying activities in Singapore increased the Company’s net income by $16 million, $20 million and $20 million and increased the Company’s net income per diluted share by $0.27, $0.33 and $0.32 for the years ended December 31, 2023, 2022 and 2021, respectively.
Business Combinations and Asset Acquisitions
We use assumptions and estimates in determining the fair value of assets acquired and liabilities assumed in a business combination. The determination of the fair value of intangible assets, which represents a significant portion of the purchase price in our recent acquisition of Wyatt, requires the use of significant judgment with regard to (i) the fair value; and (ii) whether such intangibles are amortizable or non-amortizable and, if the former, the period and the method by which the intangible asset will be amortized. We utilize commonly accepted valuation techniques, such as the income, cost and market approaches, as appropriate, in establishing the fair value of intangible assets. Typically, key assumptions include projections of cash flows that arise from identifiable intangible assets of acquired businesses as well as discount rates based on an analysis of the weighted average cost of capital, adjusted for specific risks associated with the assets.
In our recent acquisition of Wyatt, customer relationship intangible assets have been the most significant identifiable assets acquired. The customer relationships were valued using the multi-period excess earnings method under the income approach. Our cash flow projections for the customer relationships acquired included significant judgments and assumptions related to customer attrition rate, discount rate, and forecasted revenues. The value of the client relationships acquired was $331 million in fiscal year 2023, the majority of which relates to US customer relationships.
46
Table of Contents
Recent Accounting Standard Changes and Developments
Information regarding recent accounting standard changes and developments is incorporated by reference from Part II, Item 8, Financial Statements and Supplementary Data, of this document and should be considered an integral part of this Item 7. See Note 2 in the Notes to the Consolidated Financial Statements for recently adopted and issued accounting standards.
Item 7A: Quantitative and Qualitative Disclosures About Market Risk
Derivative Transactions
The Company is a global company that operates in over 35 countries and, as a result, the Company’s net sales, cost of sales, operating expenses and balance sheet amounts are significantly impacted by fluctuations in foreign currency exchange rates. The Company is exposed to currency price risk on foreign currency exchange rate fluctuations when it translates its non-U.S. dollar foreign subsidiaries’ financial statements into U.S. dollars, and when any of the Company’s subsidiaries purchase or sell products or services in a currency other than its own currency.
The Company’s principal strategies in managing exposures to changes in foreign currency exchange rates are to (1) naturally hedge the foreign-currency-denominated liabilities on the Company’s balance sheet against corresponding assets of the same currency, such that any changes in liabilities due to fluctuations in foreign currency exchange rates are typically offset by corresponding changes in assets and (2) mitigate foreign exchange risk exposure of international operations by hedging the variability in the movement of foreign currency exchange rates on a portion of its euro-denominated and yen-denominated net asset investments. The Company presents the derivative transactions in financing activities in the statement of cash flows.
Foreign Currency Exchange Contracts
The Company does not specifically enter into any derivatives that hedge foreign-currency-denominated operating assets, liabilities or commitments on its balance sheet, other than a portion of certain third-party accounts receivable and accounts payable, and the Company’s net worldwide intercompany receivables and payables, which are eliminated in consolidation. The Company periodically aggregates these net worldwide balances by currency and then enters into foreign currency exchange contracts that mature within 90 days to hedge a portion of the remaining balance to minimize some of the Company’s currency price risk exposure. The foreign currency exchange contracts are not designated for hedge accounting treatment. Principal hedged currencies include the euro, Japanese yen, British pound, Mexican peso and Brazilian real.
Cash Flow Hedges
The Company’s Credit Facility is a variable borrowing and has interest payments based on a contractually specified interest rate index. The contractually specified index on the Credit Facility is the 3-month Term SOFR. The variable rate interest payments create interest risk for the Company as interest payments will fluctuate based on changes in the contractually specified interest rate index over the life of the Credit Facility. In order to reduce interest rate risk, the Company enters into interest rate swaps that will effectively lock-in the forecasted interest payments on the variable rate borrowing over its term. The interest rate swaps represent cash flow hedges and are assessed for hedge effectiveness each reporting period. When the hedge relationship is highly effective at achieving offsetting changes in cash flows, the Company will record the entire change in fair value of the interest rate swaps in accumulated other comprehensive loss. The amount in accumulated other comprehensive loss is reclassified to earnings in the period that the underlying transaction impacts consolidated earnings. If it becomes probable that the forecasted transaction will not occur, the hedge relationship will be de-designated and amounts accumulated in other comprehensive loss will be reclassified to earnings in the current period. Interest settlements due to benchmark interest rate changes are recorded in interest income or interest expense. For the year ended December 31, 2023, the Company did not have any cash flow hedges that were deemed ineffective.
47
Table of Contents
Interest Rate Cross-Currency Swap Agreements
As of December 31, 2023, the Company had three-year interest rate cross-currency swap derivative agreements with a notional value of $625 million to hedge the variability in the movement of foreign currency exchange rates on a portion of its euro-denominated and yen-denominated net asset investments. Under hedge accounting, the change in fair value of the derivative that relates to changes in the foreign currency spot rate are recorded in the currency translation adjustment in other comprehensive income and remain in accumulated other comprehensive income in stockholders’ equity until the sale or substantial liquidation of the foreign operation. The difference between the interest rate received and paid under the interest rate cross-currency swap derivative agreement is recorded in interest income in the statement of operations.
The Company’s foreign currency exchange contracts, interest rate cross-currency swap agreements and interest rate swap agreements designated as cash flow hedges are included in the consolidated balance sheets are classified as follows (in thousands):
| December 31, 2023 | December 31, 2022 | |||||||||||||||
| Notional Value |
Fair Value |
Notional Value |
Fair Value |
|||||||||||||
| Foreign currency exchange contracts: |
||||||||||||||||
| Other current assets |
$ | 24,155 | $ | 183 | $ | 42,047 | $ | 231 | ||||||||
| Other current liabilities |
$ | 16,000 | $ | 207 | $ | 13,450 | $ | 98 | ||||||||
| Interest rate cross-currency swap agreements: |
||||||||||||||||
| Other assets |
$ | 220,000 | $ | 4,835 | $ | 400,000 | $ | 19,163 | ||||||||
| Other liabilities |
$ | 405,000 | $ | 13,384 | $ | 185,000 | $ | 4,783 | ||||||||
| Accumulated other comprehensive (loss) income |
$ | (7,975 | ) | $ | 10,026 | |||||||||||
| Interest rate swap cash flow hedges: |
||||||||||||||||
| Other liabilities |
$ | 100,000 | $ | 2,974 | $ | — | $ | — | ||||||||
| Accumulated other comprehensive (loss) income |
$ | (2,974 | ) | $ | — | |||||||||||
The following is a summary of the activity included in the consolidated statements of operations and statements of comprehensive income related to the foreign currency exchange contracts, and interest rate cross-currency swap agreements and interest rate swap agreements designated as cash flow hedges (in thousands):
| Financial Statement Classification |
Year Ended December 31, | |||||||||||||
| 2023 | 2022 | 2021 | ||||||||||||
| Foreign currency exchange contracts: |
||||||||||||||
| Realized gains (losses) on closed contracts |
Cost of sales | $ | 224 | $ | (3,855 | ) | $ | (1,973 | ) | |||||
| Unrealized losses on open contracts |
Cost of sales | (156 | ) | (176 | ) | (343 | ) | |||||||
|
|
|
|
|
|
|
|||||||||
| Cumulative net pre-tax gains (losses) |
Cost of sales | $ | 68 | $ | (4,031 | ) | $ | (2,316 | ) | |||||
|
|
|
|
|
|
|
|||||||||
| Interest rate cross-currency swap agreements: |
||||||||||||||
| Interest earned |
Interest income | $ | 10,974 | $ | 8,872 | $ | 11,084 | |||||||
| Unrealized (losses) gains on open contracts |
Accumulated other comprehensive loss |
$ | (18,001 | ) | $ | 25,969 | $ | 29,052 | ||||||
| Interest rate swap cash flow hedges: |
||||||||||||||
| Interest earned |
Interest income | $ | 326 | $ | — | $ | — | |||||||
| Unrealized losses on open contracts |
Accumulated other comprehensive loss |
$ | (2,974 | ) | $ | — | $ | — | ||||||
Assuming a hypothetical adverse change of 10% in year-end exchange rates (a strengthening of the U.S. dollar), the fair market value of the foreign currency exchange contracts outstanding as of December 31, 2023 would increase pre-tax earnings by approximately $5 million. Assuming a hypothetical adverse change of 10% in year-end exchange rates (a strengthening of the U.S. dollar), the fair market value of the interest rate
48
Table of Contents
cross-currency swap agreements outstanding as of December 31, 2023 would increase by approximately $1 million and would be recorded to foreign currency translation in other comprehensive income within stockholders’ equity. The related impact on interest income would not have a material effect on pre-tax earnings.
