Exhibit 99.1

Novavax’ NanoFlu Achieves All Primary Endpoints In
Phase 3 Clinical Trial
| · | Trial also achieves statistical significance in key secondary endpoints |
| · | Novavax to submit a U.S. BLA under FDA’s accelerated approval pathway |
| · | Company to host investor conference call today at 8:30 a.m. EDT |
Gaithersburg, MD, March 24, 2020 – Novavax, Inc. (NASDAQ: NVAX), a late-stage biotechnology company developing next-generation vaccines for serious infectious diseases, today announced positive top-line results of its pivotal Phase 3 clinical trial of NanoFlu™, its recombinant quadrivalent seasonal influenza vaccine candidate with its proprietary Matrix-M™ adjuvant, in adults aged 65 and older. Using the Food and Drug Administration’s (FDA) criteria for accelerated approval of seasonal influenza vaccines, the trial evaluated the immunogenicity and safety of NanoFlu compared to Fluzone® Quadrivalent, a U.S.-licensed quadrivalent influenza vaccine.
NanoFlu Phase 3 Primary Objectives
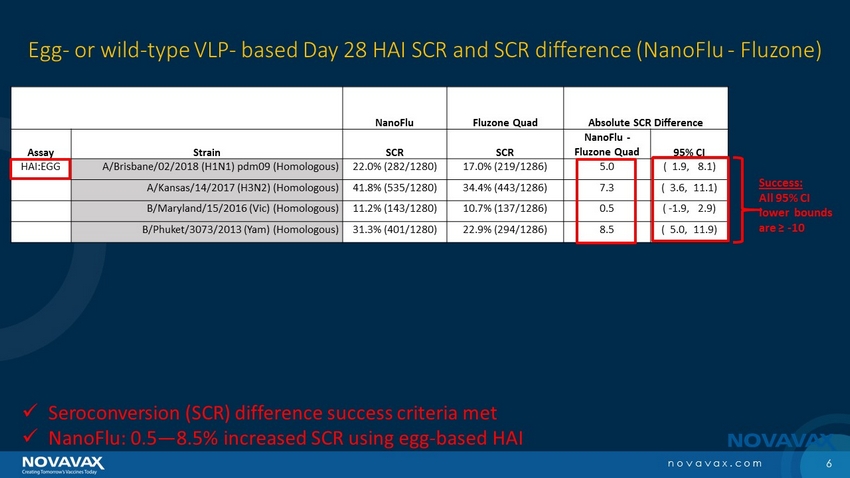
The trial’s primary objectives were to demonstrate non-inferior immunogenicity of NanoFlu compared to Fluzone Quadrivalent using the day 28 ratio of geometric mean titers (GMT) and the difference in seroconversion rates (SCR), as well as the overall safety of NanoFlu. Immunogenicity was measured by hemagglutination inhibition (HAI) assays using egg-derived reagents.
| · | NanoFlu achieved the primary endpoints, both GMT and SCR, for all four strains included in the vaccine. |
| · | NanoFlu was well-tolerated and had a safety profile comparable to Fluzone Quadrivalent with a modest increase in local adverse events (AEs). |
“With these data, we now have a clear path forward to licensure with our differentiated recombinant influenza vaccine,” said Stanley C. Erck, President and Chief Executive Officer of Novavax. “These strong Phase 3 results align with and validate our previous clinical trials, in which NanoFlu showed higher HAI antibody responses than the leading flu vaccine for older adults. We expect that both Fast Track designation and the accelerated approval pathway from the FDA will help Novavax bring NanoFlu to market as quickly as possible to address the serious public health threat of influenza.”
