Exhibit 99.1

PREVENT - 19 PHASE 3 ADOLESCENT RESULTS 1 © 2022 NOVAVAX. All rights reserved. February 10, 2022

SAFE HARBOR STATEMENT Certain information, particularly information relating to the future of Novavax, its operating plans and prospects, its partn ers hips, the ongoing development of NVX - CoV2373, including Novavax’ plans for a subsequent pediatric study planned for Q2 2022, COVID - NanoFlu combination vaccine an d other Novavax vaccine product candidates, the timing of results from clinical trials, the potential for a booster dose of NVX - CoV2373 to provi de protection against COVID - 19 (including variants), the scope and timing of future regulatory filings and actions, including Novavax’ plans to supplement g lob al regulatory filings with pediatric data in Q1 2022, anticipated manufacturing capacity, the readiness of our global supply chain and future availabili ty of NVX - CoV2373 at a global scale and the anticipated commercialization of NVX - CoV2373 constitute forward - looking statements within the meaning of the Priva te Securities Litigation Reform Act. Forward - looking statements may generally contain words such as “believe,” “may,” “could,” “will,” “possible,” “can,” “estimate,” “continue,” “ongoing,” “consider,” “intend,” “indicate,” “plan,” “project,” “expect,” “should,” “would,” or “assume” or variations of such words or oth er words with similar meanings. Novavax cautions that these forward - looking statements are subject to numerous assumptions, risks and uncertainties that change over time and may cause actual results to differ materially from the results discussed in the forward - looking statements. These risks and uncertainties include challenges satisfying, alone or together with partners, various safety, efficacy, and p rod uct characterization requirements, including those related to process qualification and assay validation, necessary to satisfy applicable regulatory authorities ; d ifficulty obtaining scarce raw materials and supplies; resource constraints, including human capital and manufacturing capacity, on the ability of Novavax t o p ursue planned regulatory pathways; challenges meeting contractual requirements under agreements with multiple commercial, governmental, and other enti tie s; and those other risk factors identified in the “Risk Factors” and “Management's Discussion and Analysis of Financial Condition and Results of Oper ati ons” sections of Novavax' Annual Report on Form 10 - K for the year ended December 31, 2020 and subsequent Quarterly Reports on Form 10 - Q, as filed with the Securities and Exchange Commission, which are available at www.sec.gov and www.novavax.com. Forward - looking statements are based on current expectations and assumptions and currently available data and are neither predic tions nor guarantees of future events or performance. Current results may not be predictive of future results. You should not place considerable reliance on forward - looking statements which speak only as of the date hereof. The Company does not undertake to update or revise any forward - looking statements after they are made, whether as a result of ne w information, future events, or otherwise, except as required by applicable law. Novavax Œ (and all associated logos) is a trademark of Novavax, Inc. Matrix - M Œ is a trademark of Novavax AB. 2 © 2022 NOVAVAX . All rights reserved.