The Company’s cash and cash equivalents are not subject to significant interest rate risk due to the short maturities of these instruments. The Company’s cash equivalents represent highly liquid investments, with original maturities of 90 days or less, primarily in bank deposits, U.S. treasury bill money market funds and commercial paper. As of December 31, 2023, the carrying value of the Company’s cash and cash equivalents approximated fair value.
The Company is exposed to the risk of interest rate fluctuations from the investments of cash generated from operations. Investments with maturities greater than 90 days are classified as investments, and are held primarily in U.S. dollar-denominated treasury bills and commercial paper, bank deposits and corporate debt securities. As of December 31, 2023, the Company estimates that a hypothetical adverse change of 100 basis points across all maturities would not have a material effect on the fair market value of its portfolio.
The Company is also exposed to the risk of exchange rate fluctuations. The Company maintains cash balances in various operating accounts in excess of federally insured limits, and in foreign subsidiary accounts in currencies other than the U.S. dollar. As of December 31, 2023 and 2022, $321 million out of $396 million and $472 million out of $481 million, respectively, of the Company’s total cash, cash equivalents and investments were held by foreign subsidiaries. In addition, $233 million out of $396 million and $336 million out of $481 million of cash, cash equivalents and investments were held in currencies other than the U.S. dollar at December 31, 2023 and 2022, respectively. As of December 31, 2023, the Company had no holdings in auction rate securities or commercial paper issued by structured investment vehicles.
Assuming a hypothetical adverse change of 10% in year-end exchange rates (a strengthening of the U.S. dollar), the fair market value of the Company’s cash, cash equivalents and investments held in currencies other than the U.S. dollar as of December 31, 2023 would decrease by approximately $23 million, of which the majority would be recorded to foreign currency translation in other comprehensive income within stockholders’ equity.
49
Table of Contents
| /s/ |
| February 27, 2024 |
December 31, |
||||||||
2023 |
2022 |
|||||||
(In thousands, except per share data) |
||||||||
ASSETS |
||||||||
Current assets: |
||||||||
Cash and cash equivalents |
$ | $ | ||||||
Investments |
||||||||
Accounts receivable, net |
||||||||
Inventories |
||||||||
Other current assets |
||||||||
Total current assets |
||||||||
Property, plant and equipment, net |
||||||||
Intangible assets, net |
||||||||
Goodwill |
||||||||
Operating lease assets |
||||||||
Other assets |
||||||||
Total assets |
$ | $ | ||||||
LIABILITIES AND STOCKHOLDERS’ EQUITY |
||||||||
Current liabilities: |
||||||||
Notes payable and debt |
$ | $ | ||||||
Accounts payable |
||||||||
Accrued employee compensation |
||||||||
Deferred revenue and customer advances |
||||||||
Current operating lease liabilities |
||||||||
Accrued income taxes |
||||||||
Accrued warranty |
||||||||
Other current liabilities |
||||||||
Total current liabilities |
||||||||
Long-term liabilities: |
||||||||
Long-term debt |
||||||||
Long-term portion of retirement benefits |
||||||||
Long-term income tax liabilities |
||||||||
Long-term operating lease liabilities |
||||||||
Other long-term liabilities |
||||||||
Total long-term liabilities |
||||||||
Total liabilities |
||||||||
Commitments and contingencies (Notes 9, 10, 11, 12, 13 and 17 ) |
||||||||
Stockholders’ equity: |
||||||||
Preferred stock, par value $ |
||||||||
Common stock, par value $ |
||||||||
Additional paid-in capital |
||||||||
Retained earnings |
||||||||
Treasury stock, at cost, |
( |
) | ( |
) | ||||
Accumulated other comprehensive loss |
( |
) | ( |
) | ||||
Total stockholders’ equity |
||||||||
Total liabilities and stockholders’ equity |
$ | $ | ||||||
Year Ended December 31, |
||||||||||||
2023 |
2022 |
2021 |
||||||||||
(In thousands, except per share data) |
||||||||||||
Revenues: |
||||||||||||
Product sales |
$ | $ | $ | |||||||||
Service sales |
||||||||||||
Total net sales |
||||||||||||
Costs and operating expenses: |
||||||||||||
Cost of product sales |
||||||||||||
Cost of service sales |
||||||||||||
Selling and administrative expenses |
||||||||||||
Research and development expenses |
||||||||||||
Purchased intangibles amortization |
||||||||||||
Litigation provision |
||||||||||||
Acquired in-process research and development |
— | |||||||||||
Total costs and operating expenses |
||||||||||||
Operating income |
||||||||||||
Other income, net |
||||||||||||
Interest expense |
( |
) | ( |
) | ( |
) | ||||||
Interest income |
||||||||||||
Income before income taxes |
||||||||||||
Provision for income taxes |
||||||||||||
Net income |
$ | $ | $ | |||||||||
Net income per basic common share |
$ | $ | $ | |||||||||
Weighted-average number of basic common shares |
||||||||||||
Net income per diluted common share |
$ | $ | $ | |||||||||
Weighted-average number of diluted common shares and equivalents |
||||||||||||
Year Ended December 31, |
||||||||||||
2023 |
2022 |
2021 |
||||||||||
(In thousands) |
||||||||||||
| Net income |
$ | $ | $ | |||||||||
| Other comprehensive income (loss): |
||||||||||||
| Foreign currency translation |
( |
) | ( |
) | ||||||||
| Unrealized losses on derivative instruments before reclassifications |
( |
) | ||||||||||
| Amounts reclassified to interest income |
( |
) | ||||||||||
| |
|
|
|
|
|
|||||||
| Unrealized losses on derivative instruments before income taxes |
( |
) | ||||||||||
| Income tax benefit |
||||||||||||
| |
|
|
|
|
|
|||||||
| Unrealized losses on derivative instruments, net of tax |
( |
) | ||||||||||
| Unrealized gains (losses) on investments before income taxes |
( |
) | ||||||||||
| Income tax (expense) benefit |
( |
) | ||||||||||
| |
|
|
|
|
|
|||||||
| Unrealized gains (losses) on investments, net of tax |
( |
) | ||||||||||
| Retirement liability adjustment before reclassifications |
( |
) | ||||||||||
| Amounts reclassified to other income, net |
( |
) | ||||||||||
| |
|
|
|
|
|
|||||||
| Retirement liability adjustment before income taxes |
( |
) | ||||||||||
| Income tax benefit (expense) |
( |
) | ( |
) | ||||||||
| |
|
|
|
|
|
|||||||
| Retirement liability adjustment, net of tax |
( |
) | ||||||||||
| Other comprehensive income (loss) |
( |
) | ||||||||||
| |
|
|
|
|
|
|||||||
| Comprehensive income |
$ | $ | $ | |||||||||
| |
|
|
|
|
|
|||||||
Year Ended December 31, |
||||||||||||
2023 |
2022 |
2021 |
||||||||||
(In thousands) |
||||||||||||
| Cash flows from operating activities: |
||||||||||||
| Net income |
$ | $ | $ | |||||||||
| Adjustments to reconcile net income to net cash provided by operating activities: |
||||||||||||
| Stock-based compensation |
||||||||||||
| Deferred income taxes |
( |
) | ( |
) | ||||||||
| Depreciation |
||||||||||||
| Amortization of intangibles |
||||||||||||
| Observable unrealized gain on investment |
( |
) | ||||||||||
| Realized gain on sale of investment |
( |
) | ||||||||||
| In-process research and development and other non-cash charges |
||||||||||||
| Change in operating assets and liabilities, net of acquisitions: |
||||||||||||
| Decrease (increase) in accounts receivable |
( |
) | ( |
) | ||||||||
| Increase in inventories |
( |
) | ( |
) | ( |
) | ||||||
| Increase in other current assets |
( |
) | ( |
) | ( |
) | ||||||
| (Increase) decrease in other assets |
( |
) | ( |
) | ||||||||
| (Decrease) increase in accounts payable and other current liabilities |
( |
) | ||||||||||
| Increase in deferred revenue and customer advances |
||||||||||||
| Decrease in other liabilities |
( |
) | ( |
) | ( |
) | ||||||
| |
|
|
|
|
|