NanoFlu Phase 3 Secondary Objectives
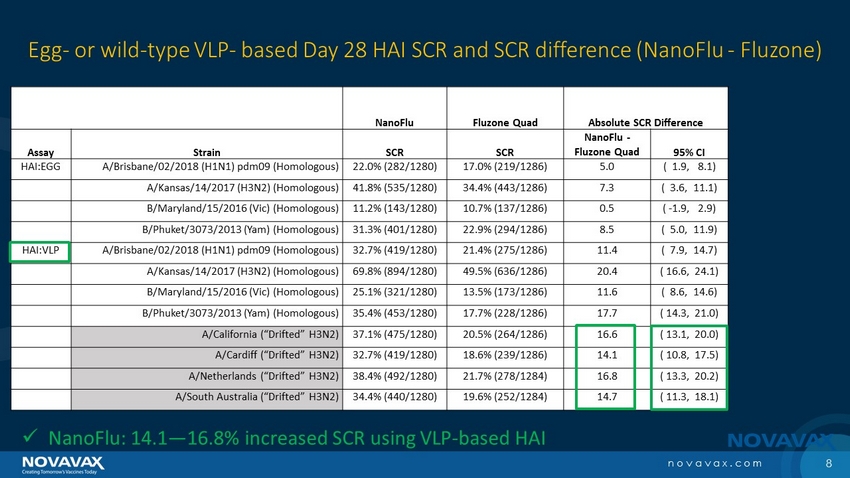
The trial’s key secondary endpoints assessed GMT and SCR, but with an HAI assay based on wild-type reagents, which are expected to provide a more accurate assessment of clinically relevant HAI antibody responses against circulating wild-type viruses.
| · | NanoFlu demonstrated significantly higher GMT and SCR than Fluzone Quadrivalent across all four strains included in the vaccine. |
| · | 24%-66% higher GMT responses; and |
| · | 11.4-20.4 higher SCR percentage points. |
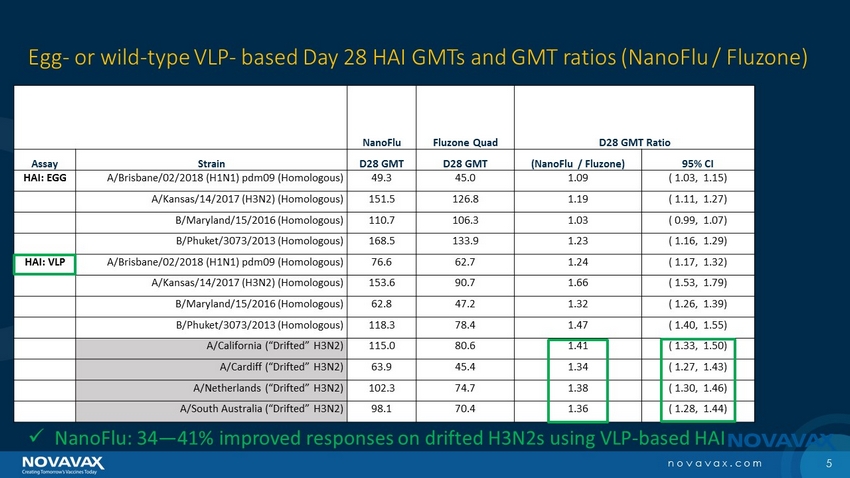
| · | NanoFlu also demonstrated significantly higher GMT and SCR than Fluzone Quadrivalent for four tested drifted H3N2 strains not included in the vaccine but circulating this year. |
| · | 34%-41% higher GMT responses; and |
| · | 14.1-16.8 higher SCR percentage points |
“In addition to meeting the primary objectives, we are very pleased to report that NanoFlu also met or exceeded its secondary endpoints for all four strains using our proprietary HAI assay based upon wild-type reagents,” said Gregory Glenn, M.D., President of Research and Development of Novavax. “NanoFlu demonstrated significant improvement against four drifted H3N2 strains that are co-circulating this year. These data, similar to what was shown in our Phase 2 clinical trial, demonstrate that NanoFlu overcomes issues related to egg-adaptation and antigenic drift. We extend our sincere appreciation to those who volunteered for this important study and to our clinical partners who worked so quickly and diligently on this trial.”