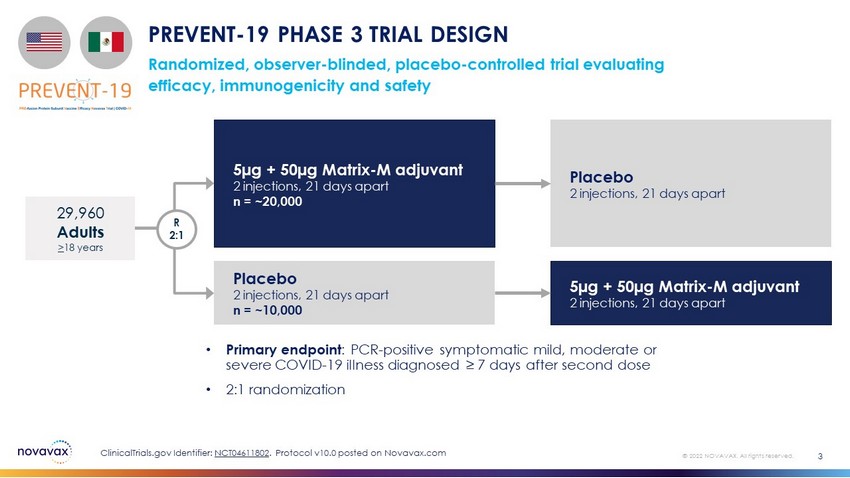
Randomized, observer - blinded, placebo - controlled trial evaluating efficacy, immunogenicity and safety PREVENT - 19 PHASE 3 TRIAL DESIGN • Primary endpoint : PCR - positive symptomatic mild, moderate or severe COVID - 19 illness diagnosed ≥ 7 days after second dose • 2:1 randomization 3 © 2022 NOVAVAX. All rights reserved. 5µg + 50µg Matrix - M adjuvant 2 injections, 21 days apart n = ~20,000 Placebo 2 injections, 21 days apart n = ~10,000 29,960 Adults > 18 years R 2:1 Placebo 2 injections, 21 days apart 5µg + 50µg Matrix - M adjuvant 2 injections, 21 days apart ClinicalTrials.gov Identifier: NCT04611802 . Protocol v10.0 posted on Novavax.com
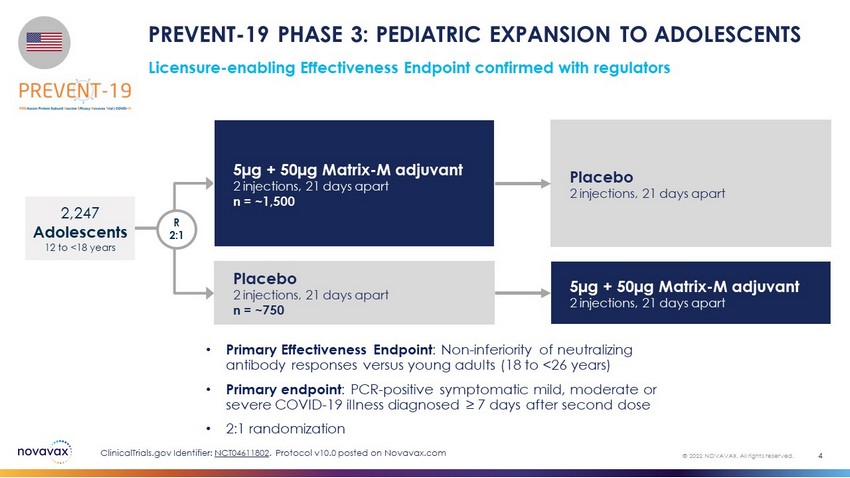
Licensure - enabling Effectiveness Endpoint confirmed with regulators PREVENT - 19 PHASE 3: PEDIATRIC EXPANSION TO ADOLESCENTS 4 © 2022 NOVAVAX. All rights reserved. • Primary Effectiveness Endpoint : Non - inferiority of neutralizing antibody responses versus young adults (18 to <26 years) • Primary endpoint : PCR - positive symptomatic mild, moderate or severe COVID - 19 illness diagnosed ≥ 7 days after second dose • 2:1 randomization 5µg + 50µg Matrix - M adjuvant 2 injections, 21 days apart n = ~1,500 Placebo 2 injections, 21 days apart n = ~750 2,247 Adolescents 12 to <18 years R 2:1 Placebo 2 injections, 21 days apart 5µg + 50µg Matrix - M adjuvant 2 injections, 21 days apart ClinicalTrials.gov Identifier: NCT04611802 . Protocol v10.0 posted on Novavax.com