|||||||
| Net cash provided by operating activities |
||||||||||||
| Cash flows from investing activities: |
||||||||||||
| Additions to property, plant, equipment and software capitalization |
( |
) | ( |
) | ( |
) | ||||||
| Asset and business acquisitions, net of cash acquired |
( |
) | ||||||||||
| Proceeds from (investments in) equity investments, net |
( |
) | ||||||||||
| Payments for intellectual property licenses |
( |
) | ( |
) | ||||||||
| Purchases of investments |
( |
) | ( |
) | ( |
) | ||||||
| Maturities and sales of investments |
||||||||||||
| |
|
|
|
|
|
|||||||
| Net cash used in investing activities |
( |
) | ( |
) | ( |
) | ||||||
| Cash flows from financing activities: |
||||||||||||
| Proceeds from debt issuances |
||||||||||||
| Payments on debt |
( |
) | ( |
) | ( |
) | ||||||
| Payments of debt issuance costs |
( |
) | ( |
) | ||||||||
| Proceeds from stock plans |
||||||||||||
| Purchases of treasury shares |
( |
) | ( |
) | ( |
) | ||||||
| Proceeds from derivative contracts |
||||||||||||
| |
|
|
|
|
|
|||||||
| Net cash provided by (used in) financing activities |
( |
) | ( |
) | ||||||||
| Effect of exchange rate changes on cash and cash equivalents |
( |
) | ( |
) | ( |
) | ||||||
| |
|
|
|
|
|
|||||||
| (Decrease) increase in cash and cash equivalents |
( |
) | ( |
) | ||||||||
| Cash and cash equivalents at beginning of period |
||||||||||||
| |
|
|
|
|
|
|||||||
| Cash and cash equivalents at end of period |
$ | $ | $ | |||||||||
| |
|
|
|
|
|
|||||||
| Supplemental cash flow information: |
||||||||||||
| Income taxes paid |
$ | $ | $ | |||||||||
| Interest paid |
$ | $ | $ | |||||||||
Number of Common Shares |
Common Stock |
Additional Paid-In Capital |
Retained Earnings |
Treasury Stock |
Accumulated Other Comprehensive Loss |
Total Stockholders’ Equity |
||||||||||||||||||||||
(In thousands) |
||||||||||||||||||||||||||||
Balance December 31, 2020 |
$ | $ | $ | $ | ( |
) | $ | ( |
) | $ | ||||||||||||||||||
Net income |
— | — | — | — | — | |||||||||||||||||||||||
Other comprehensive income |
— | — | — | — | — | |||||||||||||||||||||||
Issuance of common stock for employees: |
||||||||||||||||||||||||||||
Employee Stock Purchase Plan |
— | — | — | — | ||||||||||||||||||||||||
Stock options exercised |
— | — | — | |||||||||||||||||||||||||
Treasury stock |
— | — | — | — | ( |
) | — | ( |
) | |||||||||||||||||||
Stock-based compensation |
— | — | — | |||||||||||||||||||||||||
Balance December 31, 2021 |
$ | $ | $ | $ | ( |
) | $ | ( |
) | $ | ||||||||||||||||||
Net income |
— | — | — | — | — | |||||||||||||||||||||||
Other comprehensive loss |
— | — | — | — | — | ( |
) | ( |
) | |||||||||||||||||||
Issuance of common stock for employees: |
||||||||||||||||||||||||||||
Employee Stock Purchase Plan |
— | — | — | — | ||||||||||||||||||||||||
Stock options exercised |
— | — | — | |||||||||||||||||||||||||
Treasury stock |
— | — | — | — | ( |
) | — | ( |
) | |||||||||||||||||||
Stock-based compensation |
— | — | — | |||||||||||||||||||||||||
Balance December 31, 2022 |
$ | $ | $ | $ | ( |
) | $ | ( |
) | $ | ||||||||||||||||||
Net income |
— | — | — | — | — | |||||||||||||||||||||||
Other comprehensive income |
— | — | — | — | — | |||||||||||||||||||||||
Issuance of common stock for employees: |
||||||||||||||||||||||||||||
Employee Stock Purchase Plan |
— | — | — | — | ||||||||||||||||||||||||
Stock options exercise d |
— | — | — | |||||||||||||||||||||||||
Treasury stock |
— | — | — | — | ( |
) | — | ( |
) | |||||||||||||||||||
Stock-based compensation |
— | — | — | |||||||||||||||||||||||||
Balance December 31, 2023 |
$ | $ | $ | $ | ( |
) | $ | ( |
) | $ | ||||||||||||||||||
Balance at Beginning of Period |
Additions |
Deductions |
Balance at End of Period |
|||||||||||||
| Allowance for Credit Losses |
||||||||||||||||
| December 31, 2023 |
$ | $ | $ | ( |
) | $ | ||||||||||
| December 31, 2022 |
$ | $ | $ | ( |
) | $ | ||||||||||
| December 31, 2021 |
$ | $ | $ | ( |
) | $ | ||||||||||
Total at December 31, 2023 |
Quoted Prices in Active Markets for Identical Assets (Level 1) |
Significant Other Observable Inputs (Level 2) |
Significant Unobservable Inputs (Level 3) |
|||||||||||||
Assets: |
||||||||||||||||
Time deposits |
$ | $ | — | $ | $ | — | ||||||||||
Waters 401(k) Restoration Plan assets |
— | — | ||||||||||||||
Foreign currency exchange contracts |
— | — | ||||||||||||||
Interest rate cross-currency swap agreements |
— | — | ||||||||||||||
Total |
$ | $ | $ | $ | ||||||||||||
Liabilities: |
||||||||||||||||
Foreign currency exchange contracts |
$ | $ | — | $ | $ | — | ||||||||||
Interest rate cross-currency swap agreements |
— | — | ||||||||||||||
Interest rate swap cash flow hedge |
— | — | ||||||||||||||
Total |
$ | $ | $ | $ | ||||||||||||
Total at December 31, 2022 |
Quoted Prices in Active Markets for Identical Assets (Level 1) |
Significant Other Observable Inputs (Level 2) |
Significant Unobservable Inputs (Level 3) |
|||||||||||||
Assets: |
||||||||||||||||
Time deposits |
$ | $ | — | $ | $ | — | ||||||||||
Waters 401(k) Restoration Plan assets |
— | — | ||||||||||||||
Foreign currency exchange contracts |
— | — | ||||||||||||||
Interest rate cross-currency swap agreements |
— | — | ||||||||||||||
Total |
$ | $ | $ | $ | ||||||||||||
Liabilities: |
||||||||||||||||
Contingent consideration |
$ | $ | — | $ | — | $ | ||||||||||
Foreign currency exchange contracts |
— | — | ||||||||||||||
Interest rate cross-currency swap agreements |
— | — | ||||||||||||||
Total |
$ | $ | $ | $ | ||||||||||||
December 31, 2023 |
December 31, 2022 |
|||||||||||||||
Notional Value |
Fair Value |
Notional Value |
Fair Value |
|||||||||||||
Foreign currency exchange contracts: |
||||||||||||||||
Other current assets |
$ | $ | $ | $ | ||||||||||||
Other current liabilities |
$ | $ | $ | $ | ||||||||||||
Interest rate cross-currency swap agreements: |
||||||||||||||||
Other assets |
$ | $ | $ | $ | ||||||||||||
Other liabilities |
$ | $ | $ | $ | ||||||||||||
Accumulated other comprehensive (loss) income |
$ | ( |
) | $ | ||||||||||||
Interest rate swap cash flow hedges: |
||||||||||||||||
Other liabilities |
$ | $ | $ | — | $ | — | ||||||||||
Accumulated other comprehensive (loss) income |
$ | ( |
) | $ | — | |||||||||||
Financial Statement Classification |
Year Ended December 31, |
|||||||||||||||
2023 |
2022 |
2021 |
||||||||||||||
Foreign currency exchange contracts: |
||||||||||||||||
Realized gains (losses) on closed contracts |
Cost of sales | $ |
$ |
( |
) | $ |
( |
) | ||||||||
Unrealized losses on open contracts |
Cost of sales | ( |
) | ( |
) | ( |
) | |||||||||
Cumulative net pre-tax gains (losses) |
Cost of sales | $ | $ | ( |
) | $ | ( |
) | ||||||||
Interest rate cross-currency swap agreements: |
||||||||||||||||
Interest earned |
Interest income | $ | $ | $ | ||||||||||||
Unrealized (losses) gains on open contracts |
Accumulated other comprehensive loss | $ | ( |
) | $ | $ | ||||||||||
Interest rate swap cash flow hedges: |
||||||||||||||||
Interest earned |
Interest income | $ | $ | — | $ | — | ||||||||||
Unrealized losses on open contracts |
Accumulated other comprehensive loss | $ | ( |
) | $ | — | $ | — | ||||||||
Balance at Beginning of Period |
Accruals for Warranties |
Settlements Made |
Balance at End of Period |
|||||||||||||
Accrued warranty liability: |
||||||||||||||||
December 31, 2023 |
$ | $ | $ | ( |
) | $ | ||||||||||
December 31, 2022 |
$ | $ | $ | ( |
) | $ | ||||||||||
December 31, 2021 |
$ | $ | $ | ( |
) | $ | ||||||||||
December 31, |
||||||||||||
2023 |
2022 |
2021 |
||||||||||
Balance at the beginning of the period |
$ | $ | $ | |||||||||
Recognition of revenue included in balance at beginning of the period |
( |
) | ( |
) | ( |
) | ||||||
Revenue deferred during the period, net of revenue recognized |
||||||||||||
Balance at the end of the period |