Webcast Conference Call
Novavax will host a webcast/conference call today at 8:30 a.m. ET. The webcast can be accessed via a link on the home page of the Novavax website (novavax.com) or through the “Investor Info”/“Events” tab on the Novavax website. Listeners who wish to ask questions or don’t have internet access can dial-in to the conference call at (877) 212-6076 (domestic) or (707) 287-9331 (international) and use passcode 4498251.
A replay of the webcast will be available on the Novavax website until June 24, 2020 and a replay of the conference call only will be available starting at 11:30 a.m. ET on March 24, 2020 until 11:30 a.m. ET on March 31, 2020. To access the conference call replay, dial (855) 859-2056 (domestic) or (404) 537-3406 (international) and use passcode 4498251.
About Phase 3 Clinical Trial
The trial enrolled 2,652 healthy older adults across 19 U.S. clinical sites. Trial participants received either NanoFlu or the comparator, both of which were formulated with the four influenza strains recommended for the 2019-2020 Northern hemisphere influenza season. Participants will be followed for approximately one year after injection, with primary immunogenicity analyses of the Day 28 sera samples.
About NanoFlu™ and Matrix-M™
NanoFlu is a recombinant hemagglutinin (HA) protein nanoparticle influenza vaccine produced by Novavax in its SF9 insect cell baculovirus system. NanoFlu uses HA amino acid protein sequences that are the same as the recommended wild-type circulating virus HA sequences. The qNIV vaccine antigens were derived from A/Brisbane 02/2018 H1N1, A/Kansas 14/2017 H3N2, B/Maryland 15/2016 and B/Phuket 3073/2013. NanoFlu contains Novavax’ patented saponin-based Matrix-M adjuvant, which has demonstrated a potent and well-tolerated effect by stimulating the entry of antigen-presenting cells into the injection site and enhancing antigen presentation in local lymph nodes.
About Novavax
Novavax, Inc. (Nasdaq:NVAX), is a late-stage biotechnology company that promotes improved health globally through the discovery, development, and commercialization of innovative vaccines to prevent serious infectious diseases. NanoFlu™, its quadrivalent influenza nanoparticle vaccine, met primary and secondary endpoints in its pivotal Phase 3 clinical trial. ResVax™, its RSV vaccine for infants via maternal immunization, is the only vaccine to demonstrate efficacy in a Phase 3 clinical trial. Novavax recently initiated development of a vaccine program against COVID-19. Novavax is a leading innovator of recombinant vaccines; its proprietary recombinant technology platform combines the power and speed of genetic engineering to efficiently produce a new class of highly immunogenic nanoparticles addressing urgent global health needs.
For more information, visit www.novavax.com and connect with us on Twitter and LinkedIn.
Forward-Looking Statements
Statements herein relating to the future of Novavax and the ongoing development of its vaccine and adjuvant products are forward-looking statements. Novavax cautions that these forward-looking statements are subject to numerous risks and uncertainties, which could cause actual results to differ materially from those expressed or implied by such statements. These risks and uncertainties include those identified under the heading “Risk Factors” in the Novavax Annual Report on Form 10-K for the year ended December 31, 2019, as filed with the Securities and Exchange Commission (SEC). We caution investors not to place considerable reliance on the forward-looking statements contained in this press release. You are encouraged to read our filings with the SEC, available at sec.gov, for a discussion of these and other risks and uncertainties. The forward-looking statements in this press release speak only as of the date of this document, and we undertake no obligation to update or revise any of the statements. Our business is subject to substantial risks and uncertainties, including those referenced above. Investors, potential investors, and others should give careful consideration to these risks and uncertainties.
Contacts:
Investors
Novavax, Inc.
Erika Trahan
ir@novavax.com
240-268-2022
Westwicke
John Woolford
john.woolford@westwicke.com
443-213-0506
Media
Brandzone/KOGS Communication
Edna Kaplan
kaplan@kogspr.com
617-974-8659