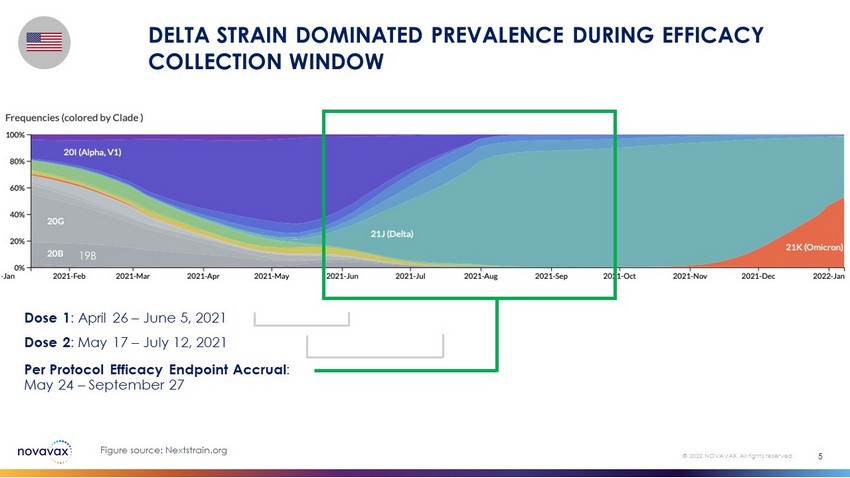
Dose 1 : April 26 – June 5, 2021 Dose 2 : May 17 – July 12, 2021 Per Protocol Efficacy Endpoint Accrual : May 24 – September 27 19B DELTA STRAIN DOMINATED PREVALENCE DURING EFFICACY COLLECTION WINDOW 5 © 2022 NOVAVAX. All rights reserved. Figure source: Nextstrain.org

DEMOGRAPHICS WELL BALANCED BETWEEN PLACEBO AND VACCINE, WITH ADEQUATE RACE AND ETHNIC REPRESENTATION 6 © 2022 NOVAVAX. All rights reserved. NVX - CoV2373 n=1,487 Placebo n=745 Female 49.2% 44.2% 12 to <15 years of age 67.1% 67.1% 15 to <18 years of age 32.9% 32.9% White 75.0% 73.2% Black or African American 13.6% 14.5% Hispanic or Latino 18.4% 18.5% Baseline anti - N positive 15.3% 16.2%

16.3% 6.4% 0.4% 0.1% 0.5% 0.0% 15.8% 6.8% 0.3% 0.1% 0.3% 0.0% 0% 20% 40% 60% 80% 100% Any AE Any Medically Attended AE Any Severe AE AE leading to vaccine discontinuation Serious AE Deaths NVX-CoV2373 Placebo Overview up to crossover SAFETY EVENTS WERE BALANCED BETWEEN VACCINE & PLACEBO 7 © 2022 NOVAVAX. All rights reserved. • No PIMMC or AESI were reported • SAEs were infrequent and balanced between groups (n=1,487) (n=745)
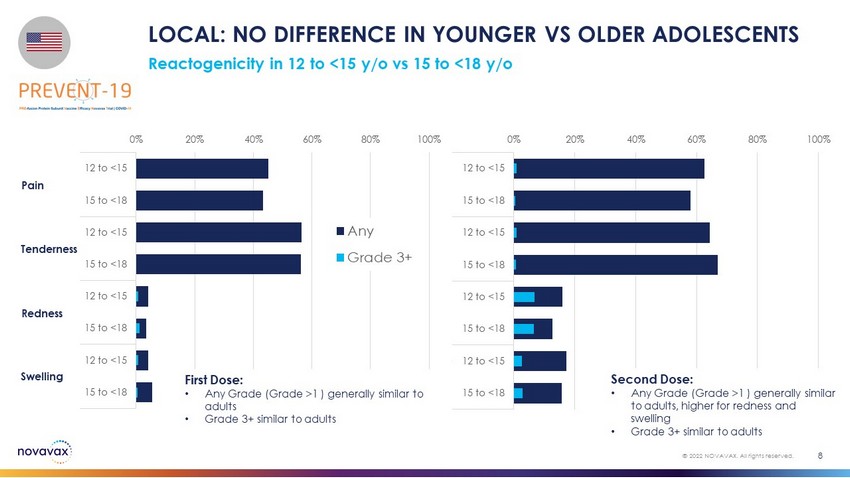
0% 20% 40% 60% 80% 100% 12 to <15 15 to <18 12 to <15 15 to <18 12 to <15 15 to <18 12 to <15 15 to <18 Pain Tenderness Redness Swelling 0% 20% 40% 60% 80% 100% 12 to <15 15 to <18 12 to <15 15 to <18 12 to <15 15 to <18 12 to <15 15 to <18 Pain Tenderness Redness Swelling Any Grade 3+ Pain Tenderness Redness Swelling Reactogenicity in 12 to <15 y/o vs 15 to <18 y/o LOCAL: NO DIFFERENCE IN YOUNGER VS OLDER ADOLESCENTS 8 © 2022 NOVAVAX. All rights reserved. First Dose: • Any Grade (Grade >1 ) generally similar to adults • Grade 3+ similar to adults Second Dose: • Any Grade (Grade >1 ) generally similar to adults, higher for redness and swelling • Grade 3+ similar to adults

Reactogenicity in 12 to <15 y/o vs 15 to <18 y/o SYSTEMIC: NO DIFFERENCE IN YOUNGER VS OLDER ADOLESCENTS 9 © 2022 NOVAVAX. All rights reserved. 0% 20% 40% 60% 80% 100% 12 to <15 15 to <18 12 to <15 15 to <18 12 to <15 15 to <18 12 to <15 15 to <18 12 to <15 15 to <18 12 to <15 15 to <18 12 to <15 15 to <18 Any Grade 3+ Muscle pain Headache Fatigue Malaise Nausea/ Vomiting Joint Pain Fever First Dose: • Any Grade (Grade >1 ) generally similar to adults, higher for muscle pain • Grade 3+ similar to adults 0% 20% 40% 60% 80% 100% 12 to <15 15 to <18 12 to <15 15 to <18 12 to <15 15 to <18 12 to <15 15 to <18 12 to <15 15 to <18 12 to <15 15 to <18 12 to <15 15 to <18 Second Dose: • Any Grade (Grade >1 ) generally lower or similar than in adults, higher for headache, fever, nausea/vomiting • Grade 3+ generally similar or slightly higher than adults
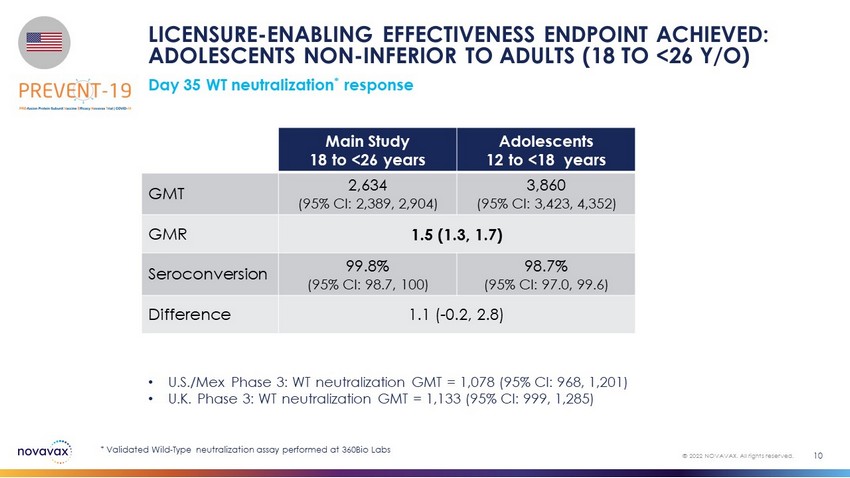
LICENSURE - ENABLING EFFECTIVENESS ENDPOINT ACHIEVED: ADOLESCENTS NON - INFERIOR TO ADULTS (18 TO <26 Y/O) Day 35 WT neutralization * response 10 © 2022 NOVAVAX. All rights reserved. Main Study 18 to <26 years Adolescents 12 to <18 years GMT 2,634 (95% CI: 2,389, 2,904) 3,860 (95% CI: 3,423, 4,352) GMR 