$ | $ | $ | |||||||||
December 31, 2023 |
||||
Deferred revenue and customer advances expected to be recognized in: |
||||
| $ | ||||
Total |
$ | |||
December 31, 2023 |
December 31, 2022 |
|||||||
Raw materials |
$ | $ | ||||||
Work in progress |
||||||||
Finished goods |
||||||||
Total inventories |
$ | $ | ||||||
December 31, |
||||||||
2023 |
2022 |
|||||||
Land and land improvements |
$ | $ | ||||||
Buildings and leasehold improvements |
||||||||
Production and other equipment |
||||||||
Construction in progress |
||||||||
Total property, plant and equipment |
||||||||
Less: accumulated depreciation and amortization |
( |
) | ( |
) | ||||
Property, plant and equipment, net |
$ | $ | ||||||
| Purchase Price |
||||
| Cash paid |
$ | |||
| Less: cash acquired |
( |
) | ||
| |
|
|||
| Net cash consideration |
||||
| |
|
|||
| Identifiable Net Assets (Liabilities) Acquired |
||||
| Accounts receivable |
||||
| Inventory |
||||
| Deferred tax assets |
||||
| Prepaid and other assets |
||||
| Property, plant and equipment |
||||
| Operating lease assets |
||||
| Intangible assets |
||||
| Accounts payable and accrued expenses |
( |
) | ||
| Operating lease liabilities |
( |
) | ||
| Tax liabilities |
( |
) | ||
| Deferred revenue |
( |
) | ||
| Other liabilities |
( |
) | ||
| |
|
|||
| Total identifiable net assets acquired |
||||
| Goodwill |
||||
| |
|
|||
| Cash consideration paid |
$ | |||
| |
|
Amount |
Weighted-Average Life |
|||||||
| Developed technology |
$ | |||||||
| Customer relationships |
||||||||
| Trade name |
||||||||
| |
|
|||||||
| Total |
$ |
|||||||
| |
|
|||||||
December 31, 2023 |
December 31, 2022 |
|||||||
| Revenue |
$ | $ | ||||||
| Net income |
||||||||
December 31, 2023 |
December 31, 2022 |
|||||||||||||||||||||||
Gross Carrying Amount |
Accumulated Amortization |
Weighted- Average Amortization Period |
Gross Carrying Amount |
Accumulated Amortization |
Weighted- Average Amortization Period |
|||||||||||||||||||
Capitalized software |
$ | $ | $ | $ | ||||||||||||||||||||
Purchased intangibles |
||||||||||||||||||||||||
Trademarks |
— | — | — | — | ||||||||||||||||||||
Licenses |
||||||||||||||||||||||||
Patents and other intangibles |
||||||||||||||||||||||||
Total |
$ | $ | $ | $ | ||||||||||||||||||||
Senior Unsecured Notes |
Term |
Interest Rate |
Face Value (in millions) |
Maturity Date | ||||
Series P |
$ |
2028 | ||||||
Series Q |
$ |
2030 |
December 31, 2023 |
December 31, 2022 |
|||||||
Senior unsecured notes - Series I - |
$ |
— | $ |
|||||
Senior unsecured notes - Series G - |
— | |||||||
Total notes payable and debt, current |
||||||||
Senior unsecured notes - Series G - |
— | |||||||
Senior unsecured notes - Series H - floating rate*, due June 2024 |
— | |||||||
Senior unsecured notes - Series K - |
||||||||
Senior unsecured notes - Series L - |
||||||||
Senior unsecured notes - Series M - |
||||||||
Senior unsecured notes - Series N - |
||||||||
Senior unsecured notes - Series O - |
||||||||
Senior unsecured notes - Series P - |
— | |||||||
Senior unsecured notes - Series Q - |
— | |||||||
Credit agreement |
||||||||
Unamortized debt issuance costs |
( |
) | ( |
) | ||||
Total long-term debt |
||||||||
Total debt |
$ | $ | ||||||
| * | Series H senior unsecured notes bear interest at a 3-month LIBOR for that floating rate interest period plus |
Total |
||||
2024 |
$ | |||
2025 |
||||
2026 |
||||
2027 |
||||
2028 |
||||
Thereafter |
||||
Total |
$ | |||
Year Ended December 31, |
||||||||||||
2023 |
2022 |
2021 |
||||||||||
The components of income before income taxes are as follows: |
||||||||||||
Domestic |
$ | $ | $ | |||||||||
Foreign |
||||||||||||
Total |
$ | $ | $ | |||||||||
Year Ended December 31, |
||||||||||||
2023 |
2022 |
2021 |
||||||||||
The components of the income tax provision were as follows: |
||||||||||||
Federal |
$ | $ | $ | |||||||||
State |
||||||||||||
Foreign |
||||||||||||
Total current tax provision |
$ | $ | $ | |||||||||
Federal |
$ | ( |
) | $ | ( |
) | $ | |||||
State |
( |
) | ( |
) | ||||||||
Foreign |
( |
) | ||||||||||
Total deferred tax provision |
( |
) | ( |
) | ||||||||
Total provision |
$ | $ | $ | |||||||||
Year Ended December 31, |
||||||||||||
2023 |
2022 |
2021 |
||||||||||
Federal tax computed at U.S. statutory income tax rate |
$ | $ | $ | |||||||||
GILTI, net of foreign tax credits |
||||||||||||
Uncertain tax positions |
( |
) | ||||||||||
State income tax, net of federal income tax benefit |
||||||||||||
Net effect of foreign operations |
( |
) | ( |
) | ( |
) | ||||||
Effect of stock-based compensation |
( |
) | ( |
) | ( |
) | ||||||
Other, net |
( |
) | ( |
) | ( |
) | ||||||
Provision for income taxes |
$ | $ | $ | |||||||||
December 31, |
||||||||
2023 |
2022 |
|||||||
Deferred tax assets: |
||||||||
Net operating losses and credits |
$ | $ | ||||||
Depreciation |
||||||||
Operating leases |
||||||||
Amortization |
||||||||
Stock-based compensation |
||||||||
Deferred compensation |
||||||||
Deferred revenue |
||||||||
Revaluation of equity investments and licenses |
||||||||
Inventory |
||||||||
Accrued liabilities and reserves |
||||||||
Capitalized interest |
||||||||
Unrealized foreign currency gain/loss |
||||||||
Capitalized Section 174 Expenditures |
||||||||
Other |
||||||||
Total deferred tax assets |
||||||||
Valuation allowance |
( |
) | ( |
) | ||||
Deferred tax assets, net of valuation allowance |
||||||||
| Deferred tax liabilities: |
||||||||
Capitalized software |
( |
) | ( |
) | ||||
Operating leases |
( |
) | ( |
) | ||||
Indefinite-lived intangibles |
( |
) | ( |
) | ||||
Deferred tax liability on foreign earnings |
( |
) | ( |
) | ||||
Total deferred tax liabilities |
( |
) | ( |
) | ||||
Net deferred tax assets |
$ | $ | ||||||
2023 |
2022 |
2021 |
||||||||||
Balance at the beginning of the period |
$ | $ | $ | |||||||||
Net reductions for settlement of tax audits |
( |
) | ( |
) | ||||||||
Net reductions for lapse of statutes taken during the period |
( |
) | ( |
) | ( |
) | ||||||
Net additions for tax positions taken during the prior period |
||||||||||||
Net additions for tax positions taken during the current period |
||||||||||||
Balance at the end of the period |
$ | $ | $ | |||||||||
Balance at Beginning of Period |
Charged to Provision for Income Taxes* |
Other** |
Balance at End of Period |
|||||||||||||
Valuation allowance for deferred tax assets: |
||||||||||||||||
2023 |
$ | $ | $ | $ | ||||||||||||
2022 |
$ | $ | ( |
) | $ | ( |
) | $ | ||||||||
2021 |
$ | $ | $ | ( |
) | $ | ||||||||||
| * | These amounts have been recorded as part of the income statement provision for income taxes. The income statement effects of these amounts have largely been offset by amounts related to changes in other deferred tax balance sheet accounts. |
| ** | The changes in the valuation allowance during the years ended December 31, 2023, 2022 and 2021 are primarily due to the effect of foreign currency translation on a valuation allowance related to a net operating loss carryforward. |
December 31, |
||||||||||
Financial Statement Classification |
2023 |
2022 |
||||||||
Assets: |
||||||||||
Property operating lease assets |
Operating lease assets | $ | $ | |||||||
Automobile operating lease assets |
Operating lease assets | |||||||||
Equipment operating lease assets |
Operating lease assets | |||||||||
Total lease assets |
$ | $ | ||||||||
Liabilities: |
||||||||||
Current operating lease liabilities |
Current operating lease liabilities | $ | $ | |||||||
Long-term operating lease liabilities |
Long-term operating lease liabilities | |||||||||
Total lease liabilities |
$ | $ | ||||||||
2024 |
$ | |||
2025 |
||||
2026 |
||||
2027 |
||||
2028 |
||||
2029 and thereafter |
||||
Total future minimum lease payments |
||||
Less: amount of lease payments representing interest |
( |
) | ||
Present value of future minimum lease payments |