1.5 (1.3, 1.7) Seroconversion 99.8% (95% CI: 98.7, 100) 98.7% (95% CI: 97.0, 99.6) Difference 1.1 ( - 0.2, 2.8 ) • U.S./Mex Phase 3: WT neutralization GMT = 1,078 (95% CI: 968, 1,201) • U.K. Phase 3: WT neutralization GMT = 1,133 (95% CI: 999, 1,285) * Validated Wild - Type neutralization assay performed at 360Bio Labs

DAY 35 I g G * MAGNITUDE HIGHER THAN 18 TO <25 Y/O ADULTS IN MAIN STUDY 11 © 2022 NOVAVAX. All rights reserved. 12 to <18 137,671 EU/mL 12 to <15 145,817 EU/mL 15 to <18 121,732 EU/mL 18 to <26 99,386 EU/mL IgG by Age Group GMR = 1.4 (95% CI: 1.2, 1.6) in adolescents vs 18 to <26 y/o * Validated prototype IgG ELISA assay performed at Novavax Clinical Immunology

Adolescent IgG following 2 - dose primary series (Day 35) VARIANT I g G RESPONSES 2 - 3X HIGHER THAN IN ADULTS; 100% SEROCONVERSION AGAINST ALL VARIANTS 12 © 2022 NOVAVAX. All rights reserved. Anti - rS IgG Titer (EC50) 1 10 100 1,000 10,000 100,000 1,000,000 Prototype Alpha Beta Delta Delta+ Gamma Mu Omicron LOD = 10 B.1.1.7 B.1.351 B.1.617.2 AY.4.2 P.1 B.1.621 B.1.1.529 Fit - for - purpose assay performed at Novavax Discovery labs
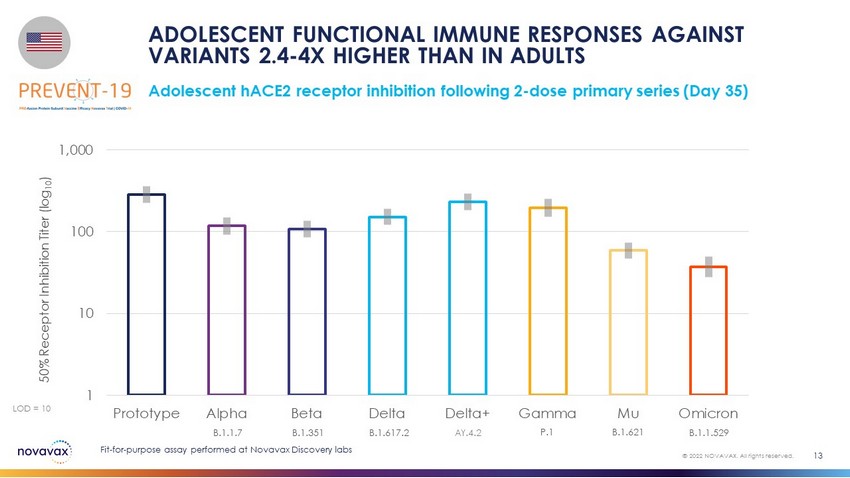
Adolescent hACE2 receptor inhibition following 2 - dose primary series (Day 35) ADOLESCENT FUNCTIONAL IMMUNE RESPONSES AGAINST VARIANTS 2.4 - 4X HIGHER THAN IN ADULTS 13 © 2022 NOVAVAX. All rights reserved. LOD = 10 50% Receptor Inhibition Titer (log 10 ) 1 10 100 1,000 Prototype Alpha Beta Delta Delta+ Gamma Mu Omicron B.1.1.7 B.1.351 B.1.617.2 AY.4.2 P.1 B.1.621 B.1.1.529 Fit - for - purpose assay performed at Novavax Discovery labs
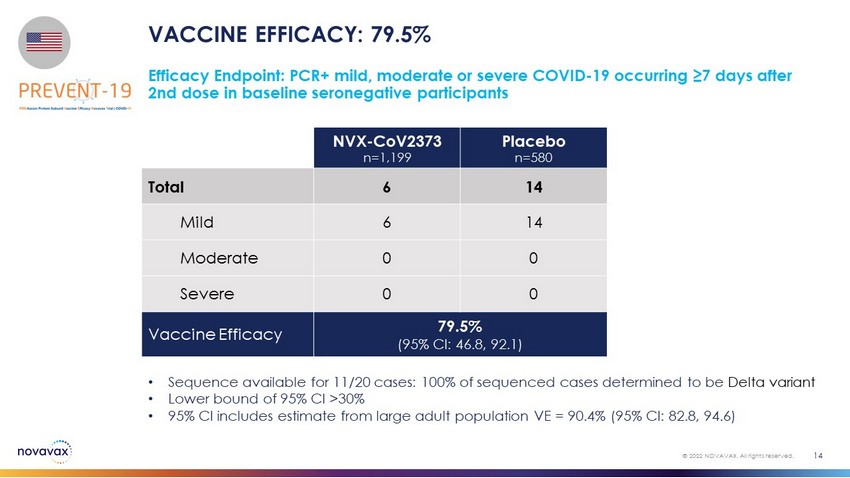
VACCINE EFFICACY: 79.5% 14 © 2022 NOVAVAX. All rights reserved. • Sequence available for 11/20 cases: 100% of sequenced cases determined to be Delta variant • Lower bound of 95% CI >30% • 95% CI includes estimate from large adult population VE = 90.4% (95% CI: 82.8, 94.6) Efficacy Endpoint: PCR+ mild, moderate or severe COVID - 19 occurring ≥7 days after 2nd dose in baseline seronegative participants NVX - CoV2373 n=1,199 Placebo n=580 Total 6 14 Mild 6 14 Moderate 0 0 Severe 0 0 Vaccine Efficacy 79.5% (95% CI: 46.8, 92.1)
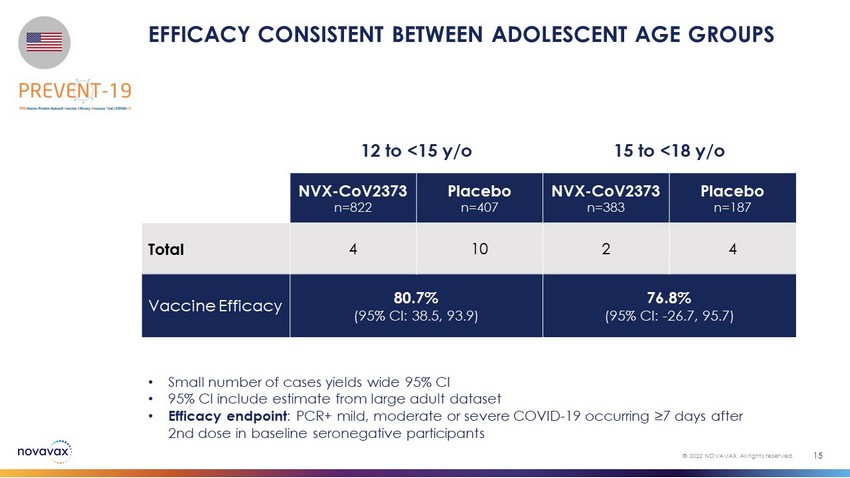
EFFICACY CONSISTENT BETWEEN ADOLESCENT AGE GROUPS 15 © 2022 NOVAVAX. All rights reserved. • Small number of cases yields wide 95% CI • 95% CI include estimate from large adult dataset • Efficacy endpoint : PCR+ mild, moderate or severe COVID - 19 occurring ≥7 days after 2nd dose in baseline seronegative participants 12 to <15 y/o 15 to <18 y/o NVX - CoV2373 n=822 Placebo n=407 NVX - CoV2373 n=383 Placebo n=187 Total 4 10 2 4 Vaccine Efficacy 80.7% (95% CI: 38.5, 93.9) 76.8% (95% CI: - 26.7, 95.7)
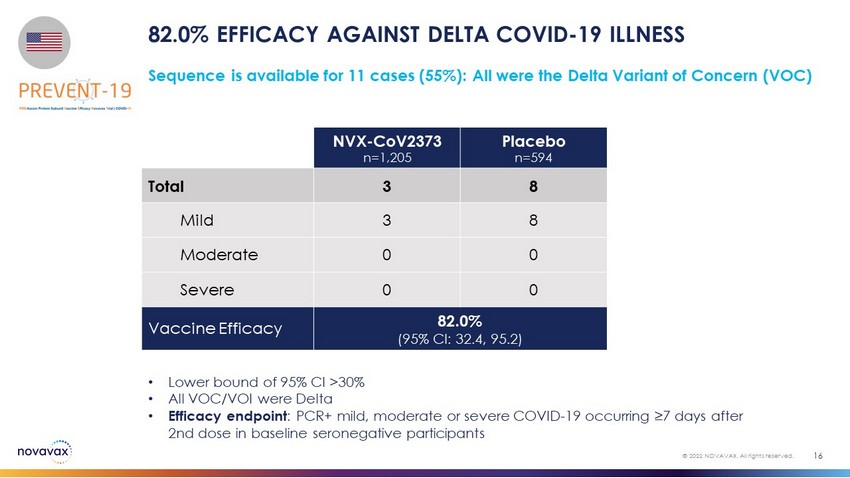
82.0% EFFICACY AGAINST DELTA COVID - 19 ILLNESS 16 © 2022 NOVAVAX. All rights reserved. • Lower bound of 95% CI >30% • All VOC/VOI were Delta • Efficacy endpoint : PCR+ mild, moderate or severe COVID - 19 occurring ≥7 days after 2nd dose in baseline seronegative participants Sequence is available for 11 cases (55%): All were the Delta Variant of Concern (VOC) NVX - CoV2373 n=1,205 Placebo n=594 Total 3 8 Mild 3 8 Moderate 0 0 Severe 0 0 Vaccine Efficacy 82.0% (95% CI: 32.4, 95.2)