||||
Less: current operating lease liabilities |
( |
) | ||
Long-term operating lease liabilities |
$ | |||
2023 |
2022 |
2021 |
||||||||||
Cost of sales |
$ | $ | $ | |||||||||
Selling and administrative expenses |
||||||||||||
Research and development expenses |
||||||||||||
Total stock-based compensation |
$ | $ | $ | |||||||||
Options Issued and Significant Weighted-Average Assumptions Used to Estimate Option Fair Values |
2023 |
2022 |
2021 |
|||||||||
Options issued in thousands |
||||||||||||
Risk-free interest rate |
% | % | % | |||||||||
Expected life in years |
||||||||||||
Expected volatility |
% | % | % | |||||||||
Expected dividends |
— | — | ||||||||||
Weighted-Average Exercise Price and Fair Value of Options on the Date of Grant |
2023 |
2022 |
2021 |
|||||||||
Exercise price |
$ | $ | $ | |||||||||
Fair value |
$ | $ | $ | |||||||||
Number of Shares |
Exercise Price per Share |
Weighted- Average Exercise Price per Share |
||||||||||||||||||
Outstanding at December 31, 2022 |
$ | to | $ | $ | ||||||||||||||||
Granted |
$ | to | $ | $ | ||||||||||||||||
Exercised |
( |
) | $ | to | $ | $ | ||||||||||||||
Canceled |
( |
) | $ | to | $ | $ | ||||||||||||||
Outstanding at December 31, 2023 |
$ | to | $ | $ | ||||||||||||||||
Exercise Price Range |
Number of Shares Outstanding |
Weighted- Average Exercise Price |
Remaining Contractual Life of Options Outstanding |
Number of Shares Exercisable |
Weighted- Average Exercise Price |
|||||||||||||||
$ |
$ | $ | ||||||||||||||||||
$ |
$ | $ | ||||||||||||||||||
$ |
$ | $ | ||||||||||||||||||
Total |
$ | $ | ||||||||||||||||||
Shares |
Weighted-Average Grant Date Fair Value per Share |
|||||||
Unvested at December 31, 2022 |
$ | |||||||
Granted |
$ | |||||||
Vested |
( |
) | $ | |||||
Forfeited |
( |
) | $ | |||||
Unvested at December 31, 2023 |
$ | |||||||
Performance Stock Units Issued and Significant Assumptions Used to Estimate Fair Values |
2023 |
2022 |
2021 |
|||||||||
Performance stock units issued in thousands |
||||||||||||
Risk-free interest rate |
% | % | % | |||||||||
Expected life in years |
||||||||||||
Expected volatility |
% | % | % | |||||||||
Average volatility of peer companies |
% | % | % | |||||||||
Correlation Coefficient |
% | % | % | |||||||||
Expected dividends |
— | — | ||||||||||
Shares |
Weighted-Average Fair Value per Share |
|||||||
Unvested at December 31, 2022 |
$ | |||||||
Granted |
$ | |||||||
Vested |
( |
) | $ | |||||
Forfeited |
( |
) | $ | |||||
Change in performance shares in the year due to exceeding performance targets |
$ |
|||||||
Unvested at December 31, 2023 |
$ | |||||||
Year Ended December 31, 2023 |
||||||||||||
Net Income |
Weighted-Average Shares |
Per Share |
||||||||||
(Numerator) |
(Denominator) |
Amount |
||||||||||
Net income per basic common share |
$ | $ | ||||||||||
Effect of dilutive stock option, restricted stock, performance stock unit and restricted stock unit securities |
— | ( |
) | |||||||||
Net income per diluted common share |
$ | $ | ||||||||||
Year Ended December 31, 2022 |
||||||||||||
Net Income |
Weighted-Average Shares |
Per Share |
||||||||||
(Numerator) |
(Denominator) |
Amount |
||||||||||
Net income per basic common share |
$ | $ | ||||||||||
Effect of dilutive stock option, restricted stock, performance stock unit and restricted stock unit securities |
— | ( |
) | |||||||||
Net income per diluted common share |
$ | $ | ||||||||||
Year Ended December 31, 2021 |
||||||||||||
Net Income |
Weighted-Average Shares |
Per Share |
||||||||||
(Numerator) |
(Denominator) |
Amount |
||||||||||
Net income per basic common share |
$ | $ | ||||||||||
Effect of dilutive stock option, restricted stock, performance stock unit and restricted stock unit securities |
— | ( |
) | |||||||||
Net income per diluted common share |
$ | $ | ||||||||||
Currency Translation |
Unrealized Gain (Loss) on Retirement Plans |
Unrealized Gain (Loss) on Investments |
Unrealized Loss on Derivative Instruments |
Accumulated Other Comprehensive Loss |
||||||||||||||||
Balance at December 31, 2021 |
$ | ( |
) | $ | ( |
) | $ | ( |
) | $ | $ | ( |
) | |||||||
Other comprehensive (loss) income, net of tax |
( |
) | ( |
) | ||||||||||||||||
Balance at December 31, 2022 |
$ | ( |
) | $ | $ | $ | $ | ( |
) | |||||||||||
Other comprehensive (loss) income, net of tax |
( |
) | ( |
) | ||||||||||||||||
Balance at December 31, 2023 |
$ | ( |
) | $ | ( |
) | $ | $ | ( |
) | $ | ( |
) | |||||||
2023 |
2022 |
|||||||||||||||
U.S. Retiree Healthcare Plan |
Non-U.S. Pension Plans |
U.S. Retiree Healthcare Plan |
Non-U.S. Pension Plans |
|||||||||||||
Projected benefit obligation, January 1 |
$ | $ | $ | $ | ||||||||||||
Service cost |
||||||||||||||||
Employee contributions |
||||||||||||||||
Interest cost |
||||||||||||||||
Actuarial losses (gains) |
( |
) | ( |
) | ||||||||||||
Benefits paid |
( |
) | ( |
) | ( |
) | ( |
) | ||||||||
Plan amendments |
— | ( |
) | — | ||||||||||||
Plan settlements |
— | ( |
) | — | ( |
) | ||||||||||
Currency impact |
— | — | ( |
) | ||||||||||||
Projected benefit obligation, December 31 |
$ | $ | $ | $ | ||||||||||||
2023 |
2022 |
|||||||||||||||
U.S. Retiree |
Non-U.S. |
U.S. Retiree |
Non-U.S. |
|||||||||||||
Healthcare |
Pension |
Healthcare |
Pension |
|||||||||||||
Plan |
Plans |
Plan |
Plans |
|||||||||||||
| Fair value of plan assets, January 1 |
$ | $ | $ | $ | ||||||||||||
| Actual return on plan assets |
( |
) | ( |
) | ||||||||||||
| Company contributions |
||||||||||||||||
| Employee contributions |
||||||||||||||||
| Plan settlements |
— | ( |
) | — | ( |
) | ||||||||||
| Benefits paid |
( |
) | ( |
) | ( |
) | ( |
) | ||||||||
| Currency impact |
— | — | ( |
) | ||||||||||||
| |
|
|
|
|
|
|
|
|||||||||
| Fair value of plan assets, December 31 |
$ | $ | $ | $ | ||||||||||||
| |
|
|
|
|
|
|
|
|||||||||
2023 |
2022 |
|||||||||||||||
U.S. Retiree |
Non-U.S. |
U.S. Retiree |
Non-U.S. |
|||||||||||||
Healthcare |
Pension |
Healthcare |
Pension |
|||||||||||||
Plan |
Plans |
Plan |
Plans |
|||||||||||||
| Projected benefit obligation |
$ | ( |
) | $ | ( |
) | $ | ( |
) | $ | ( |
) | ||||
| Fair value of plan assets |
||||||||||||||||
| |
|
|
|
|
|
|
|
|||||||||
| Funded status |
$ | ( |
) | $ | ( |
) | $ | ( |
) | $ | ||||||
| |
|
|
|
|
|
|
|
|||||||||
2023 |
2022 |
|||||||||||||||
U.S. Retiree |
Non-U.S. |
U.S. Retiree |
Non-U.S. |
|||||||||||||
Healthcare |
Pension |
Healthcare |
Pension |
|||||||||||||
Plan |
Plans |
Plan |
Plans |
|||||||||||||
| Long-term assets |
$ | — | $ | $ | — | $ | ||||||||||
| Long-term liabilities |
( |
) | ( |
) | ( |
) | ( |
) | ||||||||
| |
|
|
|
|
|
|
|
|||||||||
| Net amount recognized at December 31 |
$ | ( |
) | $ | ( |
) | $ | ( |
) | $ | ||||||
| |
|
|
|
|
|
|
|
|||||||||
2023 |
2022 |
|||||||
| Accumulated benefit obligations |
$ | $ | ||||||
| Fair value of plan assets |
$ | $ | ||||||
2023 |
2022 |
|||||||
| Projected benefit obligations |
$ | $ | ||||||
| Fair value of plan assets |
$ | $ | ||||||
2023 |
2022 |
2021 |
||||||||||||||||||||||
U.S. Retiree Healthcare Plan |
Non-U.S. Pension Plans |
U.S. Retiree Healthcare Plan |
Non-U.S. Pension Plans |
U.S. Retiree Healthcare Plan |
Non-U.S. Pension Plans |
|||||||||||||||||||
| Service cost |
$ | $ | $ | $ | $ | $ | ||||||||||||||||||
| Interest cost |
||||||||||||||||||||||||
| Expected return on plan assets |
( |
) | ( |
) | ( |
) | ( |
) | ( |
) | ( |
) | ||||||||||||
| Settlement loss |
— | — | — | |||||||||||||||||||||
| Net amortization: |
||||||||||||||||||||||||
| Prior service credit |
( |
) | ( |
) | ( |
) | ( |
) | ( |
) | ( |
) | ||||||||||||
| Net actuarial (gain) loss |
— | ( |
) | |||||||||||||||||||||
| |
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
| Net periodic pension cost |
$ | $ | $ | $ | $ | $ | ||||||||||||||||||
| |
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
2023 |