SUMMARY 17 © 2022 NOVAVAX. All rights reserved. • No safety signal observed in preliminary dataset up to crossover • Vaccine was well tolerated • Reactogenicity was generally similar or lower magnitude and severity compared to adult part of the study • Overall IgG and wild - type neutralization responses higher than observed in adults • IgG and hACE2 - inhibition against variants also higher than observed in adults • Achieved Primary Effectiveness endpoint with neutralizing antibody responses non - inferior to young adults • Adolescent neutralization responses ~1.5 - fold higher than adults • Efficacy against symptomatic Delta variant infection 82.0% (95% CI: 32.4, 95.2 ) • Overall efficacy 79.5% (95% CI: 46.8, 92.1) • Achieved statistical success criteria with lower bound of 95% CI >30% • All illness in this dataset was mild, often more difficult to prevent with vaccination • Small number of cases resulted in broad confidence internal which overlap with adult part of study ( VE = 90.4% (95% CI: 82.8,94.6) • Findings consistent between 12 to <15 y/o and 15 to <18 y/o

NEXT STEPS IN PEDIATRIC PROGRAM 18 © 2022 NOVAVAX. All rights reserved. • Supplement Global Filings with Pediatric Data: Q1 2022 • Pediatric investigations plan: Agreed to by FDA, MHRA, EMA • Subsequent pediatric study planned for Q2 2022