2022 |
2021 |
||||||||||||||||||||||
U.S. Retiree Healthcare Plan |
Non-U.S. Pension Plans |
U.S. Retiree Healthcare Plan |
Non-U.S. Pension Plans |
U.S. Retiree Healthcare Plan |
Non-U.S. Pension Plans |
|||||||||||||||||||
| Prior service credit |
$ | — | $ | $ | — | $ | $ | $ | ( |
) | ||||||||||||||
| Net (loss) gain arising during the year |
( |
) | ( |
) | ||||||||||||||||||||
| Amortization: |
||||||||||||||||||||||||
| Prior service credit |
( |
) |
( |
) | ( |
) | ( |
) | ( |
) | ( |
) | ||||||||||||
| Net loss |
||||||||||||||||||||||||
| Currency impact |
— | ( |
) | — | — | |||||||||||||||||||
| |
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
| Total recognized in other comprehensive (loss) income |
$ | ( |
) | $ | ( |
) | $ | $ | $ | $ | ||||||||||||||
| |
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
2023 |
2022 |
|||||||||||||||
U.S. Retiree Healthcare Plan |
Non-U.S. Pension Plans |
U.S. Retiree Healthcare Plan |
Non-U.S. Pension Plans |
|||||||||||||
Net actuarial (loss) gain |
$ | ( |
) |
$ | ( |
) |
$ | ( |
) | $ | ||||||
Prior service credit (cost) |
( |
) | ( |
) | ||||||||||||
Total |
$ | ( |
) | $ | ( |
) | $ | ( |
) | $ | ||||||
2023 |
2022 |
|||||||||||||||
U.S. Retiree Healthcare Plan |
Non-U.S. Pension Plans |
U.S. Retiree Healthcare Plan |
Non-U.S. Pension Plans |
|||||||||||||
Equity securities |
% | % | % | % | ||||||||||||
Debt securities |
% | % | % | % | ||||||||||||
Cash and cash equivalents |
% | % | % | % | ||||||||||||
Insurance contracts and other |
% | % | % | % | ||||||||||||
Total |
% | % | % | % | ||||||||||||
U.S. Retiree Healthcare Plan |
Non-U.S. Pension Plans Policy Target |
|||||||||||
Policy Target |
Range |
|||||||||||
Equity securities |
% | % | ||||||||||
Debt securities |
% | % | ||||||||||
Cash and cash equivalents |
% | % | ||||||||||
Insurance contracts and other |
% | % | ||||||||||
Level 1: |
The fair value of these types of investments is based on market and observable sources from daily quoted prices on nationally recognized securities exchanges. | |
Level 2: |
The fair value of these types of investments utilizes data points other than quoted prices in active markets that are observable either directly or indirectly. | |
Level 3: |
These bank and insurance investment contracts are issued by well-known, highly-rated companies. The fair value disclosed represents the present value of future cash flows under the terms of the respective contracts. Significant assumptions used to determine the fair value of these contracts include the amount and timing of future cash flows and counterparty credit risk. | |
Total at December 31, 2023 |
Quoted Prices in Active Markets for Identical Assets (Level 1) |
Significant Other Observable Inputs (Level 2) |
Significant Unobservable Inputs (Level 3) |
|||||||||||||
U.S. Retiree Healthcare Plan: |
||||||||||||||||
Mutual funds (a) |
— | — | ||||||||||||||
Total U.S. Retiree Healthcare Plan |
— | — | ||||||||||||||
Non-U.S. Pension Plans: |
||||||||||||||||
Cash equivalents (b) |
— | — | ||||||||||||||
Mutual funds (c) |
— | — | ||||||||||||||
Bank and insurance investment contracts (d) |
— | — | ||||||||||||||
Total Non-U.S. Pension Plans |
— | |||||||||||||||
Total fair value of retirement plan assets |
$ | $ | $ | — | $ | |||||||||||
Total at December 31, 2022 |
Quoted Prices in Active Markets for Identical Assets (Level 1) |
Significant Other Observable Inputs (Level 2) |
Significant Unobservable Inputs (Level 3) |
|||||||||||||
U.S. Retiree Healthcare Plan: |
||||||||||||||||
Mutual funds (e) |
— | — | ||||||||||||||
Total U.S. Retiree Healthcare Plan |
— | — | ||||||||||||||
Non-U.S. Pension Plans: |
||||||||||||||||
Cash equivalents (b) |
— | — | ||||||||||||||
Mutual funds (f) |
— | — | ||||||||||||||
Bank and insurance investment contracts (d) |
— | — | ||||||||||||||
Total Non-U.S. Pension Plans |
— | |||||||||||||||
Total fair value of retirement plan assets |
$ | $ | $ | — | $ | |||||||||||
| a) | The mutual fund balance in the U.S. Retiree Healthcare Plan is invested in the following categories: large-cap U.S. companies, |
| b) | Primarily represents deposit account funds held with various financial institutions. |
| c) | The mutual fund balance in the Non-U.S. Pension Plans is primarily invested in the following categories: |
| d) | Amount represents bank and insurance guaranteed investment contracts. |
| e) | The mutual fund balance in the U.S. Retiree Healthcare Plan is invested in the following categories: large-cap U.S. companies, |
| f) | The mutual fund balance in the Non-U.S. Pension Plans is invested in the following categories: |
Insurance Guaranteed Investment Contracts |
||||
Fair value of assets, December 31, 2021 |
$ | |||
Net purchases (sales) and appreciation (depreciation) |
( |
) | ||
Fair value of assets, December 31, 2022 |
||||
Net purchases (sales) and appreciation (depreciation) |
||||
Fair value of assets, December 31, 2023 |
$ | |||
2023 |
2022 |
2021 |
||||||||||||||||||||||
U.S. |
Non-U.S. |
U.S. |
Non-U.S. |
U.S. |
Non-U.S. |
|||||||||||||||||||
Discount rate |
% | % | % | % | % | % | ||||||||||||||||||
Increases in compensation levels |
* | * | % | * | * | % | * | * | % | |||||||||||||||
Interest crediting rate |
% | % | % | % | % | % | ||||||||||||||||||
| ** | Not applicable |
2023 |
2022 |
2021 |
||||||||||||||||||||||
U.S. |
Non-U.S. |
U.S. |
Non-U.S. |
U.S. |
Non-U.S. |
|||||||||||||||||||
Discount rate |
% | % | % | % | % | % | ||||||||||||||||||
Return on plan assets |
% | % | % | % | % | % | ||||||||||||||||||
Increases in compensation levels |
* | * | % | * | * | % | * | * | % | |||||||||||||||
Interest crediting rate |
% | % | % | % | % | % | ||||||||||||||||||
| ** | Not applicable |
U.S. Retiree Healthcare Plans |
Non-U.S. Pension Plans |
Total |
||||||||||
2024 |
$ | $ | $ | |||||||||
2025 |
||||||||||||
2026 |
||||||||||||
2027 |
||||||||||||
2028 |
||||||||||||
2029 - 2033 |
||||||||||||
2023 |
2022 |
2021 |
||||||||||
Product net sales: |
||||||||||||
Waters instrument systems |
$ | $ | $ | |||||||||
Chemistry consumables |
||||||||||||
TA instrument systems |
||||||||||||
Total product sales |
||||||||||||
Service net sales: |
||||||||||||
Waters service |
||||||||||||
TA service |
||||||||||||
Total service sales |
||||||||||||
Total net sales |
$ | $ | $ | |||||||||
2023 |
2022 |
2021 |
||||||||||
Net Sales: |
||||||||||||
Asia: |
||||||||||||
China |
$ | $ | $ | |||||||||
Japan |
||||||||||||
Asia Other |
||||||||||||
Total Asia |
||||||||||||
Americas: |
||||||||||||
United States |
||||||||||||
Americas Other |
||||||||||||
Total Americas |
||||||||||||
Europe |
||||||||||||
Total net sales |
$ | $ | $ | |||||||||
2023 |
2022 |
2021 |
||||||||||
Pharmaceutical |
$ | $ | $ | |||||||||
Industrial |
||||||||||||
Academic and government |
||||||||||||
Total net sales |
$ | $ | $ | |||||||||
2023 |
2022 |
2021 |
||||||||||
Net sales recognized at a point in time: |
||||||||||||
Instrument systems |
$ | $ | $ | |||||||||
Chemistry consumables |
||||||||||||
Service sales recognized at a point in time (time & materials) |
||||||||||||
Total net sales recognized at a point in time |
||||||||||||
Net sales recognized over time: |
||||||||||||
Service and software maintenance sales recognized over time (contracts) |
||||||||||||
Total net sales |
$ | $ | $ | |||||||||
December 31, |
||||||||||||
2023 |
2022 |
2021 |
||||||||||
Long-lived assets: |
||||||||||||
United States |
$ | $ | $ | |||||||||
Americas Other |
||||||||||||
Total Americas |
||||||||||||
Europe |
||||||||||||
Asia |
||||||||||||
Total long-lived assets |
$ | $ | $ | |||||||||
First |
Second |
Third |
Fourth |
|||||||||||||||||
2023 |
Quarter |
Quarter |
Quarter |
Quarter |
Total |
|||||||||||||||
Net sales |
$ | $ | $ | $ | $ | |||||||||||||||
Costs and operating expenses: |
||||||||||||||||||||
Cost of sales |
||||||||||||||||||||
Selling and administrative expenses |
||||||||||||||||||||
Research and development expenses |
||||||||||||||||||||
Purchased intangibles amortization |
||||||||||||||||||||
Total costs and operating expenses |
||||||||||||||||||||
Operating income |
||||||||||||||||||||
Other income (expense), net |
( |
) | ( |
) | ||||||||||||||||
Interest expense |
( |
) | ( |
) | ( |
) | ( |
) | ( |
) | ||||||||||
Interest income |
||||||||||||||||||||
Income before income taxes |
||||||||||||||||||||
Provision for income taxes |
||||||||||||||||||||
Net income |
$ | $ | $ | $ | $ | |||||||||||||||
Net income per basic common share |
||||||||||||||||||||
Weighted-average number of basic common shares |
||||||||||||||||||||
Net income per diluted common share |
||||||||||||||||||||
Weighted-average number of diluted common shares and equivalents |
||||||||||||||||||||
First |
Second |
Third |
Fourth |
|||||||||||||||||
| 2022 |
Quarter |
Quarter |
Quarter |
Quarter |
Total |
|||||||||||||||
| Net sales |
$ | $ | $ | $ | $ | |||||||||||||||
| Costs and operating expenses: |
||||||||||||||||||||
| Cost of sales |
||||||||||||||||||||
| Selling and administrative expenses |
||||||||||||||||||||
| Research and development expenses |
||||||||||||||||||||
| Purchased intangibles amortization |
||||||||||||||||||||
| Acquired in-process research and development |
— | — | — | |||||||||||||||||
| |
|
|
|
|
|
|
|
|
|
|||||||||||
| Total costs and operating expenses |
||||||||||||||||||||
| |
|
|
|
|
|
|
|
|
|
|||||||||||
| Operating income |
||||||||||||||||||||
| Other income (expense), net |
( |
) | ||||||||||||||||||
| Interest expense |
( |
) | ( |
) | ( |
) | ( |
) | ( |
) | ||||||||||
| Interest income |
||||||||||||||||||||
| |
|
|
|
|
|
|
|
|
|
|||||||||||
| Income before income taxes |
||||||||||||||||||||
| Provision for income taxes |
||||||||||||||||||||
| |
|
|
|
|
|
|
|
|
|
|||||||||||
| Net income |
$ | $ | $ | $ | $ | |||||||||||||||
| |
|
|
|
|
|
|
|
|
|
|||||||||||
| Net income per basic common share |
||||||||||||||||||||
| Weighted-average number of basic common shares |
||||||||||||||||||||
| Net income per diluted common share |
||||||||||||||||||||
| Weighted-average number of diluted common shares and equivalents |
||||||||||||||||||||
Table of Contents
Item 9: Changes in and Disagreements with Accountants on Accounting and Financial Disclosure
None.
Item 9A: Controls and Procedures
Evaluation of Disclosure Controls and Procedures
The Company’s chief executive officer and chief financial officer (principal executive officer and principal financial officer), with the participation of management, evaluated the effectiveness of the Company’s disclosure controls and procedures (as defined in Rules 13a-15(e) and 15d-15(e) under the Exchange Act) as of the end of the period covered by this annual report on Form 10-K. Based on this evaluation, the Company’s chief executive officer and chief financial officer concluded that the Company’s disclosure controls and procedures were effective as of December 31, 2023 (1) to ensure that information required to be disclosed by the Company, including its consolidated subsidiaries, in the reports that it files or submits under the Exchange Act is accumulated and communicated to the Company’s management, including its chief executive officer and chief financial officer, to allow timely decisions regarding the required disclosure and (2) to provide reasonable assurance that information required to be disclosed by the Company in the reports that it files or submits under the Exchange Act is recorded, processed, summarized and reported within the time periods specified in the SEC’s rules and forms.
Management’s Annual Report on Internal Control Over Financial Reporting
See Management’s Report on Internal Control Over Financial Reporting in Item 8 on page 50 of this Annual Report.
Report of the Independent Registered Public Accounting Firm
See the report of PricewaterhouseCoopers LLP in Item 8 beginning on page 51 of this Annual Report.
Changes in Internal Control Over Financial Reporting
No change was identified in the Company’s internal control over financial reporting (as defined in Rules 13a-15(f) and 15d-15(f) under the Exchange Act) during the quarter ended December 31, 2023 that has materially affected, or is reasonably likely to materially affect, the Company’s internal control over financial reporting.
Item 9B: Other Information
Insider Trading Arrangements and Related Disclosures
None.
Amendment and Restatement of Bylaws
On February 23, 2024, the Board of Directors of the Company approved an amendment and restatement of the bylaws of the Company (the “Amended Bylaws”), effective as of such date.
Among other matters, the Amended Bylaws:
| (1) | revise procedures and disclosure requirements for the nomination of directors and the submission of proposals for consideration at meetings of the stockholders of the Company, including, among other things, limiting the scope of persons to whom such disclosure requirements apply and adding a requirement that a stockholder seeking to nominate director(s) at an annual meeting deliver to the Company reasonable evidence that it has complied with the requirements of Rule 14a-19 of the Exchange Act, no less than seven business days prior to the meeting; |
101
Table of Contents
| (2) | clarify the applicability of the majority voting standard for contested elections of directors; |
| (3) | clarify the position, duties and powers of the Chairman and Vice Chairman within the Company structure; |
| (4) | make certain administrative, modernizing, clarifying and conforming changes, including making updates to reflect recent amendments to the General Corporation Law of the State of Delaware; and |
| (5) | adopt gender-neutral terms when referring to particular positions, offices or title holders, including the adoption of the title Chair in place of Chairman. |
The foregoing description of the Amended Bylaws does not purport to be complete and is qualified in its entirety by reference to the full text of the Amended Bylaws, a copy of which is attached hereto as Exhibit 3.5 and incorporated herein by reference.
Item 9C: Disclosure Regarding Foreign Jurisdictions that Prevent Inspections
None.
PART III
| Item 10: | Directors, Executive Officers and Corporate Governance |
INFORMATION ABOUT OUR EXECUTIVE OFFICERS
Officers of the Company are elected annually by the Board of Directors and hold office at the discretion of the Board of Directors. The following persons serve as executive officers of the Company:
Dr. Udit Batra, 53, was appointed a Director of the Company as well as President and CEO on September 1, 2020. He most recently served as Chief Executive Officer of the Life Science business of Merck KGaA, Darmstadt, Germany, which operates as MilliporeSigma in the United States and Canada, and as a member of its Executive Board, roles he held from 2014 and 2016, respectively, through July 2020. Prior to that, Dr. Batra served as President and Chief Executive Officer of Merck KGaA, Darmstadt, Germany’s Consumer Health business. Dr. Batra oversaw the company’s Bioethics Advisory Panel and had Board responsibility for the global Information Technology function. Before joining Merck KGaA, Darmstadt, Germany, Dr. Batra held several positions of increasing responsibility at Novartis, including Global Head of Corporate Strategy in Switzerland, Country President for the Pharma Business of Novartis in Australia and New Zealand and the Global Head of Public Health and Market Access in Cambridge, Massachusetts. Dr. Batra also served at the global consultancy McKinsey & Company across the healthcare, consumer and non-profit sectors. Dr. Batra started his career at Merck Research Labs in West Point, Pennsylvania as a research engineer.
Jianqing Bennett, 54, was appointed Senior Vice President of TA Instruments Division on May 1, 2021. Previously, Ms. Bennett served as Senior Vice President, High Growth Markets at Beckman Coulter Diagnostics from November 2017 to March 2021. Prior to that, from 2007-2017, she held various senior management positions at Carestream Health Inc, including serving as President, Medical Digital Solutions from August 2015 to November 2017.
Amol Chaubal, 48, was appointed Chief Financial Officer of Waters Corporation on May 12, 2021. Previously, Mr. Chaubal was Chief Financial Officer of Quanterix Corporation, a life sciences company, where he served as Chief Financial Officer since April 2019. Before Quanterix, Mr. Chaubal served as Chief Financial Officer, Global Operations at Smith & Nephew, a global medical devices company, from October 2017 to April 2019. Prior to his time at Smith & Nephew, he served as Corporate Vice President and Head of Finance for the Clinical Research Services and Access business at Parexel from July 2015 to October 2017.
102
Table of Contents
Information regarding the Company’s directors, any material changes to the process by which security holders may recommend nominees to the Board of Directors and the information required by the Item will be contained in our definitive proxy statement for the 2024 Annual Meeting of Stockholders, to be filed with the SEC not later than 120 days after the close of business of the fiscal year and is incorporated in this report by reference (the “2024 Proxy Statement”), under the headings “Election of Directors”, “Directors Meetings and Board Committees”, “Corporate Governance”, “Report of the Audit and Finance Committee of the Board of Directors” and “Compensation of Directors and Executive Officers”. Information regarding compliance with Section 16(a) of the Exchange Act will be contained in the 2024 Proxy Statement, under the heading “Delinquent Section 16(a) Reports”. Information regarding the Company’s Audit and Finance Committee and Audit and Finance Committee Financial Expert will be contained in the 2024 Proxy Statement, under the headings “Report of the Audit and Finance Committee of the Board of Directors” and “Directors Meetings and Board Committees”. Such information is incorporated herein by reference.
The Company has adopted a Global Code of Business Conduct & Ethics (the “Code”) that applies to all of the Company’s employees (including its executive officers) and directors and that is in compliance with Item 406 of Regulation S-K. The Code has been distributed to all employees of the Company. In addition, the Code is available on the Company’s website, https://www.waters.com, under the caption “Corporate Governance”. The Company intends to satisfy the disclosure requirement regarding any amendment to, or waiver of a provision of, the Code applicable to any executive officer or director by posting such information on its website. The Company shall also provide to any person without charge, upon request, a copy of the Code. Any such request must be made in writing to the Secretary of the Company, c/o Waters Corporation, 34 Maple Street, Milford, MA 01757.
The Company’s corporate governance guidelines and the charters of the audit committee, compensation committee and nominating and corporate governance committee of the Board of Directors are available on the Company’s website, https://www.waters.com, under the caption “Corporate Governance”. The Company shall provide to any person without charge, upon request, a copy of any of the foregoing materials. Any such request must be made in writing to the Secretary of the Company, c/o Waters Corporation, 34 Maple Street, Milford, MA 01757.
| Item 11: | Executive Compensation |
This information will be contained in the 2024 Proxy Statement, under the headings “Compensation of Directors and Executive Officers”, “Compensation Committee Interlocks and Insider Participation” and “Compensation Committee Report”. Such information is incorporated herein by reference.
Item 12: Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters
Except for the Equity Compensation Plan information set forth below, this information will be contained in the 2024 Proxy Statement, under the heading “Security Ownership of Certain Beneficial Owners and Management”. Such information is incorporated herein by reference.
103
Table of Contents
Equity Compensation Plan Information
The following table provides information as of December 31, 2023 about the Company’s common stock that may be issued upon the exercise of options, warrants and rights under its existing equity compensation plans (in thousands):
| A | B | C | ||||||||||
| Number of Securities to be Issued Upon Exercise of Outstanding Options, Warrants and Rights (1) |
Weighted-Average Exercise Price of Outstanding Options, Warrants and Rights (1) |
Number of Securities Remaining Available for Future Issuance Under Equity Compensation Plans (excluding securities reflected in column (A)) |
||||||||||
| Equity compensation plans approved by security holders |
939 | $ | 265.17 | 6,680 | ||||||||
| Equity compensation plans not approved by security holders |
— | — | — | |||||||||
|
|
|
|
|
|
|
|||||||
| Total |
939 | $ | 265.17 | 6,680 | ||||||||
|
|
|
|
|
|
|
|||||||
| (1) | Column (a) includes an aggregate of 352 shares of common stock to be issued upon settlement of restricted stock, restricted stock units and performance stock units. The weighted-average share price in column (b) does not take into account restricted stock, restricted stock units or performance stock units, which do not have an exercise price. |
See Note 14, Stock-Based Compensation, in the Notes to Consolidated Financial Statements for a description of the material features of the Company’s equity compensation plans.
| Item 13: | Certain Relationships and Related Transactions and Director Independence |
This information is contained in the 2024 Proxy Statement, under the headings “Directors Meetings and Board Committees”, “Corporate Governance” and “Compensation of Directors and Executive Officers”. Such information is incorporated herein by reference.
| Item 14: | Principal Accountant Fees and Services |
This information is contained in the 2024 Proxy Statement, under the headings “Ratification of Selection of Independent Registered Public Accounting Firm” and “Report of the Audit and Finance Committee of the Board of Directors”. Such information is incorporated herein by reference.
104
Table of Contents
| (1) | Financial Statements: |
| (2) | Exhibits: |
Exhibit Number |
Description of Document | |
| 21.1 | Subsidiaries of Waters Corporation. | |
| 23.1 | Consent of PricewaterhouseCoopers LLP, an independent registered public accounting firm. | |
| 31.1 | Chief Executive Officer Certification Pursuant to Section 302 of the Sarbanes-Oxley Act of 2002. | |
| 31.2 | Chief Financial Officer Certification Pursuant to Section 302 of the Sarbanes-Oxley Act of 2002. | |
| 32.1 | Chief Executive Officer Certification Pursuant to 18 U.S.C. Section 1350, as Adopted Pursuant to Section 906 of the Sarbanes-Oxley Act of 2002.** | |
| 32.2 | Chief Financial Officer Certification Pursuant to 18 U.S.C. Section 1350, as Adopted Pursuant to Section 906 of the Sarbanes-Oxley Act of 2002.** | |
| 97 | Waters Mandatory Clawback Policy | |
| 101 | The following materials from Waters Corporation’s Annual Report on Form 10-K for the year ended December 31, 2023, formatted in iXBRL (Inline eXtensible Business Reporting Language): (i) the Consolidated Balance Sheets, (ii) the Consolidated Statements of Operations, (iii) the Consolidated Statements of Comprehensive Income, (iv) the Consolidated Statements of Cash Flows, (v) the Consolidated Statements of Stockholders’ Equity (Deficit) and (vi) Notes to Consolidated Financial Statements. | |
| 104 | Cover Page Interactive Date File (formatted in iXBRL and contained in Exhibit 101). | |
| + | Paper Filing |
| * | Management contract or compensatory plan required to be filed as an Exhibit to this Annual Report. |
| ** | This exhibit shall not be deemed “filed” for purposes of Section 18 of the Exchange Act, or otherwise subject to the liability of that section, nor shall it be deemed incorporated by reference into any filing under the Securities Act, or the Exchange Act, whether made before or after the date hereof and irrespective of any general incorporation language in any filing, except to the extent the Company specifically incorporates it by reference. |
| (b) | See Item 15 (a) (2) above. |
Item 16: |
Form 10-K Summary |
Table of Contents
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| WATERS CORPORATION |
| /s/ Amol Chaubal |
| Amol Chaubal |
| Senior Vice President and Chief Financial Officer |
| (Principal Financial Officer) |
| (Principal Accounting Officer) |
Date: February 27, 2024
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed by the following persons on behalf of the registrant and in the capacities indicated on February 27, 2024.
| /s/ Dr. Flemming Ornskov, M.D., M.P.H. | Chairman of the Board of Directors | |||
| Dr. Flemming Ornskov, M.D., M.P.H. | ||||
| /s/ Udit Batra, Ph.D. | President and Chief Executive Officer | |||
| Udit Batra, Ph.D. | Director (Principal Executive Officer) | |||
| /s/ Amol Chaubal | Senior Vice President and Chief Financial Officer | |||
| Amol Chaubal | (Principal Financial Officer) | |||
| (Principal Accounting Officer) | ||||
| /s/ Linda Baddour | Director | |||
| Linda Baddour | ||||
| /s/ Dan Brennan | Director | |||
| Dan Brennan | ||||
| /s/ Richard Fearon | Director | |||
| Richard Fearon | ||||
| /s/ Pearl S. Huang, Ph.D. | Director | |||
| Pearl S. Huang, Ph.D. | ||||
| /s/ Wei Jiang | Director | |||
| Wei Jiang | ||||
| /s/ Christopher A. Kuebler | Director | |||
| Christopher A. Kuebler | ||||
| /s/ Mark Vergnano | Director | |||
| Mark Vergnano | ||||
109